10.7: Plastics and the Environment
- Page ID
- 153847
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
- Know the problems associated with plastics.
- Identify the type of polymer associated with each recycling number.
- Know the different plastic recycling processes.
Problems with Plastics
Due to their low cost, ease of manufacture, versatility, and imperviousness to water, plastics are used in a multitude of products of different scale, including paper clips and spacecraft. They have prevailed over traditional materials, such as wood, stone, horn and bone, leather, metal, glass, and ceramic, in some products previously left to natural materials. However, there are numerous problems encountered with plastic use.
Small-molecule release
Many kinds of polymers contain small molecules — either unreacted monomers, or substances specifically added (plasticizers, uv absorbers, flame retardants, etc.) to modify their properties. Many of these smaller molecules are able to diffuse through the material and be released into any liquid or air in contact with the plastic — and eventually into the aquatic environment. Those that are used for building materials (in mobile homes, for example) can build up in closed environments and contribute to indoor air pollution.
Residual monomer
Formation of long polymer chains is a complicated and somewhat random process that is never perfectly stoichiometric. It is therefore not uncommon for some unreacted monomer to remain in the finished product. Some of these monomers, such as formaldehyde, styrene (from polystyrene, including polystyrene foam food take-out containers), vinyl chloride, and bisphenol-A (from polycarbonates) are known carcinogens. Although there is little evidence that the small quantities that diffuse into the air or leach out into fluids pose a quantifiable health risk, people are understandably reluctant to tolerate these exposures, and public policy is gradually beginning to regulate them.
Perfluorooctanoic acid (PFOA), the monomer from which Teflon is made, has been the subject of a 2004 lawsuit against a DuPont factory that contaminated groundwater. Small amounts of PFOA have been detected in gaseous emissions from hot fluorocarbon products.
Decomposition products
Most commonly-used polymers are not readily biodegradable, particularly under the anaerobic conditions of most landfills. And what decomposition does occur will combine with rainwater to form leachates that can contaminate nearby streams and groundwater supplies. Partial photodecomposition, initiated by exposure to sunlight, is a more likely long-term fate for exposed plastics, resulting in tiny broken-up fragments. Many of these materials are less dense than seawater, and once they enter the oceans through coastal sewage outfalls or from marine vessel wastes, they tend to remain there indefinitely.
Open burning of polymeric materials containing chlorine (polyvinyl chloride, for example) is known to release compounds such as dioxins that persist in the environment. Incineration under the right conditions can effectively eliminate this hazard. Disposed products containing fluorocarbons (Teflon-coated ware, some personal-care, waterproofing and anti-stick materials) break down into perfluorooctane sulfonate which has been shown to damage aquatic animals.
Hazards to animals
There are two general types of hazards that polymers can introduce into the aquatic environment. One of these relates to the release of small molecules that act as hormone disrupters as described above. It is well established that small aquatic animals such as fish are being seriously affected by such substances in many rivers and estuarine systems, but details of the sources and identities of these molecules have not been identified. One confounding factor is the release of sewage water containing human birth-control drugs (which have a feminizing effect on sexual development) into many waterways.
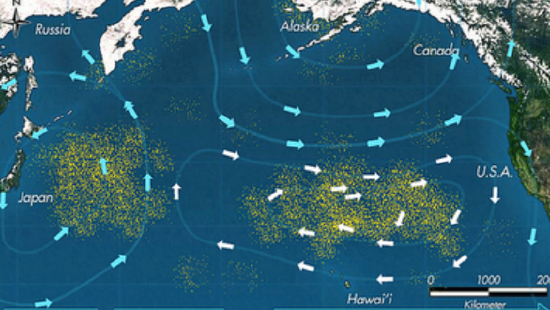
The other hazard relates to pieces of plastic waste that aquatic animals mistake for food or become entangled in (Figure \(\PageIndex{1}\)) .


These dangers occur throughout the ocean, but are greatly accentuated in regions known as gyres. These are regions of the ocean in which a combination of ocean currents drives permanent vortices that tend to collect and concentrate floating materials. The most notorious of these are the Great Pacific Gyres that have accumulated astounding quantities of plastic waste.
.
Recycling
The huge quantity (one estimate is 108 metric tons per year) of plastic materials produced for consumer and industrial use has created a gigantic problem of what to do with plastic waste which is difficult to incinerate safely and which, being largely non-biodegradable, threatens to overwhelm the capacity of landfills. An additional consideration is that de novo production most of the major polymers consumes non-renewable hydrocarbon resources.

