14.6: Making Water Fit to Drink
- Page ID
- 152229
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Know the primary standards for various contaminants in drinking water.
- Describe the importance of the different steps in water treatment.
- Of the approximately 155,693 public water systems in the United States, 52,110 (33.5%) are community systems and 103,583 (66.5%) are noncommunity systems, including 84,744 transient systems and 18,839 nontransient systems 1.
- Over 286 million Americans get their tap water from a community water system 1.
- 8% of U.S. community water systems provide water to 82% of the U.S. population through large municipal water systems 1.
- Although the majority of community water systems (78%) are supplied by ground water, more people (68%) are supplied year-round by community water systems that use surface water .
Note: EPA. Factoids: drinking water and ground water statistics for 2007. March 2008, April 2008.
Safe Drinking Water Act
Up until 1974, public drinking water supplies in the United States were monitored and regulated by state and local authorities. Lists of contaminants with their various concentrations could vary from state to state. As the chemical industry grew, these same state agencies noted the increased presence of existing and new organic chemicals in public water systems. In order to standardize drinking water across the country, the Environmental Protection Agency (EPA) enacted the Safe Water Drinking Act of 1974.

The 1974 act enabled the EPA to monitor and regulate public water systems that serve over 25 people. Implementation and enforcement of drinking water standards would still be performed by each state. Regarding drinking water sources (surface and ground), the EPA and state agencies protect and monitor these as well. Levels of contaminants would be defined using the concentration terms Maximum Contaminant Level (MCL) and Treatment Technique (TT)
National Primary Standards for Drinking Water
The first set of drinking water standards included only 22 chemicals and/or pathogens. EPA established two major types of contaminants: primary and secondary. The first of these types (primary) contaminants are substances (examples could include Hg, As, and U) that can be toxic in small amounts. On the other hand, secondary contaminants are less toxic species (Fe and Zn) and would include cosmetic issues (color, taste, and odor) of drinking water.
All primary contaminants have enforceable concentration values. For the majority of these pollutants, EPA lists concentration limits by using the term Maximum Contaminant Level (MCL). If a water supplier exceeds a given MCL for a toxin, then fines and penalties could by imposed by the EPA. A few pathogens (Giardia Lamblia and Legionella) use Treatment Technique (TT) notation rather than numerical MCL concentrations. Water that contains any amount of these pathogens must be sanitized immediately with a standardized EPA procedure.
A detailed list of acceptable levels of different contaminants by EPA classification can be found on the the link below.
https://www.epa.gov/ground-water-and-drinking-water/national-primary-drinking-water-regulations
The complete table of standards is given below.
https://www.epa.gov/sites/production/files/2016-06/documents/npwdr_complete_table.pdf
| Contaminant | MCLG1(mg/L)2 | MCL or TT1(mg/L)2 | Potential Health Effects from Long-Term Exposure Above the MCL (unless specified as short-term) | Sources of Contaminant in Drinking Water |
|---|---|---|---|---|
|
Microorganisms- Cryptosporidium |
zero | TT3 | Gastrointestinal illness (such as diarrhea, vomiting, and cramps) | Human and animal fecal waste |
|
Microorganisms- Giardia lamblia |
zero | TT | Gastrointestinal illness (such as diarrhea, vomiting, and cramps) | Human and animal fecal waste |
|
Microorganisms- Total Coliforms (including fecal coliform and E. Coli) |
zero | 5.0% | Not a health threat in itself; it is used to indicate whether other potentially harmful bacteria may be present5 | Coliforms are naturally present in the environment; as well as feces; fecal coliforms and E. coli only come from human and animal fecal waste. |
| Viruses (enteric) | zero | TT | Gastrointestinal illness (such as diarrhea, vomiting, and cramps) | Human and animal fecal waste |
|
Inorganic Chemicals- Antimony |
0.006 | 0.006 | Increase in blood cholesterol; decrease in blood sugar | Discharge from petroleum refineries; fire retardants; ceramics; electronics; solder |
|
Inorganic Chemicals- Chromium (total) |
0.1 | 0.1 | Allergic dermatitis | Discharge from steel and pulp mills; erosion of natural deposits |
|
Inorganic Chemicals- Nitrate (measured as Nitrogen) |
10 | 10 | Infants below the age of six months who drink water containing nitrate in excess of the MCL could become seriously ill and, if untreated, may die. Symptoms include shortness of breath and blue-baby syndrome. | Runoff from fertilizer use; leaking from septic tanks, sewage; erosion of natural deposits |
|
Organic Chemicals- Benzene |
zero | 0.005 | Anemia; decrease in blood platelets; increased risk of cancer | Discharge from factories; leaching from gas storage tanks and landfills |
|
Organic Chemicals- Carbon tetrachloride |
zero | 0.005 | Liver problems; increased risk of cancer | Discharge from chemical plants and other industrial activities |
|
Organic Chemicals- Styrene |
0.1 | 0.1 | Liver, kidney, or circulatory system problems | Discharge from rubber and plastic factories; leaching from landfills |
|
Radionuclides Alpha particles |
none ---------- zero | 15 picocuries per Liter (pCi/L) | Increased risk of cancer | Erosion of natural deposits of certain minerals that are radioactive and may emit a form of radiation known as alpha radiation |
|---|
Notes
1Definitions:
- Maximum Contaminant Level Goal (MCLG) - The level of a contaminant in drinking water below which there is no known or expected risk to health. MCLGs allow for a margin of safety and are non-enforceable public health goals.
