14.2: Water in Nature
- Page ID
- 152225
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Know the distribution of earth's water.
- Describe the water (hydrologic) cycle.
- Know the different natural water contaminants
Water 's influence on the world around us is affected by its unique properties some of which where discussed in the previous section. Without water, life might not be able to exist on Earth, and it certainly would not have the tremendous complexity and diversity that we see.
Distribution of Earth's Water
Earth’s oceans contain 97% of the planet’s water, so just 3% is fresh water, water with low concentrations of salts (Figure \(\PageIndex{1}\)). The majority of the Earth's water can be classified as being saline (or salt containing). Most freshwater is trapped as ice in the vast glaciers and ice sheets of Greenland. A storage location for water such as an ocean, glacier, pond, or even the atmosphere is known as a reservoir.
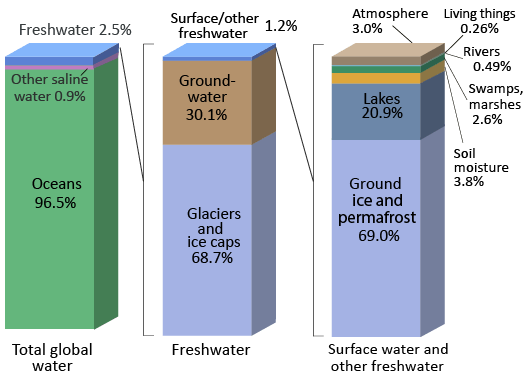
The Water (Hydrologic) Cycle and Natural Contaminants
Because Earth’s water is present in all three states, it can get into a variety of environments around the planet. The movement of water around Earth’s surface is the water cycle (Figure \(\PageIndex{2}\)).
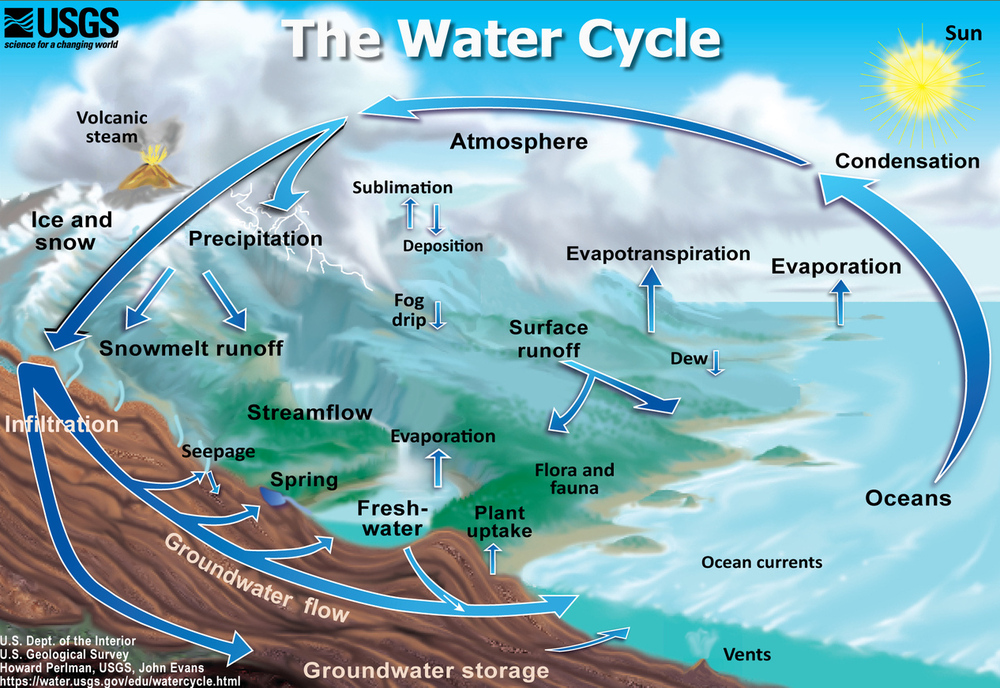
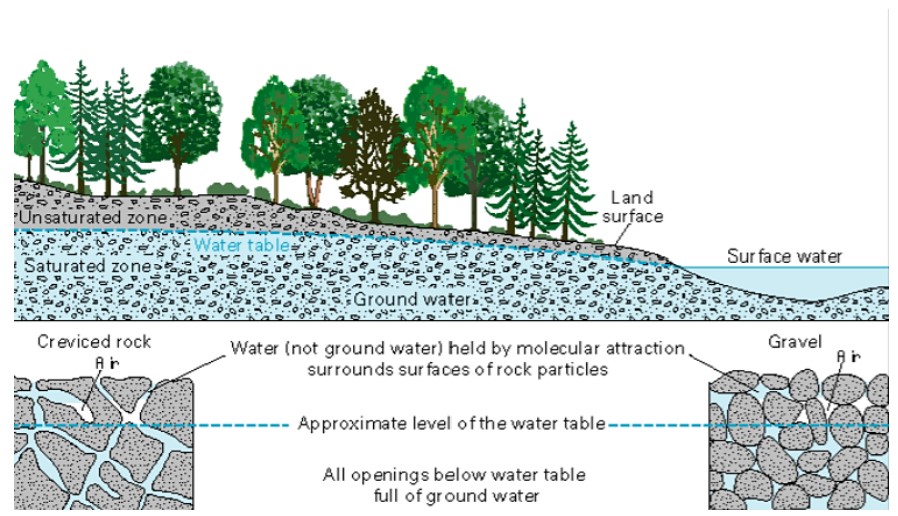
Water changes from a liquid to a gas by evaporation to become water vapor. The Sun’s energy can evaporate water from the ocean surface or from lakes, streams, or puddles on land. Only the water molecules evaporate; the salts remain in the ocean or a freshwater reservoir. The water vapor remains in the atmosphere until it undergoes condensation and then precipitation. Precipitation can be rain, sleet, hail, or snow. At the surface, water or melted water, may eventually evaporate and reenter the atmosphere. A significant amount of water infiltrates into the ground. Water may seep through dirt and rock below the soil through pores infiltrating the ground to go into Earth’s groundwater system (Figure \(\PageIndex3}\)). Groundwater enters aquifers which are bodies of rock or sediment that store (and yield) large amounts of usable water in their pores. Alternatively, the water may come to the surface through springs or find its way back to the oceans.
Substances Present in Natural Waters
The amounts of water present in the atmosphere and on land (as surface runoff, lakes and streams) is great enough to make it a significant agent in transporting substances between the lithosphere (the rigid surface of the earth) and the oceans. Water interacts with both the atmosphere and the lithosphere (the rigid surface of the earth) acquiring solutes from each, and thus provides the major chemical link between these two realms. The various transformations undergone by water through the different stages of the hydrologic cycle act to transport both dissolved and particulate substances between different geographic locations.
