10.5: Condensation Polymers
- Page ID
- 152200
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Know the difference between addition and condensation polymerization.
- Know the properties and uses of common synthetic condensation polymers.
A large number of important and useful polymeric materials are not formed by addition polymerizaiton, but proceed instead by conventional functional group transformations of polyfunctional reactants. These polymerizations often (but not always) occur with loss of a small byproduct, such as water, and generally (but not always) combine two different components in an alternating structure. The polyester Dacron and the polyamide Nylon 66, shown here, are two examples of synthetic condensation polymers, also known as step-growth polymers. In contrast to addition polymerizaion, most of which grow by carbon-carbon bond formation, step-growth polymers generally grow by carbon-heteroatom bond formation (C-O & C-N in Dacron & Nylon respectively). Although polymers of this kind might be considered to be alternating copolymers, the repeating monomeric unit is usually defined as a combined moiety.
Examples of naturally occurring condensation polymers are cellulose, starch, the polypeptide chains of proteins, and poly(β-hydroxybutyric acid), a polyester synthesized in large quantity by certain soil and water bacteria.
Nylon and Other Polyamides
Condensation polymerization (also known as step-growth) requires that the monomers possess two or more kinds of functional groups that are able to react with each other in such a way that parts of these groups combine to form a small molecule (often H2O) which is eliminated from the two pieces. The now-empty bonding positions on the two monomers can then join together .
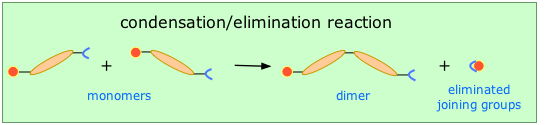
One important class of condensation polymers are polyamides. They arise from the reaction of carboxylic acid and an amine. Examples include nylons and proteins.
When prepared from diamines and dicarboxylic acids, e.g. the production of nylon 66, the polymerization produces two molecules of water per repeat unit:
n H2N-X-NH2 + n HO2C-Y-CO2H → [HN-X-NHC(O)-Y-C(O)]n + 2n H2O
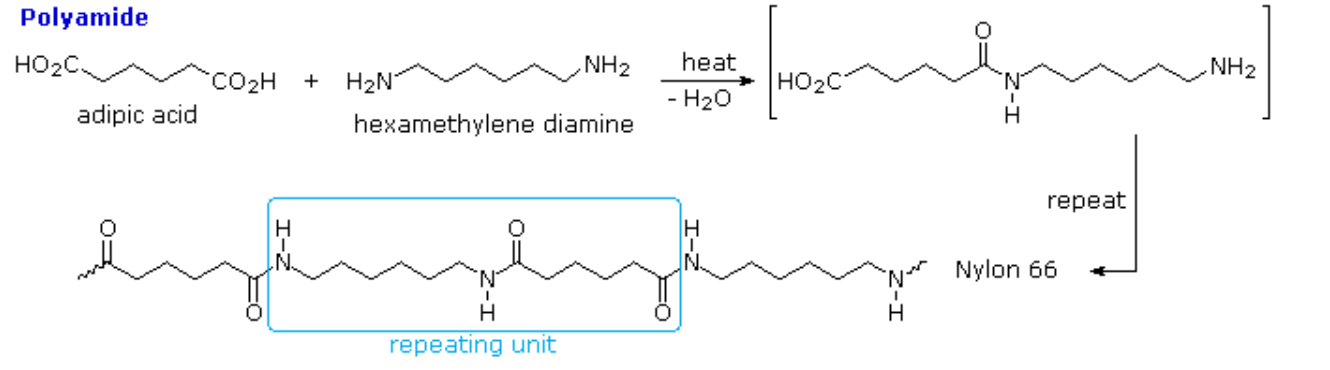
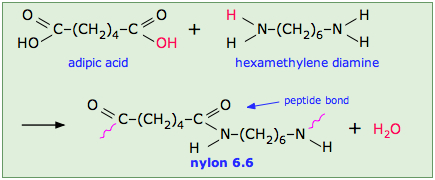
Note that the monomeric units that make up the polymer are not identical with the starting components.
Nylon is a thermoplastic silky material that can be melt-processed into fibers, films, or shapes.:2 It is made of repeating units linked by amide links similar to the peptide bonds in proteins. Nylon polymers can be mixed with a wide variety of additives to achieve many different property variations. Nylon polymers have found significant commercial applications in fabric and fibers (apparel, flooring and rubber reinforcement), in shapes (molded parts for cars, electrical equipment, etc.), and in films (mostly for food packaging).

