22.2: Poisons and How They Act
- Page ID
- 152296
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Describe the modes of action and treatments for different poisons.
Corrosive Substances
A corrosive substance is one that will damage or destroy other substances with which it comes into contact by means of a chemical reaction. Corrosives are different from poisons in that corrosives are immediately dangerous to the tissues they contact, whereas poisons may have systemic toxic effects that require time to become evident. Colloquially, corrosives may be called poisons but the concepts are technically distinct. However, there is nothing which precludes a corrosive from being a poison; there are substances that are both corrosives and poisons.
There are several household products that contain chemical substances which are considered highly reactive and thus are very dangerous upon ingestion or skin contact. Chemical poisoning with corrosive agents occurs by ingestion of: acids (hydrochloric, nitric, sulfuric, perchloric, chloric), alkalis (sodium and potassium, soaps, detergents), heavy metal salts (sublimate), strong oxidizing agents, formalin, iodine tincture and many other chemical substances. Strong acids or bases, readily decomposes proteins and lipids through amide and ester hydrolysis upon contact with living tissues, such as skin and flesh.
Oxidizing Agents as Poisons
Household cleaning products with strong oxidizing agents like sodium hypochlorite (NaOCl) and hydrogen peroxide (H2O2) are dangerous when ingested. Generally speaking, the ingestion of bleaches (Figure \(\PageIndex{2}\)) will cause damage to the esophagus and stomach, possibly leading to death. On contact with the skin or eyes, it causes irritation, drying, and potentially burns. Inhalation of bleach fumes can damage the lungs. Personal protective equipment should always be used when using bleach. Bleach should never be mixed with vinegar or other acids as this will create highly toxic chlorine gas and can cause severe burns internally and externally. Mixing bleach with ammonia similarly produces toxic chloramine gas, which can burn the lungs. Mixing bleach with hydrogen peroxide results in an exothermic chemical reaction that releases oxygen, and may cause the contents to splatter and cause skin and eye injury. Heating bleach and boiling it may produce chlorates, a strong oxidizer which may lead to a fire or explosion.

Ozone. For the last few decades, scientists studied the effects of acute and chronic ozone exposure on human health. Hundreds of studies suggest that ozone is harmful to people at levels currently found in urban areas. Ozone has been shown to affect the respiratory, cardiovascular and central nervous system. Early death and problems in reproductive health and development are also shown to be associated with ozone exposure.
Acute ozone exposure ranges from hours to a few days. Because ozone is gas, it directly affects the lungs and the entire respiratory system. Inhaled ozone causes inflammation and acute -but reversible- changes in lung function, as well as airway hyperresponsiveness. These changes lead to shortness of breath, wheezing, and coughing which may exacerbate lung diseases, like asthma or chronic obstructive pulmonary disease (COPD) resulting in the need to receive medical treatment. Acute and chronic exposure to ozone has been shown to cause an increased risk of respiratory infections, due to the following mechanism.
Multiple studies have been conducted to determine the mechanism behind ozone's harmful effects, particularly in the lungs. These studies have shown that exposure to ozone causes changes in the immune response within the lung tissue, resulting in disruption of both the innate and adaptive immune response, as well as altering the protective function of lung epithelial cells. It is thought that these changes in immune response and the related inflammatory response are factors that likely contribute to the increased risk of lung infections, and worsening or triggering of asthma and reactive airways after exposure to ground-level ozone pollution.
Inhaling ozone not only affects the immune system and lungs, but it may also affect the heart as well. Ozone causes short-term autonomic imbalance leading to changes in heart rate and reduction in heart rate variability; and high levels exposure for as little as one-hour results in a supraventricular arrhythmia in the elderly, both increase the risk of premature death and stroke. Ozone may also lead to vasoconstriction resulting in increased systemic arterial pressure contributing to increased risk of cardiac morbidity and mortality in patients with pre-existing cardiac diseases.
Metabolic Poisons
Carbon monoxide and cyanide are chemical substances that interfere with the normal bodily functions i.e. during the delivery of oxygen to bodily tissues and cellular respiration.
Carbon Monoxide Poisoning
Carbon monoxide poisoning typically occurs from breathing in carbon monoxide (CO) at excessive levels. Symptoms (Figure \(\PageIndex{2}\)) are often described as "flu-like" and commonly include headache, dizziness, weakness, vomiting, chest pain, and confusion. Large exposures can result in loss of consciousness, arrhythmias, seizures, or death. The classically described "cherry red skin" rarely occurs. Long-term complications may include chronic fatigue, trouble with memory, and movement problems.