The Safe Drinking Water Act defines the term "contaminant" as meaning any physical, chemical, biological, or radiological substance or matter in water. Therefore, the law defines "contaminant" very broadly as being anything other than water molecules.
Gases
The carbon dioxide in natural water creates an interesting phenomenon. Rainwater is saturated with CO2, and it dissolves limestones. When CO2 is lost due to temperature changes or escaping from water drops, the reverse reaction takes place. The solid formed, however, may be a less stable phase called aragonite, which has the same chemical formula as, but a different crystal structure than that of calcite.
The rain dissolves calcium carbonate by the two reactions shown above. The water carries the ions with it, sips through the crack of the rocks. When it reached the ceiling of a cave, the drop dangles there for a long time before fallen. During this time, the carbon dioxide escapes and the pH of the water increases. Calcium carbonate crystals begin to appear. Calcite, aragonite, stalactite, and stalagmite are four common solids found in the formation of caves.
Dissolved Minerals
Table \(\PageIndex{1}\) list the major ions present in seawater. The composition does vary, depending on region, depth, latitude, and water temperature. Waters at the river mouths contain less salt. If the ions are utilized by living organism, its contents vary according to the populations of organisms.
Dust particles and ions present in the air are nucleation center of water drops. Thus, waters from rain and snow also contain such ions: Ca2+, Mg2+, Na+, K+, NH4+. These cations are balanced by anions, HCO3-, SO4-, NO2-, Cl-, and NO3-. The pH of rain is between 5.5 and 5.6. Rain and snow waters eventually become river or lake waters. When the rain or snow waters fall, they interact with vegetation, top soil, bed rock, river bed and lake bed, dissolving whatever is soluble. Bacteria, algae, and water insects also thrive. Solubilities of inorganic salts are governed by the kinetics and equilibria of dissolution. The most common ions in lake and river waters are the same as those present in rainwater, but at higher concentrations. The pH of these waters depends on the river bed and lake bed. Natural waters contain dissolved minerals. Waters containing Ca2+ and Mg2+ ions are usually called hard water.