Nylon was the first commercially successful synthetic thermoplastic polymer. DuPont began its research project in 1927. The first example of nylon (nylon 6,6) was produced using diamines on February 28, 1935, by Wallace Hume Carothers (Figure \(\PageIndex{1}\)) at DuPont's research facility at the DuPont Experimental Station. In response to Carothers' work, Paul Schlack at IG Farben developed nylon 6, a different molecule based on caprolactam, on January 29, 1938.:10
Nylon was first used commercially in a nylon-bristled toothbrush in 1938, followed more famously in women's stockings or "nylons" which were shown at the 1939 New York World's Fair and first sold commercially in 1940. During World War II, almost all nylon production was diverted to the military for use in parachutesand parachute cord. Wartime uses of nylon and other plastics greatly increased the market for the new materials.
Other polyamides of practical use include nylon 6 and kevlar. Nylon-6 is made from a monomer called caprolactam.
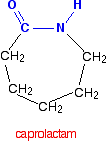
Notice that this already contains an amide link. When this molecule polymerizes, the ring opens, and the molecules join up in a continuous chain. Nylon 6 fibers are tough, possessing high tensile strength, as well as elasticity and lustre. They are wrinkleproof and highly resistant to abrasion and chemicals such as acids and alkalis. The fibers can absorb up to 2.4% of water, although this lowers tensile strength.
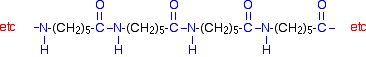
Kevlar is similar in structure to nylon-6,6 except that instead of the amide links joining chains of carbon atoms together, they join benzene rings. The two monomers are benzene-1,4-dicarboxylic acid and 1,4-diaminobenzene.
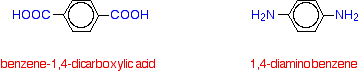
If you line these up and remove water between the -COOH and -NH2 groups in the same way as we did with nylon-6,6, you get the structure of Kevlar:
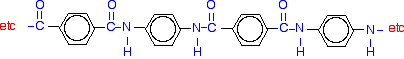
Kevlar is a very strong material - about five times as strong as steel, weight for weight. It is used in bulletproof vests, in composites for boat construction, in lightweight mountaineering ropes, and for lightweight skis and racquets - amongst many other things.
Polyethylene Terephthalate and Other Polyesters
One important class of condensation polymers are polyesters. They arise from the reaction of carboxylic acid and an alcohol. Examples include polyesters, e.g. polyethyleneterephthalate:
- n HO-X-OH + n HO2C-Y-CO2H → [O-X-O2C-Y-C(O)]n + (3n-2) H2O
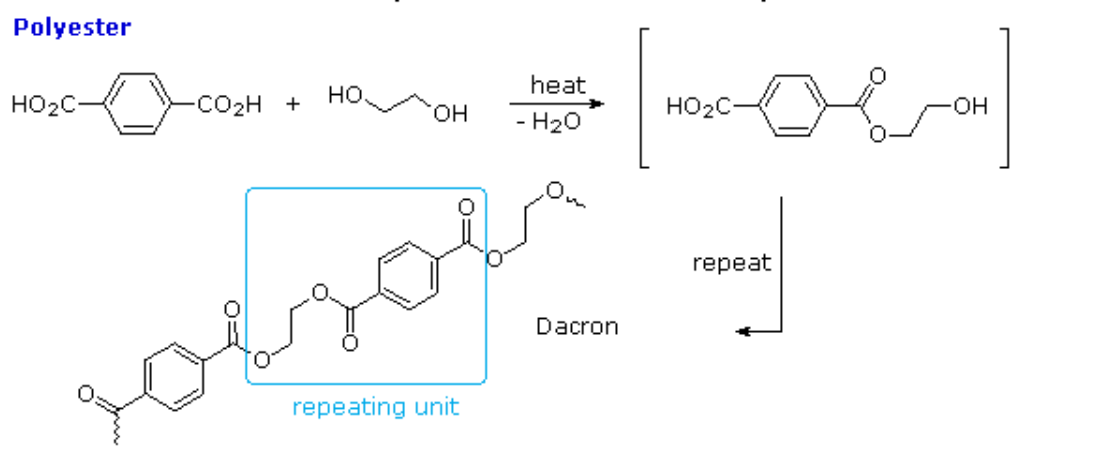
Polyethylene terephthalate (sometimes written poly(ethylene terephthalate)), commonly abbreviated PET, PETE, or the obsolete PETP or PET-P, is the most common thermoplastic polymer resin of the polyester family and is used in fibres for clothing, containers for liquids and foods, thermoforming for manufacturing, and in combination with glass fibre for engineering resins.