Plastics recycling has become a major industry, greatly aided by enlightened trash management policies in the major developed nations. However, it is plagued with some special problems of its own:
- Recycling is only profitable when there is a market for the regenerated material. Such markets vary with the economic cycle (they practically disappeared during the recession that commenced in 2008.)
- The energy-related costs of collecting and transporting plastic waste, and especially of processing it for re-use, are frequently the deciding factor in assessing the practicability of recycling.
- Collection of plastic wastes from diverse sources and locations and their transport to processing centers consumes energy and presents numerous operational problems.
- Most recycling processes are optimized for particular classes of polymers. The diversity of plastic types necessitates their separation into different waste streams — usually requiring manual (i.e., low-cost) labor. This in turn encourages shipment of these wastes to low-wage countries, thus reducing the availability of recycled materials in the countries in which the plastics originated.
Some of the major recycling processes include
- Thermal decomposition processes that can accommodate mixed kinds of plastics and render them into fuel oil, but the large inputs of energy they require have been a problem.
- A very small number of condensation polymers can be depolymerized so that the monomers can be recovered and re-used.
- Thermopolymers can be melted and pelletized, but those of widely differing types must be treated separately to avoid incompatability problems.
- Thermosets are usually shredded and used as filler material in recycled thermopolymers.
Other processes
A process has also been developed in which many kinds of plastic can be used as a carbon source in the recycling of
scrap steel. There is also a possibility of mixed recycling of different plastics, which does not require their separation. It is called compatibilization and requires use of special chemical bridging agents compatibilizers. It can help to keep the quality of recycled material and to skip often expensive and inefficient preliminary scanning of waste plastics streams and their separation/purification.

Recycled Plastics
Seven groups of plastic polymers, each with specific properties, are used worldwide for packaging applications (see Table \(\PageIndex{1}\)). Each group of plastic polymer can be identified by its plastic identification code (PIC), usually a number or a letter abbreviation. For instance, low-density polyethylene can be identified by the number "4" or the letters "LDPE". The PIC appears inside a three-chasing-arrow recycling symbol. The symbol is used to indicate whether the plastic can be recycled into new products.
The PIC was introduced by the Society of the Plastics Industry, Inc., to provide a uniform system for the identification of various polymer types and to help recycling companies separate various plastics for reprocessing. Manufacturers of plastic products are required to use PIC labels in some countries/regions and can voluntarily mark their products with the PIC where there are no requirements. Consumers can identify the plastic types based on the codes usually found at the base or at the side of the plastic products, including food/chemical packaging and containers.

Not all categories are accepted by all local recycling authorities, so residents need to be informed about which kinds should be placed in recycling containers and which should be combined with ordinary trash.
| Plastic identification code | Type of plastic polymer | Properties | Common packaging applications | Melting temperatures (°C) |
|---|---|---|---|---|

|
Polyethylene terephthalate(PET, PETE) | Clarity, strength, toughness, barrier to gas and moisture. | Soft drink, water and salad dressing bottles; peanut butter and jam jars; ice cream cone lids; small consumer electronics | Tm = 250 |

|
High-density polyethylene(HDPE) | Stiffness, strength, toughness, resistance to moisture, permeability to gas | Water pipes, Gas & Fire Pipelines, Electrical & Communications conduit, hula hoop rings, five gallon buckets, milk, juice and water bottles; grocery bags, some shampoo/toiletry bottles | Tm = 130 |

|
Polyvinyl chloride(PVC) | Versatility, ease of blending, strength, toughness. | Blister packaging for non-food items; cling films for non-food use. May be used for food packaging with the addition of the plasticisers needed to make natively rigid PVC flexible. Non-packaging uses are electrical cable insulation; rigid piping; vinyl records. | Tm = 240 |

|
Low-density polyethylene(LDPE) | Ease of processing, strength, toughness, flexibility, ease of sealing, barrier to moisture | Frozen food bags; squeezable bottles, e.g. honey, mustard; cling films; flexible container lids | Tm = 120 |

|
Polypropylene(PP) | Strength, toughness, resistance to heat, chemicals, grease and oil, versatile, barrier to moisture. | Reusable microwaveable ware; kitchenware; yogurt containers; margarine tubs; microwaveable disposable take-away containers; disposable cups; soft drink bottle caps; plates. | Tm = 173 |

|
Polystyrene(PS) | Versatility, clarity, easily formed | Egg cartons; packing peanuts; disposable cups, plates, trays and cutlery; disposable take-away containers | Tm = 240 |