- Maximum Contaminant Level (MCL) - The highest level of a contaminant that is allowed in drinking water. MCLs are set as close to MCLGs as feasible using the best available treatment technology and taking cost into consideration. MCLs are enforceable standards.
- Maximum Residual Disinfectant Level Goal (MRDLG) - The level of a drinking water disinfectant below which there is no known or expected risk to health. MRDLGs do not reflect the benefits of the use of disinfectants to control microbial contaminants.
- Treatment Technique (TT) - A required process intended to reduce the level of a contaminant in drinking water.
- Maximum Residual Disinfectant Level (MRDL) - The highest level of a disinfectant allowed in drinking water. There is convincing evidence that addition of a disinfectant is necessary for control of microbial contaminants.
2 Units are in milligrams per liter (mg/L) unless otherwise noted. Milligrams per liter are equivalent to parts per million (PPM).
Calculations of Parts per Million and Parts per Billion
In addition to percentage units, the units for expressing the concentration of extremely dilute solutions are parts per million (ppm) and parts per billion (ppb). Both of these units are mass based and are defined as follows:
\[\mathrm{ppm=\dfrac{mass\: of\: solute}{mass\: of\: solution}\times1,000,000} \nonumber \]
\[\mathrm{ppb=\dfrac{mass\: of\: solute}{mass\: of\: solution}\times1,000,000,000} \nonumber \]
Similar to parts per million and parts per billion, related units include parts per thousand (ppth) and parts per trillion (ppt).
Concentrations of trace elements in the body—elements that are present in extremely low concentrations but are nonetheless necessary for life—are commonly expressed in parts per million or parts per billion. Concentrations of poisons, contaminants, and pollutants are also described in these units. For example, cobalt is present in the body at a concentration of 21 ppb, while the State of Oregon’s Department of Agriculture limits the concentration of arsenic in fertilizers to 9 ppm.
In aqueous solutions, 1 ppm is essentially equal to 1 mg/L, and 1 ppb is equivalent to 1 µg/L.
\[\text{1 ppm}= \dfrac{\text{1 mg Solute}}{\text{1 L Solution}} \nonumber \]
\[\text{1 ppb} = \dfrac{1\; \mu \text{g Solute}}{\text{1 L Solution}} \nonumber \]
Amendment SDWA of 1986
This change to the Safe Drinking Water Act established MCLGs and increased the total of regulated contaminants to 83. The EPA would also install more monitoring devices to detect organic contaminants. Research was done to detect pathogens more effectively to reduce disease. Lastly, public notifications from water systems to the consumers would be made announced if severe water issues occurred.
Amendment of SDWA of 1996
This particular legislation provided more protection and assessment of water sources (lakes, river, streams). In addition, water companies were required to provide consumers with a water quality report. States could seek federal money for upgrading their water quality processes. Cost benefit analysis would be done to determine risk and reward of lowering the concentration of a contaminant. For small regulated water companies, more financial and technical assistance would be offered to help them maintain drinking water standards.