It may also be referred to by the brand names Terylene in the UK, Lavsan in Russia and the former Soviet Union, and Dacron in the US.
The majority of the world's PET production is for synthetic fibres (in excess of 60%), with bottle production accounting for about 30% of global demand. In the context of textile applications, PET is referred to by its common name, polyester, whereas the acronym PET is generally used in relation to packaging. Polyester makes up about 18% of world polymer production and is the fourth-most-produced polymer after polyethylene (PE), polypropylene (PP) and polyvinyl chloride (PVC).
Phenol-Formaldehyde and Related Resins
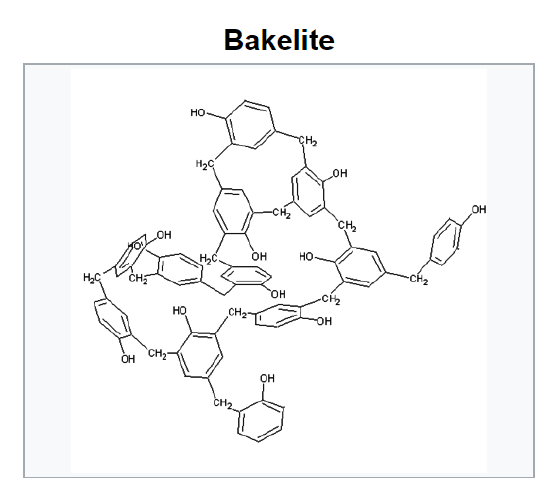
Bakelite was patented on December 7, 1909. The creation of a synthetic plastic was revolutionary for its electrical nonconductivity and heat-resistant properties in electrical insulators, radio and telephone casings and such diverse products as kitchenware, jewelry, pipe stems, children's toys, and firearms.
In recent years the "retro" appeal of old Bakelite products has made them collectible.
Bakelite was designated a National Historic Chemical Landmark on November 9, 1993, by the American Chemical Society in recognition of its significance as the world's first synthetic plastic.
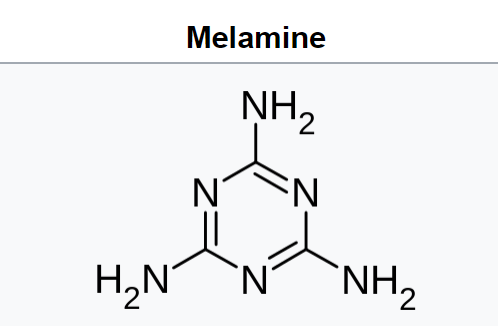
Melamine /ˈmɛləmiːn/ (![]() listen) is an organic compound with the formula C3H6N6. This white solid is a trimer of cyanamide, with a 1,3,5-triazine skeleton. Like cyanamide, it contains 67% nitrogen by mass, and its derivatives have fire retardant properties due to its release of nitrogen gas when burned or charred. Melamine can be combined with formaldehyde and other agents to produce melamine resins. Such resins are characteristically durable thermosetting plastic used in high pressure decorative laminates such as Formica, melamine dinnerware, laminate flooring, and dry erase boards. Melamine foam is used as insulation, soundproofing material and in polymeric cleaning products, such as Magic Eraser.
listen) is an organic compound with the formula C3H6N6. This white solid is a trimer of cyanamide, with a 1,3,5-triazine skeleton. Like cyanamide, it contains 67% nitrogen by mass, and its derivatives have fire retardant properties due to its release of nitrogen gas when burned or charred. Melamine can be combined with formaldehyde and other agents to produce melamine resins. Such resins are characteristically durable thermosetting plastic used in high pressure decorative laminates such as Formica, melamine dinnerware, laminate flooring, and dry erase boards. Melamine foam is used as insulation, soundproofing material and in polymeric cleaning products, such as Magic Eraser.
Other Condensation Polymers
Polycarbonates (PC) are a group of thermoplastic polymers containing carbonate groups in their chemical structures. Polycarbonates used in engineering are strong, tough materials, and some grades are optically transparent. They are easily worked, molded, and thermoformed. Because of these properties, polycarbonates find many applications. Polycarbonates received their name because they are polymers containing carbonate groups (−O−(C=O)−O−). A balance of useful features, including temperature resistance, impact resistance and optical properties, positions polycarbonates between commodity plastics and engineering plastics.