|
Other (often polycarbonateor ABS) | Dependent on polymers or combination of polymers | Beverage bottles, baby milk bottles. Non-packaging uses for polycarbonate, compact discs, "unbreakable" glazing, electronic apparatus housing, lenses (including sunglasses), prescription glasses, automotive headlamps, riot shields, instrument panels. |
Polycarbonate: Tm = 225 |
Tire Recycling
The large number of rubber tires that are disposed of, together with the increasing reluctance of landfills to accept them, has stimulated considerable innovation in the re-use of this material, especially in the construction industry.
Plastics and Fire Hazards
The term fire (or flame)-retardant as applied to organic (i.e., containing carbon) materials, is intended to refer to reduced fire hazard, as all will burn under certain circumstances. Fabric flammability is an important textile issue, especially for stage drapery that will be used in a public space such as a school, theatre or special event venue. In the United States, Federal regulations require that drapery fabrics used in such spaces be certified as flame or fire-retardant. For draperies and other fabrics used in public places, this is known as the NFPA 701 Test, which follows standards developed by the National Fire Protection Association (NFPA). Although all fabrics will burn, some are naturally more resistant to fire than others. Those that are more flammable can have their fire resistance drastically improved by treatment with fire-retardant chemicals. Inherently flame-retardant fabrics such as polyester are commonly used for flame retardant curtain fabrics.
The deaths in fiery crashes of race car drivers Fireball Roberts at Charlotte, and Eddie Sachs and Dave MacDonald at Indianapolis in 1964 led to the use of flame-resistant ics such as Nomex. Nomex and related aramid polymers are related to nylon, but have aromatic backbones, and hence are more rigid and more durable. Nomex is an example of a meta variant of the aramids (Kevlar is a para aramid). Unlike Kevlar, Nomex strands cannot align during filament polymerization and has less strength. However, it has excellent thermal, chemical, and radiation resistance for a polymer material.
A Nomex hood is a common piece of racing and firefighting equipment. It is placed on the head on top of a firefighter's face mask. The hood protects the portions of the head not covered by the helmet and face mask from the intense heat of the fire.

Wildland firefighters wear Nomex shirts and trousers as part of their personal protective equipment during wildfire suppression activities.
Racing car drivers wear driving suits constructed of Nomex and or other fire retardant materials, along with Nomex gloves, long underwear, balaclavas, socks, helmet lining and shoes, to protect them in the event of a fire.
Military pilots and aircrew wear flight suits made of over 92 percent Nomex to protect them from the possibility of cockpit fires and other mishaps. Recently, troops riding in ground vehicles have also begun wearing Nomex. Kevlar thread is often used to hold the fabric together at seams.
Military tank drivers also typically use Nomex hoods as protection against fire.
Plasticizers and Pollution
Plasticizers (UK: plasticisers) or dispersants are additives that increase the plasticity or decrease the viscosity of a material. These substances are compounded into certain types of plastics to render them more flexible by lowering the glass transition temperature. They accomplish this by taking up space between the polymer chains and acting as lubricants to enable the chains to more readily slip over each other. Many (but not all) are small enough to be diffusible and a potential source of health problems.
Polyvinyl chloride polymers are one of the most widely-plasticized types, and the odors often associated with flexible vinyl materials such as garden hoses, waterbeds, cheap shower curtains, raincoats and upholstery are testament to their ability to migrate into the environment.
 The well-known "new car smell" is largely due to plasticizer release from upholstery and internal trim.
The well-known "new car smell" is largely due to plasticizer release from upholstery and internal trim.
According to 2014 data, the total global market for plasticizers was 8.4 million metric tonnes including 1.3 million metric tonnes in Europe.
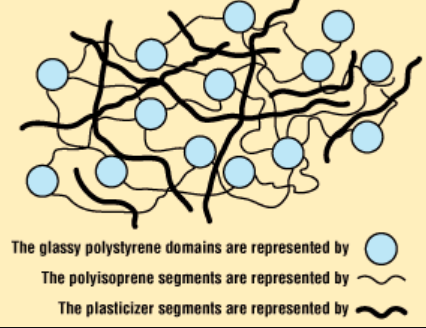
Substantial concerns have been expressed over the safety of some plasticizers, especially because some low molecular weight ortho-phthalates have been classified as potential endocrine disruptors with some developmental toxicity reported.
Summary
- Plastics are found everywhere due to its low cost, versatility, ease of use etc.
- Plastics pose a threat to the environment due to residual or degradation products that contribute to air and water pollution.
- Plastics hazards to animals and marine life as these living creatures mistake them for food.
- Plastic polymers are classified into seven groups for recycling purposes.
Contributors and Attributions
Stephen Lower, Professor Emeritus (Simon Fraser U.) Chem1 Virtual Textbook
- Wikipedia