CO is a colorless and odorless gas which is initially non-irritating. It is produced during incomplete burning of organic matter. This can occur from motor vehicles, heaters, or cooking equipment that run on carbon-based fuels. Carbon monoxide primarily causes adverse effects by combining with hemoglobin to form carboxyhemoglobin (HbCO) preventing the blood from carrying oxygen and expelling carbon dioxide as carbaminohemoglobin. Additionally, many other hemoproteins such as myoglobin, Cytochrome P450, and mitochondrial cytochrome oxidase are affected, along with other metallic and non-metallic cellular targets.
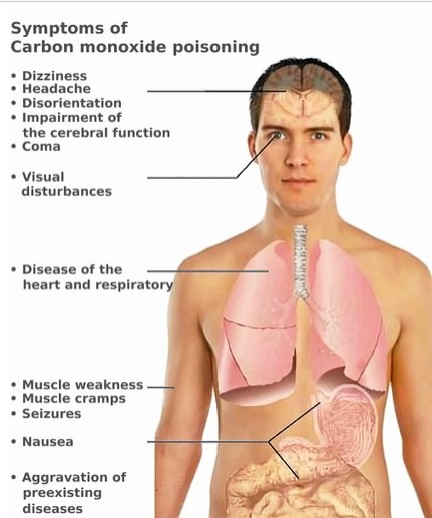
Initial treatment for carbon monoxide poisoning is to immediately remove the person from the exposure without endangering further people. Those who are unconscious may require CPR on site. Administering oxygen via non-rebreather mask shortens the half-life of carbon monoxide from 320 minutes, when breathing normal air, to only 80 minutes. Oxygen hastens the dissociation of carbon monoxide from carboxyhemoglobin, thus turning it back into hemoglobin. Due to the possible severe effects in the baby, pregnant women are treated with oxygen for longer periods of time than non-pregnant people.
Cyanide Poisoning
Cyanide poisoning is poisoning that results from exposure to a number of forms of cyanide. Early symptoms include headache, dizziness, fast heart rate, shortness of breath, and vomiting. This may then be followed by seizures, slow heart rate, low blood pressure, loss of consciousness, and cardiac arrest. Onset of symptoms is usually within a few minutes. If a person survives, there may be long-term neurological problems.
Toxic cyanide-containing compounds include hydrogen cyanide gas and a number of cyanide salts. Poisoning is relatively common following breathing in smoke from a house fire. Other potential routes of exposure include workplaces involved in metal polishing, certain insecticides, and the medication nitroprusside. Liquid forms of cyanide can be absorbed through the skin. Cyanide ions interfere with cellular respiration, resulting in the body's tissues being unable to use oxygen.
In addition to its uses as a pesticide and insecticide, cyanide is contained in tobacco smoke and smoke from building fires, and is present in many seeds or kernels such as those of almonds, apricots, apples, oranges, and in foods including cassava (also known as tapioca, yuca or manioc), and bamboo shoots. Vitamin B12, in the form of hydroxocobalamin (also spelled hydroxycobalamin), may reduce the negative effects of chronic exposure, and a deficiency can lead to negative health effects following exposure. Flaxseed also contains cyanogenic glycosides.
Cyanide is a potent cytochrome c oxidase (COX, a.k.a Complex IV) inhibitor. As such, cyanide poisoning is a form of histotoxic hypoxia, because it interferes with an essential step in aerobic metabolism called oxidative phosphorylation.
The United States standard cyanide antidote kit first uses a small inhaled dose of amyl nitrite, followed by intravenous sodium nitrite, followed by intravenous sodium thiosulfate. Hydroxocobalamin is newly approved in the US and is available in Cyanokit antidote kits. Sulfanegen TEA, which could be delivered to the body through an intra-muscular (IM) injection, detoxifies cyanide and converts the cyanide into thiocyanate, a less toxic substance. Alternative methods of treating cyanide intoxication are used in other countries.
Arsenic Poisoning
Arsenic poisoning is a medical condition that occurs due to elevated levels of arsenic in the body. If arsenic poisoning occurs over a brief period of time, symptoms may include vomiting, abdominal pain, encephalopathy, and watery diarrhea that contains blood. Long-term exposure can result in thickening of the skin, darker skin, abdominal pain, diarrhea, heart disease, numbness, and cancer.