The main polycarbonate material is produced by the reaction of bisphenol A (BPA) and phosgene COCl2. The overall reaction can be written as follows:

Polycarbonate is mainly used for electronic applications that capitalize on its collective safety features. Being a good electrical insulator and having heat-resistant and flame-retardant properties. The second largest consumer of polycarbonates is the construction industry, e.g. for domelights, flat or curved glazing, and sound walls, which all use extruded flat solid or multiwall sheet, or corrugated sheet. A major application of polycarbonate is the production of Compact Discs, DVDs, and Blu-ray Discs.
Polyurethane (PUR and PU) is a polymer composed of organic units joined by carbamate (urethane) links. While most polyurethanes are thermosetting polymers that do not melt when heated, thermoplastic polyurethanes are also available.
Polyurethanes are in the class of compounds called reaction polymers, which include epoxies, unsaturated polyesters, and phenolics. Polyurethanes are produced by reacting an isocyanate containing two or more isocyanate groups per molecule (R−(N=C=O)n) with a polyol containing on average two or more hydroxyl groups per molecule (R′−(OH)n) in the presence of a catalyst or by activation with ultraviolet light.
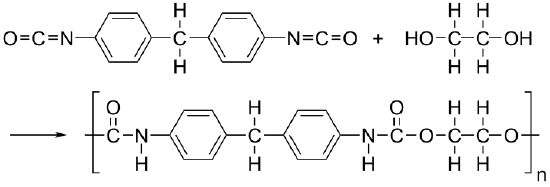
Polyurethanes are used in the manufacture of high-resilience foam seating, rigid foam insulation panels, microcellular foam seals and gaskets, durable elastomeric wheels and tires (such as roller coaster, escalator, shopping cart, elevator, and skateboard wheels), automotive suspension bushings, electrical potting compounds, high performance adhesives, surface coatings and surface sealants, synthetic fibers (e.g., Spandex), carpet underlay, hard-plastic parts (e.g., for electronic instruments), condoms, and hoses.

Fully reacted polyurethane polymer is chemically inert. No exposure limits have been established in the U.S. by OSHA (Occupational Safety and Health Administration) or ACGIH (American Conference of Governmental Industrial Hygienists). It is not regulated by OSHA for carcinogenicity.

Polyurethane polymer is a combustible solid and can be ignited if exposed to an open flame. Decomposition from fire can produce significant amounts of carbon monoxide and hydrogen cyanide, in addition to nitrogen oxides, isocyanates, and other toxic products.Because of the flammability of the material, it has to be treated with flame retardants (at least in case of furniture), almost all of which are considered harmful. California later issued Technical Bulletin 117 2013 which allowed most polyurethane foam to pass flammability tests without the use of flame retardants. Green Science Policy Institute states: "Although the new standard can be met without flame retardants, it does NOT ban their use. Consumers who wish to reduce household exposure to flame retardants can look for a TB117-2013 tag on furniture, and verify with retailers that products do not contain flame retardants."
Liquid resin blends and isocyanates may contain hazardous or regulated components. Isocyanates are known skin and respiratory sensitizers. Additionally, amines, glycols, and phosphate present in spray polyurethane foams present risks.
Exposure to chemicals that may be emitted during or after application of polyurethane spray foam (such as isocyanates) are harmful to human health and therefore special precautions are required during and after this process.
In the United States, additional health and safety information can be found through organizations such as the Polyurethane Manufacturers Association (PMA) and the Center for the Polyurethanes Industry (CPI), as well as from polyurethane system and raw material manufacturers. Regulatory information can be found in the Code of Federal Regulations Title 21 (Food and Drugs) and Title 40 (Protection of the Environment). In Europe, health and safety information is available from ISOPA, the European Diisocyanate and Polyol Producers Association.
Epoxy is either any of the basic components or the cured end products of epoxy resins, as well as a colloquial name for the epoxide functional group. Epoxy resins, also known as polyepoxides, are a class of reactive prepolymers and polymers which contain epoxide groups.
Epoxy resins may be reacted (cross-linked) either with themselves through catalytic homopolymerisation, or with a wide range of co-reactants including polyfunctional amines, acids (and acid anhydrides), phenols, alcohols and thiols (usually called mercaptans). These co-reactants are often referred to as hardeners or curatives, and the cross-linking reaction is commonly referred to as curing. The structure of bisphenol-A diglycidyl ether epoxy resin is shown below: n denotes the number of polymerized subunits and is typically in the range from 0 to 25

Reaction of polyepoxides with themselves or with polyfunctional hardeners forms a thermosetting polymer, often with favorable mechanical properties and high thermal and chemical resistance. Epoxy has a wide range of applications, including metal coatings, use in electronics/electrical components/LEDs, high tension electrical insulators, paint brush manufacturing, fiber-reinforced plastic materials and structural adhesives. Epoxy is sometimes used as a glue (see image at right).

