24.3: Synthesis of Amides
- Page ID
- 22352
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)From Carboxylic Acids
Formation of amides from carboxylic acid derivatives already has been discussed in some detail (Section 23-9A):
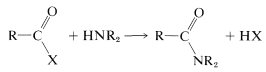 \(\tag{24-1}\)
\(\tag{24-1}\)
The ease of formation of amides by the reaction of Equation 24-1 depends a lot on the nature of the leaving group \(\ce{X}\). The characteristics of a good leaving group were discussed in Sections 8-7C and 8-7D in connection with \(S_\text{N}\) reactions, and similar considerations apply here. Some idea of the range of acid derivatives used in amide synthesis can be obtained from Table 24-1, which lists various \(\ce{RCOX}\) compounds and the p\(K_a\) values of \(\ce{HX}\). As a reasonable rule of thumb, the stronger \(\ce{HX}\) is as an acid, the better \(\ce{X}\) is as a leaving group.
Table 24-1: Derivatives and Reactivity of Carboxylic Acids Commonly Used in Amide Formation
\[\ce{RCOX} + \ce{H_2NR'} \rightarrow \ce{RCONHR'} + \ce{HX}\]
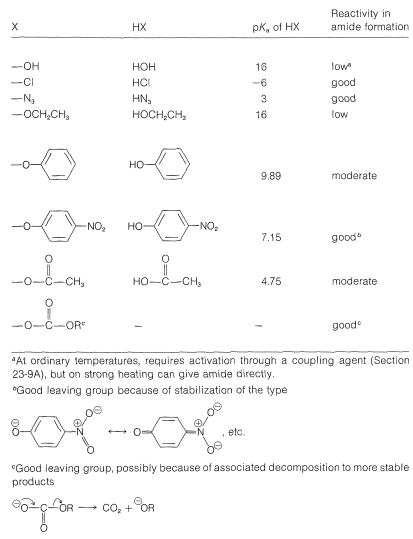
Amides generally are formed from acid chlorides, acid azides, acid anhydrides, and esters. It is not practical to prepare them directly from an amine and a carboxylic acid without strong heating or unless the reaction is coupled to a second reaction that "activates" the acid. Notice that esters of phenols are more reactive toward amines than esters of alcohols because phenols are stronger acids than alcohols.
From Nitriles
The hydrolysis of nitriles is a satisfactory method for preparation of unsubstituted amides and is particularly convenient when hydrolysis is induced under mildly basic conditions by hydrogen peroxide:
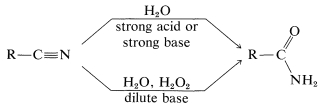
For the preparation of amides of the type \(\ce{R_3CNHCOR}\), which have a tertiary alkyl group bonded to nitrogen, the Ritter reaction of an alcohol or alkene with a nitrile or hydrogen cyanide is highly advantageous. This reaction involves formation of a carbocation by action of strong sulfuric acid on an alkene or an alcohol (Equation 24-2), combination of the carbocation with the unshared electrons on nitrogen of \(\ce{RCN}\) (Equation 24-3), and then addition of water (Equation 24-4). We use here the preparation of an \(\ce{N}\)-tert-butylalkanamide as an example; \(\ce{RC \equiv N}\) can be an alkyl cyanide such as ethanenitrile or hydrogen cyanide itself:
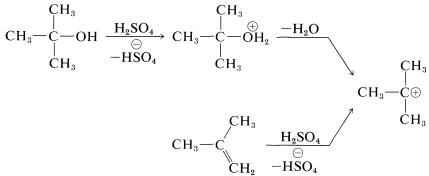 \(\tag{24-2}\)
\(\tag{24-2}\)
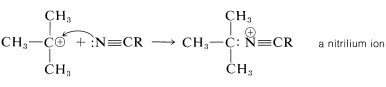 \(\tag{24-3}\)
\(\tag{24-3}\)
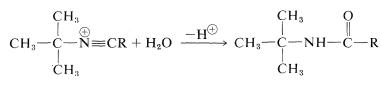 \(\tag{24-4}\)
\(\tag{24-4}\)
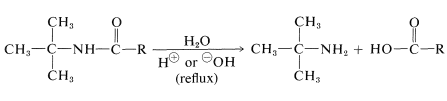
This reaction also is useful for the preparation of primary amines by hydrolysis of the amide. It is one of the relatively few practical methods for synthesizing amines with a tertiary alkyl group on the nitrogen:
