17.12: Cyclopropanones and Cyclopropenones
- Page ID
- 22286
Cyclopropanones deserve special comment, not because of their practical importance (they have no commercial value at this time), but because of their novel behavior and reactivity. No unambiguous synthesis of cyclopropanones was known prior to 1965, and the older textbooks usually contained statements such as "cyclopropanones apparently cannot exist". However, they had been postulated as intermediates in various reactions (see, for example, the Favorskii rearrangement, Section 17-2C), but until recently had defied isolation and identification. The problem is that the three-ring ketone is remarkably reactive, especially towards nucleophiles. Because of the associated relief of angle strain, nucleophiles readily add to the carbonyl group without the aid of a catalyst and give good yields of adducts from which the cyclopropanone is not easily recovered:
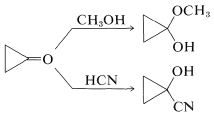
To avoid destructive side reactions, cyclopropanones have to be prepared at low temperatures in the absence of nucleophiles. A good example is the synthesis of cyclopropanone itself from ketene and diazomethane (see Section 16-4A):
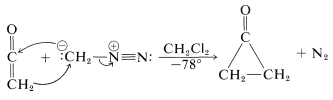
When seemingly simple organic structures defy isolation, this usually stimulates many theoretical and experimental studies in an effort to rationalize anomalous behavior. In the case of cyclopropanone, the possibility was considered that the molecule might preferably exist as an open-chain dipolar structure rather than as the cyclic ketone:
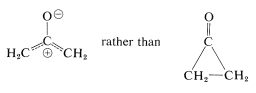
Although the spectral properties of cyclopropanones and the easy formation of hydrates and hemiketals are inconsistent with the dipolar form, some reactions of cyclopropanones do indicate that the ring carbons are much more electrophilic than in other cyclic or acyclic ketones. For example, nucleophilic ring opening often occurs easily:
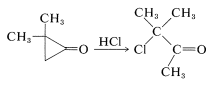
Also, both [3 + 4] cycloadditions of cyclopropanone to dienes and [3 + 2] additions to carbonyl groups have been observed. These reactions seem easiest to understand if cyclopropanone can behave as if it had, or could be converted to, a dipolar open-chain structure:
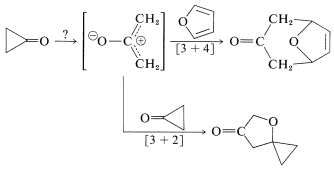
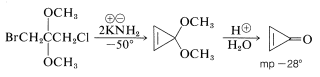
Cyclopropenone has been prepared by a route that illustrates the value of the acetal grouping in protecting ketone groups (Section 16-8):
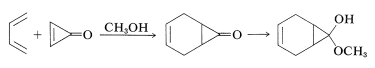
Cyclopropenone undergoes many interesting reactions - one example is Diels-Alder addition, the product of which in methanol solution is a hemiketal. That the hemiketal is favored for the adduct, but not for cyclopropenone, indicates that the double bond of cyclopropenone has a considerable effect on the reactivity of the carbonyl group.
Contributors and Attributions
John D. Robert and Marjorie C. Caserio (1977) Basic Principles of Organic Chemistry, second edition. W. A. Benjamin, Inc. , Menlo Park, CA. ISBN 0-8053-8329-8. This content is copyrighted under the following conditions, "You are granted permission for individual, educational, research and non-commercial reproduction, distribution, display and performance of this work in any format."


