24.2: Amides as Acids and Bases
- Page ID
- 22351
Acidity
Amides with \(\ce{N-H}\) bonds are weakly acidic, the usual \(K_a\) being about \(10^{-16}\):
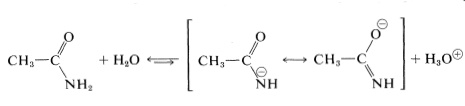
Nonetheless, amides clearly are far more acidic than ammonia \(\left( K_a \sim 10^{-33} \right)\), and this difference reflects a substantial degree of stabilization of the amide anion. However, amides still are very weak acids (about as weak as water) and, for practical purposes, are regarded as neutral compounds.
Where there are two carbonyl groups to stabilize the amide anion, as in the 1,2-benzenedicarboximide (phthalimide) anion (Section 18-10C), the acidity increases markedly and imides can be converted to their conjugate bases with concentrated aqueous hydroxide ion. We have seen how imide salts can be used for the synthesis of primary amines. (Gabriel synthesis, Section 23-9D and Table 23-6).
Basicity
The degree of basicity of amides is very much less than that of aliphatic amines. For ethanamide, \(K_b\) is about \(10^{-15}\) (\(K_a\) of the conjugate acid is \(\sim 10\)):
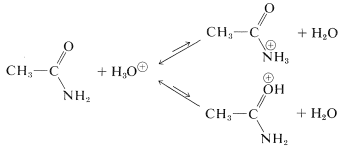
The proton can become attached either to nitrogen or to oxygen, and the choice between the assignments is not an easy one. Of course, nitrogen is intrinsically more basic than oxygen; but formation of the \(\ce{N}\)-conjugate acid would cause loss of all the amide stabilization energy. Addition to oxygen actually is favored, but amides are too weakly basic for protonation to occur to any extent in water solution.
Contributors and Attributions
John D. Robert and Marjorie C. Caserio (1977) Basic Principles of Organic Chemistry, second edition. W. A. Benjamin, Inc. , Menlo Park, CA. ISBN 0-8053-8329-8. This content is copyrighted under the following conditions, "You are granted permission for individual, educational, research and non-commercial reproduction, distribution, display and performance of this work in any format."