The most common reason for long-term exposure is contaminated drinking water. Groundwater most often becomes contaminated naturally; however, contamination may also occur from mining or agriculture. It may also be found in the soil and air. Recommended levels in water are less than 10–50 µg/L (10–50 parts per billion). Other routes of exposure include toxic waste sites and traditional medicines. Most cases of poisoning are accidental. Arsenic acts by changing the functioning of around 200 enzymes. Diagnosis is by testing the urine, blood, or hair.
Prevention is by using water that does not contain high levels of arsenic. This may be achieved by the use of special filters or using rainwater. There is not good evidence to support specific treatments for long-term poisoning. For acute poisonings treating dehydration is important. Dimercaptosuccinic acid (DMSA) or dimercaptopropane sulfonate (DMPS) may be used while dimercaprol (BAL) is not recommended. Hemodialysis may also be used.
More Information on arsenic can be found on the link below
https://www.niehs.nih.gov/health/topics/agents/arsenic/index.cfm
Heavy Metal Poisoning
Lead poisoning, also known as plumbism and saturnism, is a type of metal poisoning caused by lead in the body. The brain is the most sensitive. Symptoms may include abdominal pain, constipation, headaches, irritability, memory problems, infertility, and tingling in the hands and feet. It causes almost 10% of intellectual disability of otherwise unknown cause and can result in behavioral problems. Some of the effects are permanent. In severe cases, anemia, seizures, coma, or death may occur.
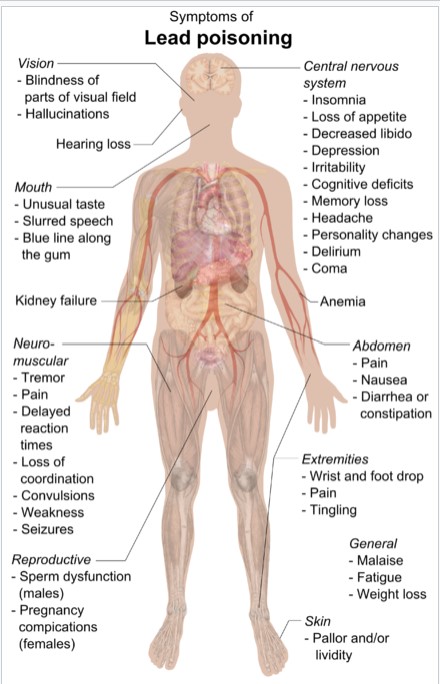
Exposure to lead can occur by contaminated air, water, dust, food, or consumer products. Children are at greater risk as they are more likely to put objects in their mouth such as those that contain lead paint and absorb a greater proportion of the lead that they eat. Exposure at work is a common cause of lead poisoning in adults with certain occupations at particular risk. Diagnosis is typically by measurement of the blood lead level. The Centers for Disease Control (US) has set the upper limit for blood lead for adults at 10 µg/dl (10 µg/100 g) and for children at 5 µg/dl. Elevated lead may also be detected by changes in red blood cells or dense lines in the bones of children as seen on X-ray.
Lead has no known physiologically relevant role in the body, and its harmful effects are myriad. Lead and other heavy metals create reactive radicals which damage cell structures including DNA and cell membranes. Lead also interferes with DNA transcription, enzymes that help in the synthesis of vitamin D, and enzymes that maintain the integrity of the cell membrane. Anemia may result when the cell membranes of red blood cells become more fragile as the result of damage to their membranes. Lead interferes with metabolism of bones and teeth and alters the permeability of blood vessels and collagen synthesis. Lead may also be harmful to the developing immune system, causing production of excessive inflammatory proteins; this mechanism may mean that lead exposure is a risk factor for asthma in children. Lead exposure has also been associated with a decrease in activity of immune cells such as polymorphonuclear leukocytes. Lead also interferes with the normal metabolism of calcium in cells and causes it to build up within them.