Composite Materials
A composite material (also called a composition material or shortened to composite, which is the common name) is a material made from two or more constituent materials with significantly different physical or chemical properties that, when combined, produce a material with characteristics different from the individual components. Composite materials are generally used for buildings, bridges, and structures such as boat hulls, swimming pool panels, racing car bodies, shower stalls, bathtubs, storage tanks, imitation granite and cultured marble sinks and countertops.
Composites are made up of individual materials referred to as constituent materials. There are two main categories of constituent materials: matrix (binder) and reinforcement. At least one portion of each type is required. The matrix material surrounds and supports the reinforcement materials by maintaining their relative positions. The reinforcements impart their special mechanical and physical properties to enhance the matrix properties. A synergism produces material properties unavailable from the individual constituent materials, while the wide variety of matrix and strengthening materials allows the designer of the product or structure to choose an optimum combination. Many commercially produced composites use a polymer matrix material often called a resin solution. There are many different polymers available depending upon the starting raw ingredients. There are several broad categories, each with numerous variations. The most common are known as polyester, vinyl ester, epoxy, phenolic, polyimide, polyamide, polypropylene, PEEK (polyether ether ketone), and others. Common fibres used for reinforcement include glass fibres, carbon fibres, cellulose (wood/paper fibre and straw) and high strength polymers for example aramid. Silicon carbide fibers are used for some high temperature applications.
One of the most common and familiar composite is fibreglass, in which small glass fibre are embedded within a polymeric material (normally an epoxy or polyester). The glass e is relatively strong and stiff (but also brittle), whereas the polymer is ductile (but also weak and flexible). Thus the resulting fibreglass is relatively stiff, strong, flexible, and ductile.
Figure \(\PageIndex{6}\) Glass reinforcements used for fiberglass are supplied in different physical forms, microspheres, chopped or woven.
Silicones
Silicones, also known as polysiloxanes, are polymers that include any synthetic compound made up of repeating units of siloxane. Silicones consist of an inorganic silicon-oxygen backbone chain (⋯–Si–O–Si–O–Si–O–⋯) with organic side groups attached to the silicon atoms. Silicones have in general the chemical formula [R2SiO]n, where R is an organic group such as an alkyl (methyl, ethyl) or phenyl group. A silicone polymer tha consist of repeated units of dimethyl silicone is shown below.

They are typically heat-resistant and either liquid or rubber-like. Silicones are used in many products. Ullmann's Encyclopedia of Industrial Chemistry lists the following major categories of application: Electrical (e.g., insulation), electronics (e.g., coatings), household (e.g., sealants and cooking utensils), automobile (e.g., gaskets), aeroplane (e.g., seals), office machines (e.g., keyboard pads), medicine and dentistry (e.g., tooth impression molds), textiles and paper (e.g., coatings). For these applications, an estimated 400,000 tonnes of silicones were produced in 1991.

Silicone is often confused with silicon, but they are distinct substances. Silicon is a chemical element, a hard dark-grey semiconducting metalloid which in its crystalline form is used to make integrated circuits ("electronic chips") and solar cells. Silicones are compounds that contain silicon, carbon, hydrogen, oxygen, and perhaps other kinds of atoms as well, and have very different physical and chemical properties.
Summary
- Condensation polymerization (also known as step-growth) requires that the monomers possess two or more kinds of functional groups that are able to react with each other in such a way that parts of these groups combine to form a small molecule (often H2O) which is eliminated from the two pieces. The now-empty bonding positions on the two monomers can then join together .
- Examples of natural condensation polymers include cellulose, starch, and polypeptide chains of proteins.
- Several synthetic condensation polymers discussed include nylon, kevlar, polyester, Bakelite, Melamine, polycarbonates, polyurethanes, epoxies.
- Synthetic condensation polymers have a wide array of household, industrial, commercial, and medical uses and applications.
Contributors and Attributions
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry
Stephen Lower, Professor Emeritus (Simon Fraser U.) Chem1 Virtual Textbook
Jim Clark (Chemguide.co.uk)
- Wikipedia