Water Treatment (US CDC)
Drinking water supplies in the United States are among the safest in the world. However, even in the U.S., drinking water sources can become contaminated, causing sickness and disease from waterborne germs, such as Cryptosporidium, E. coli, Hepatitis A, Giardia intestinalis, and other pathogens.
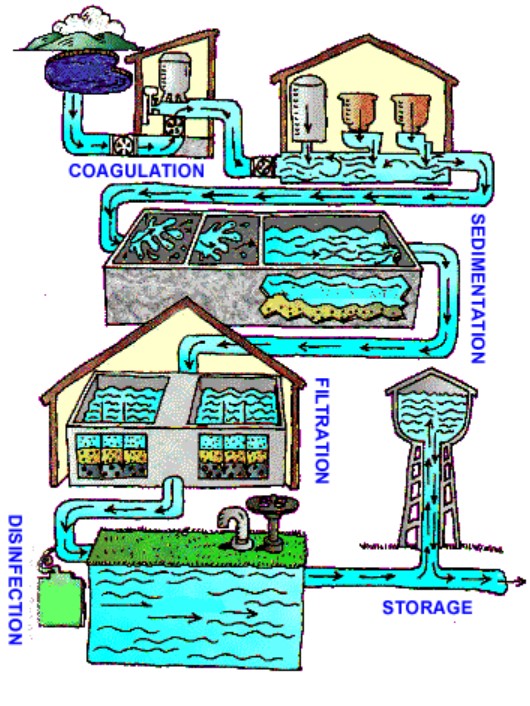
Drinking water sources are subject to contamination and require appropriate treatment to remove disease-causing agents. Public drinking water systems use various methods of water treatment to provide safe drinking water for their communities. Today, the most common steps in water treatment (Figure \(\PageIndex{1}\)) used by community water systems (mainly surface water treatment) include:
Coagulation and Flocculation
Coagulation and flocculation are often the first steps in water treatment. Chemicals with a positive charge are added to the water. The positive charge of these chemicals neutralizes the negative charge of dirt and other dissolved particles in the water. When this occurs, the particles bind with the chemicals and form larger particles, called floc. Historically, dirty water is cleaned by treating with alum, Al2(SO4)3.12 H2O, and lime, Ca(OH)2. These electrolytes cause the pH of the water to change due to the following reactions:
Al2(SO4)3.12 H2O, -> Al3+(aq) + 3 SO42-(aq) + 12 H2O
SO42-(aq) + H2O -> HSO4-(aq) + OH- (causing pH change)
Ca(OH)2 -> Ca2+(aq) + 2 OH- (causing pH change)
The slightly basic water causes Al(OH)3, Fe(OH)3 and Fe(OH)2 to precipitate, bringing the small particulates with them and the water becomes clear. Some records have been found that Egyptians and Romans used these techniques as early as 2000 BC.
Suspension of iron oxide particulates and humic organic matter in water gives water the yellow muddy appearance. Both iron oxide particulates and organic matter can be removed from coagulation and flocculation. The description given here is oversimplified, and many more techniques have been applied in the treatment of water. Coagulation is a major application of lime in the treatment of wastewater.
Other salts such as iron sulfates Fe2(SO4)3 and FeSO4, chromium sulfate Cr2(SO4)3, and some special polymers are also useful. Other ions such as sodium, chloride, calcium, magnesium, and potassium also affect the coagulation process. So do temperature, pH, and concentration.
Disposal of coagulation sludge is a concern, however.
Sedimentation
During sedimentation, floc settles to the bottom of the water supply, due to its weight. This settling process is called sedimentation. The floc particles are then removed from the bottom of the basins.
Aeration
Bringing air into intimate contact with water for the purpose of exchanging certain components between the two phases is called aeration. Oxygenation is one of the purposes of aeration. Others are removal of volatile organic substances, hydrogen sulfide, ammonia, and volatile organic compounds
Filtration
Once the floc has settled to the bottom of the water supply, the clear water on top will pass through filters of varying compositions (sand, gravel, and charcoal) and pore sizes, in order to remove dissolved particles, such as dust, parasites, bacteria, viruses, and chemicals.
Treatment with Chlorine and/or Chloramine
Most communities use either chlorine or chloramines. Some communities switch back and forth between chlorine and chloramines at different times of the year or for other operational reasons. Less commonly, utilities use other disinfectants, such as chlorine dioxide. Some water systems that use water from a groundwater source (like community wells) do not have to add a disinfectant at all.