Lead poisoning is preventable. This includes individual efforts such as removing lead-containing items from the home, workplace efforts such as improved ventilation and monitoring, state laws that ban the use of and national policies such as laws that ban lead in products such as paint, gasoline, ammunition, wheel weights, and fishing weights reduce allowable levels in water or soil, and provide for cleanup of contaminated soil. Workers' education could be helpful as well. The major treatments are removal of the source of lead and the use of medications that bind lead so it can be eliminated from the body, known as chelation therapy. Chelation therapy in children is recommended when blood levels are greater than 40–45 µg/dl. Medications used include dimercaprol, edetate calcium disodium, and succimer.
Links to various lead and mercury related topics can be found below
Mercury poisoning is a type of metal poisoning due to exposure to mercury. Symptoms depend upon the type, dose, method, and duration of exposure. They may include muscle weakness, poor coordination, numbness in the hands and feet, skin rashes, anxiety, memory problems, trouble speaking, trouble hearing, or trouble seeing. High-level exposure to methylmercury is known as Minamata disease. Methylmercury exposure in children may result in acrodynia (pink disease) in which the skin becomes pink and peels. Long-term complications may include kidney problems and decreased intelligence. The effects of long-term low-dose exposure to methylmercury are unclear.
Forms of mercury exposure include metal, vapor, salt, and organic compound. Most exposure is from eating fish, amalgam based dental fillings, or exposure at work. In fish, those higher up in the food chain generally have higher levels of mercury. Less commonly, poisoning may occur as a method of attempted suicide. Human activities that release mercury into the environment include the burning of coal and mining of gold. Tests of the blood, urine, and hair for mercury are available but do not relate well to the amount in the body.
The primary mechanism of mercury toxicity involves its irreversible inhibition of selenoenzymes, such as thioredoxin reductase. High mercury exposures deplete the amount of cellular selenium available for the biosynthesis of thioredoxin reductase and other selenoenzymes that prevent and reverse oxidative damage, which, if the depletion is severe and long lasting, results in brain cell dysfunctions that can ultimately cause death.
Prevention includes eating a diet low in mercury, removing mercury from medical and other devices, proper disposal of mercury, and not mining further mercury. In those with acute poisoning from inorganic mercury salts, chelation with either dimercaptosuccinic acid (DMSA) or dimercaptopropane sulfonate (DMPS) appears to improve outcomes if given within a few hours of exposure. Chelation for those with long-term exposure is of unclear benefit.
Cadmium poisoning. Cadmium is a naturally occurring toxic metal with common exposure in industrial workplaces, plant soils, and from smoking. Due to its low permissible exposure in humans, overexposure may occur even in situations where trace quantities of cadmium are found. Cadmium is used extensively in electroplating, although the nature of the operation does not generally lead to overexposure. Cadmium is also found in some industrial paints and may represent a hazard when sprayed. Operations involving removal of cadmium paints by scraping or blasting may pose a significant hazard. The primary use of cadmium is in the manufacturing of NiCd rechargeable batteries. The primary source for cadmium is as a byproduct of refining zinc metal. Exposures to cadmium are addressed in specific standards for the general industry, shipyard employment, the construction industry, and the agricultural industry.
Acute exposure to cadmium fumes may cause flu-like symptoms including chills, fever, and muscle ache sometimes referred to as "the cadmium blues." Symptoms may resolve after a week if there is no respiratory damage. More severe exposures can cause tracheo-bronchitis, pneumonitis, and pulmonary edema. Symptoms of inflammation may start hours after the exposure and include cough, dryness and irritation of the nose and throat, headache, dizziness, weakness, fever, chills, and chest pain.
Inhaling cadmium-laden dust quickly leads to respiratory tract and kidney problems which can be fatal (often from kidney failure). Ingestion of any significant amount of cadmium causes immediate poisoning and damage to the liver, bones, and the kidneys. Compounds containing cadmium are also carcinogenic.
Summary
- Corrosive poisons are chemicals (that can be found in many household products) that are considered highly reactive and thus are very dangerous upon ingestion or skin contact.
- Metabolic poisons such as carbon monoxide and cyanide interfere with the bodily functions.
- Human exposure to lead, mercury, and cadmium have affected the nervous system, respiratory system, brain, and other various organs.
Contributors
- Wikipedia
- US NIH MedlinePlus
- Chibishev, Andon, et al. “Corrosive Poisonings in Adults.” Materia Socio Medica, vol. 24, no. 2, 2012, p. 125., doi:10.5455/msm.2012.24.125-130.
- Robert J. Lancashire (University of West Indies-Mona)