The Beckmann Rearrangement of Oximes
You may recall that ketones react with \(\ce{RNH_2}\) compounds to give products with a double bond to nitrogen, \(\ce{-C=NR}\) (Section 16-4C). When the \(\ce{RNH_2}\) compound is azanol (hydroxylamine), \(\ce{HO-NH_2}\), the product is called a ketoxime, or oxime:
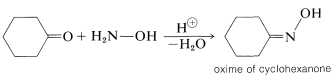
Oximes rearrange when heated with a strong acid, and this reaction provides a useful synthesis of amides:
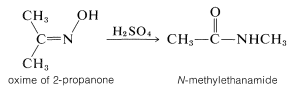
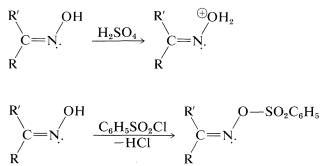
This intriguing reaction is known as the Beckmann rearrangement. It has been the subject of a number of mechanistic studies that have shown the acid or acid halide (\(\ce{PCl_3}\), \(\ce{C_6H_5SO_2Cl}\)) makes the hydroxyl group on nitrogen into a better leaving group by forming \(\ce{-OH_2^+}\) or ester intermediates:
Thereafter, a rearrangement occurs resembling the reactions of carbocations (Sections 8-9B and 15-5E). When the cleavage of the \(\ce{N-O}\) bond occurs, the nitrogen atom would be left with only six valence electrons. However, as the bond breaks, a substituent \(\ce{R}\) on the neighboring carbon moves with its bonding electron pair to the developing positive nitrogen (Equation 24-5):
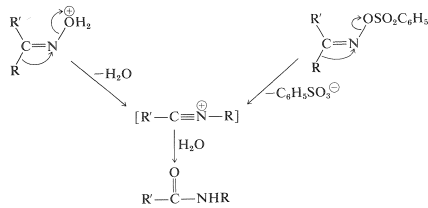 \(\tag{24-5}\)
\(\tag{24-5}\)
Oximes with \(\ce{R}\) and \(\ce{R'}\) as different groups exist as \(E\) and \(Z\) isomers (Section 19-7) and you will notice in Equation 24-5 that the group that migrates is the one that is trans to the leaving group. To some extent the Beckmann rearrangement is an internal \(S_\text{N}2\) reaction with inversion at the nitrogen. Section 21-10F gives a theoretical treatment of this kind of reaction. The rearrangement product is a nitrilium ion, as in the Ritter reaction (Section 24-3B), which adds water to form the amide.
The synthesis of aza-2-cycloheptanone (\(\varepsilon\)-caprolactam) by the Beckmann rearrangement of the oxime of cyclohexanone is of commercial importance because the lactam is an intermediate in the synthesis of a type of nylon (a polyamide called "nylon-6"\(^2\)):
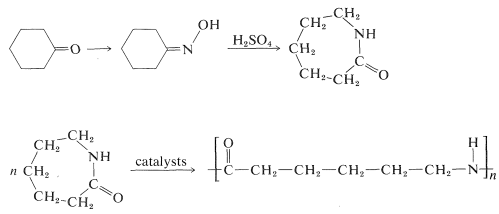
\(^2\)The number 6 specifies the number of carbons in each monomer unit comprising the polyamide structure. By this code, nylon-6,6 is \(\ce{(-NH(CH_2)_6NHCO(CH_2)_4CO-)}_n\).
Contributors and Attributions
John D. Robert and Marjorie C. Caserio (1977) Basic Principles of Organic Chemistry, second edition. W. A. Benjamin, Inc. , Menlo Park, CA. ISBN 0-8053-8329-8. This content is copyrighted under the following conditions, "You are granted permission for individual, educational, research and non-commercial reproduction, distribution, display and performance of this work in any format."


