30.6: Some Nitrogen-Containing Natural Products
- Page ID
- 22406
Alkaloids
Basic nitrogen compounds in plants are classified as alkaloids. Several examples were given previously of this large and remarkably heterogeneous class of compounds, many of which have very complex structures (Section 23-2). It is difficult to give a coherent account of alkaloid chemistry in the limited space available to us here.
The biosynthesis of alkaloids has been extensively studied, and although for a time it was thought that alkaloids arose primarily from amino acid precursors, strong evidence now is available that ethanoate also is involved. The mode of alkaloid biosynthesis is not yet as well understood as that of the terpenes and steroids. One experimental problem is the difficulty of feeding suitably labeled precursors to plants.
Vitamin B\(_{12}\)
Vitamin B\(_{12}\) is the nutritional factor required for the prevention of pernicious anemia. Its structure was determined in 1956 through the chemical studies of Alexander Todd (Nobel Prize, 1957) and the x-ray diffraction studies of Dorothy Hodgkin (Nobel Prize, 1964). It is one of the most complex natural products known, yet it has features that are not unfamiliar. It is related to the metalloporphyrins discussed previously (Section 25-8B), but the ring system surrounding the cobalt atom has one less carbon bridging two of the nitrogen-containing rings than the porphyrin ring of heme or chlorophyll. The B\(_{12}\) ring system is called a corrin ring, and the vitamin is a cobalt-corrin complex.
The corrin ring includes methyl, ethanamide, and propanamide groups, and one of these is linked through a nucleotide residue to the cobalt atom. There are five nitrogen ligands around the cobalt, and a sixth ligand is attached through carbon - here a cyano group - so that an alternate name for vitamin B\(_{12}\) is cyanocobalamin:
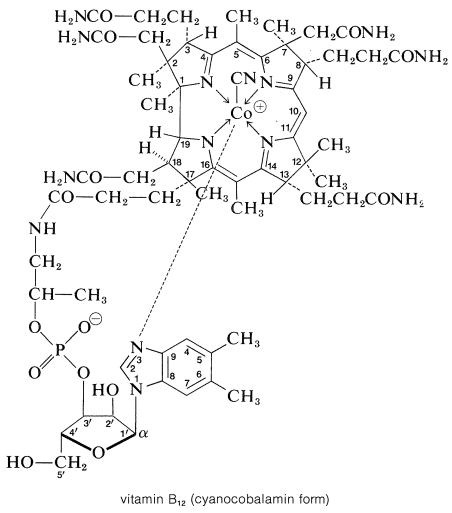
A total synthesis of vitamin B\(_{12}\) was announced in 1972, as the result of a collaborative effort between R. B. Woodward (Harvard) and A. Eschenmoser (Zurich). The synthesis was completed after 11 years of effort involving 100 co-workers from 19 countries. A number of important techniques and reactions of synthetic value were developed during the course of this work, including the principle of conservation of orbital symmetry (the Woodward-Hoffman rules, Section 21-10). The biochemical action of vitamin B\(_{12}\) is considered in Chapter 31.
Penicillins and Cephalosporins
The first antibiotics of medicinal value were discovered by Alexander Fleming in 1929 as metabolites of the microorganism Penicillium notatum. They became known as penicillins, but their development as useful drugs was slow in coming. However, the urgent need for nontoxic antibiotics was recognized during World War II, and resulted in a team effort by English and American scientists to develop efficient methods for preparing penicillin by fermentation and to undertake clinical and chemical studies. By 1943, penicillin was available in quantity for the treatment of war wounded. By 1945, the basic structure and stereochemistry was deduced through chemical degradation and x-ray diffraction studies:
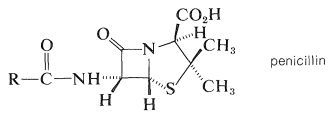
The structure is unusual in that it has a four-membered cyclic amide ring (\(\beta\)-lactam). It was the first example to be discovered of a natural product with this ring structure.
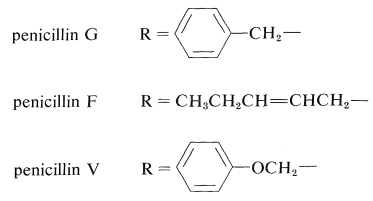
Fermentation can produce penicillins that differ only in the nature of the side-chain group \(\ce{R}\). The common natural penicillin is penicillin G, in which \(\ce{R} \: =\) phenylmethyl (benzyl):
The cephalosporins are antibiotics produced by the bacterial strain cephalosporium. They are closely related to the penicillins. Thus cephalosporin C has a \(\beta\)-lactam ring but a six-membered sulfur-containing ring:
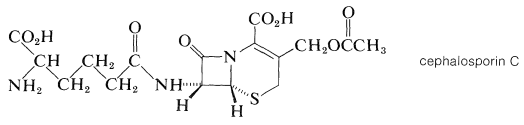
Both the cephalosporins and the penicillins owe their antibacterial action to their ability to block bacterial cell-wall biosynthesis. Cephalosporin C is less active than the penicillins, but is less susceptible to enzymatic destruction by \(\beta\)-lactamases, which are enzymes that cleave the lactam ring. In fact, the so-called resistance of staph bacteria to penicillins is attributed to the propagation of strains that produce \(\beta\)-lactamase. Numerous semisynthetic penicillins and cephalosporins have been made in the hope of finding new broad-spectrum antibiotics with high activity but with greater \(\beta\)-lactam stability. Several of these are in clinical use.
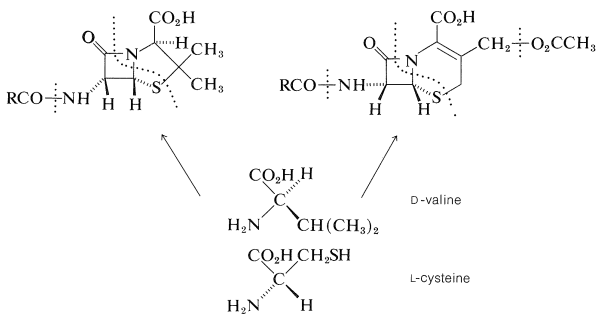
The total synthesis of penicillin V was achieved by J. C. Sheehan (1957) and of cephalosporin by R. B. Woodward (1966). Biosynthetic routes have been worked out in part, and the precursors to both ring systems are \(L\)-cysteine and \(D\)-valine:
Contributors and Attributions
John D. Robert and Marjorie C. Caserio (1977) Basic Principles of Organic Chemistry, second edition. W. A. Benjamin, Inc. , Menlo Park, CA. ISBN 0-8053-8329-8. This content is copyrighted under the following conditions, "You are granted permission for individual, educational, research and non-commercial reproduction, distribution, display and performance of this work in any format."