|
cations
|
g/kg
|
anions
|
g/kg
|
|---|---|---|---|
| Na+ |
10.77
|
Cl– |
19.354
|
| Mg2+ |
1.29
|
SO42– |
2.712
|
| Ca2+ |
0.412
|
Br– |
0.087
|
| K+ |
0.399
|
||
| Sr2+ |
0.0079
|
||
| Al3+ |
0.005
|
Although most elements are found in seawater only at trace levels, marine organisms may selectively absorb them and make them more detectable. Iodine, for example, was discovered in marine algae (seaweeds) 14 years before it was found in seawater. Other elements that were not detected in seawater until after they were found in marine organisms include barium, cobalt, copper, lead, nickel, silver and zinc. Si-32, presumably deriving from cosmic ray bombardment of Ar, has been discovered in marine sponges.
Hard Water
Minerals usually dissolve in natural water bodies such as lakes, rivers, springs, and underground waterways (ground waters). Calcium carbonate, CaCO3, is one of the most common inorganic compounds in the Earth crust. It is the ingredient for both calcite and aragonite. These two minerals have different crystal structures and appearance. This photograph shows crystals of typical Calcite.
Calcium-carbonate minerals dissolve in water, with a solubility product as shown below.
\[CaCO_3 \rightleftharpoons Ca^{2+} + CO_3^{2-} \;\;\; K_{sp} = 5 \times 10^{-9} \nonumber \]
From the solubility product, we can (see example 1) evaluate the molar solubility to be 7.1x10-5 M or 7.1 mg/L (7.1 ppm of CaCO3 in water). The solubility increases as the pH decrease (increase acidity). This is compounded when the water is saturated with carbon dioxide, CO2. Saturated CO2 solution contains carbonic acid, which help the dissolution due to the reaction:
\[H_2O + CO_2 \rightleftharpoons H_2CO_3 \nonumber \]
\[CaCO_3 + H_2CO_3 \rightleftharpoons Ca^{2+} + 2 HCO_3^- \nonumber \]
Because of these reactions, some natural waters contain more than 300 ppm calcium carbonates or its equivalents.
The composition of the ocean has attracted the attention of some of the more famous names in science, including Robert Boyle, Antoine Lavoisier and Edmund Halley. Their early investigations tended to be difficult to reproduce, owing to the different conditions under which they crystallized the various salts. As many as 54 salts, double salts and hydrated salts can be obtained by evaporating seawater to dryness. At least 73 elements are now known to be present in seawater.
Organic Matter
Most of the organic carbon in seawater is present as dissolved material, with only about 1-2% in particulates. The total organic carbon content ranges between 0.5 mg/L in deep water to 1.5 mg/L near the surface. There is still considerable disagreement about the composition of the dissolved organic matter; much of it appears to be of high molecular weight, and may be polymeric. Substances qualitatively similar to the humic acids found in soils can be isolated. The greenish color that is often associated with coastal waters is due to a mixture of fluorescent, high molecular weight substances of undetermined composition known as “Gelbstoffe”. It is likely that the significance of the organic fraction of seawater may be much greater than its low abundance would suggest. For one thing, many of these substances are lipid-like and tend to adsorb onto surfaces. It has been shown that any particle entering the ocean is quickly coated with an organic surface film that may influence the rate and extent of its dissolution or decomposition. Certain inorganic ions may be strongly complexed by humic-like substances. The surface of the ocean is mostly covered with an organic film, only a few molecular layers thick. This is believed to consist of hydrocarbons, lipids, and the like, but glycoproteins and proteoglycans have been reported. If this film is carefully removed from a container of seawater, it will quickly be reconstituted. How significant this film is in its effects on gas exchange with the atmosphere is not known.
Summary
- The water (hydrologic) cycle describes the continuous movement of water on, above and below the surface of the Earth. The water moves from one reservoir to another, such as from river to ocean, or from the ocean to the atmosphere, by the physical processes of evaporation, condensation, precipitation, infiltration, surface runoff, and subsurface flow. In doing so, the water goes through different forms: liquid, solid (ice) and capor.
- Different substances that can be found in natural waters include dissolved minerals, dissolved gases, and organic matter.
Contributors and Attributions
- Template:ContribCandelaGeo
- TextMap: Introduction to Environmental Science (Zendher et al.)
Chung (Peter) Chieh (Professor Emeritus, Chemistry @ University of Waterloo)
Stephen Lower, Professor Emeritus (Simon Fraser U.) Chem1 Virtual Textbook