Ozone Disinfection
Ozone disinfection, or ozonation, is an unstable molecule which readily gives up one atom of oxygen providing a powerful oxidizing agent which is toxic to most waterborne organisms. It is a very strong, broad spectrum disinfectant that is widely used in Europe and in a few municipalities in the United States and Canada. It is an effective method to inactivate harmful protozoa that form cysts. It also works well against almost all other pathogens. Ozone is made by passing oxygen through ultraviolet light or a "cold" electrical discharge. To use ozone as a disinfectant, it must be created on-site and added to the water by bubble contact. Some of the advantages of ozone include the production of fewer dangerous by-products and the absence of taste and odor problems (in comparison to ). No residual ozone is left in the water. In the absence of a residual disinfectant in the water, chlorine or chloramine may be added throughout a distribution system to remove any potential pathogens in the distribution piping.
Ultraviolet Disinfection
Ultraviolet disinfection of water is a purely physical, chemical-free process. Even parasites such as Cryptosporidium or Giardia, which are extremely resistant to chemical disinfectants, are efficiently reduced. UV can also be used to remove chlorine and chloramine species from water; this process is called photolysis, and requires a higher dose than normal disinfection. The dead microorganisms are not removed from the water. UV disinfection does not remove dissolved organics, inorganic compounds or particles in the water. The world's largest water disinfection plant treats drinking water for New York city. The Catskill-Delaware Water Ultraviolet Disinfection Facility, commissioned on 8 October 2013, incorporates a total of 56 energy-efficient UV reactors treating up to 2.2 billion US gallons (8,300,000 m3) a day.
Ultraviolet can also be combined with ozone or hydrogen peroxide to produce hydroxyl radicals to break down trace contaminants through an advanced oxidation process.
Fluoridation (US CDC)
The mineral fluoride occurs naturally on earth and is released from rocks into the soil, water, and air. All water contains some fluoride. Usually, the fluoride level in water is not enough to prevent tooth decay; however, some groundwater and natural springs can have naturally high levels of fluoride.
Fluoride has been proven to protect teeth from decay. Bacteria in the mouth produce acid when a person eats sugary foods. This acid eats away minerals from the tooth’s surface, making the tooth weaker and increasing the chance of developing cavities. Fluoride helps to rebuild and strengthen the tooth’s surface, or enamel. Water fluoridation prevents tooth decay by providing frequent and consistent contact with low levels of fluoride. By keeping the tooth strong and solid, fluoride stops cavities from forming and can even rebuild the tooth’s surface.
Community water fluoridation is the process of adjusting the amount of fluoride found in water to achieve optimal prevention of tooth decay.
Although other fluoride-containing products, such as toothpaste, mouth rinses, and dietary supplements are available and contribute to the prevention and control of tooth decay, community water fluoridation has been identified as the most cost-effective method of delivering fluoride to all, reducing tooth decay by 25% in children and adults.1
Benefits: Strong Teeth (US CDC)
Fluoride benefits children and adults throughout their lives. For children younger than age 8, fluoride helps strengthen the adult (permanent) teeth that are developing under the gums. For adults, drinking water with fluoride supports tooth enamel, keeping teeth strong and healthy. The health benefits of fluoride include having:
- Fewer cavities.
- Less severe cavities.
- Less need for fillings and removing teeth.
- Less pain and suffering because of tooth decay.
Fluoride in the Water Today
In 2012, more than 210 million people, or 75% of the US population, were served by community water systems that contain enough fluoride to protect their teeth.5 However, approximately 100 million Americans still do not have access to water with fluoride. Because it is so beneficial, the United States has a national goal for 80% of Americans to have water with enough fluoride to prevent tooth decay by 2020.
https://www.youtube.com/watch?v=0_ZcCqqpS2o&feature=youtu.be
Summary
- The Safe Drinking Water Act (SDWA) (originally passed by Congress in 1974) protects public health by regulating the nation's public drinking water supply.
- The different steps in water treatment include floculation, sedimentation, filtration, aeration, and disinfection or combinations thereof. Fluoride is also added to municipal water systems for prevention of tooth decay.
Contributors and Attributions
Chung (Peter) Chieh (Professor Emeritus, Chemistry @ University of Waterloo)
- US EPA
- Center for Disease Control (CDC)
- Wikipedia

