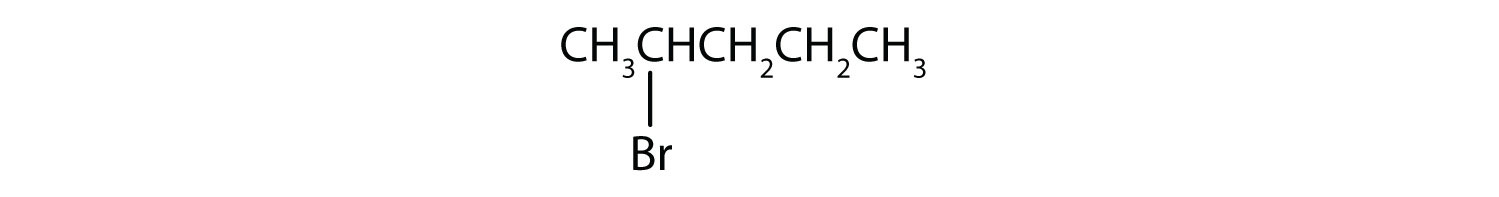9.3: Halogenated Hydrocarbons- Many Uses, Some Hazards
- Page ID
- 152192
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
- Identify haloalkanes (alkyl halides) and know their occurrence, uses, and properties.
- Learn how to name simple haloalkanes.
Haloalkanes also known as alkyl halides or halogenoalkanes, are a group of organic compounds in which one or more halogen atoms are substituted for one or more hydrogen atoms in a hydrocarbon. Haloalkanes are widely used commercially and, consequently, are known under many chemical and commercial names. Although these compounds were once widely used as dry cleaning solvents, coolants in refrigerators and air conditioners, and propellants in hairsprays and deodorants, increasing awareness of their toxicity and impact on the environment has led to a widespread decrease in applications for these materials.
The general formulas for organic molecules with functional groups use the letter \(\ce{R}\) to stand for the rest of the molecule outside of the functional group. Because there are four possible halogen atoms (fluorine, chlorine, bromine, or iodine) that can act as the functional group, we use the general formula \(\ce{R-X}\) to represent an alkyl halide.
Haloalkanes have higher boiling points than alkanes containing the same number of carbons. They are at best only slightly soluble in water but tend to dissolve in organic solvents.
Chloromethane (or methylchloride, CH3Cl) was a widely used refrigerant, but its use has been discontinued due to its toxicity and flammability. Methyl chloride was also once used for producing lead-based gasoline additives (tetramethyllead).
The most important use of chloromethane today is as a chemical intermediate in the production of silicone polymers. Smaller quantities are used as a solvent in the manufacture of butyl rubber and in petroleum refining.
Large amounts of methyl chloride are produced naturally in the oceans by the action of sunlight on biomass and chlorine in sea foam. However, all methyl chloride that is used in industry is produced synthetically. Most methyl chloride is prepared by reacting methanol with hydrogen chloride, according to the chemical equation
\[ CH_3OH + HCl → CH_3Cl + H_2O \nonumber \]
This can be carried out either by bubbling hydrogen chloride gas through boiling methanol with or without a zinc chloride catalyst, or by passing combined methanol and hydrogen chloride vapors over an alumina catalyst at 350 °C (662 °F).
A smaller amount of chloromethane is produced by heating a mixture of methane and chlorine to over 400 °C (752 °F). However, this method also results in more highly chlorinated compounds such as dichloromethane, chloroform, and carbon tetrachloride and is usually only used when these other products are also desired.
Short chain haloalkanes such as dichloromethane (CH2Cl2), trichloromethane (chloroform, CHCl3) and tetrachloromethane (carbon tetrachloride, CCl4) are commonly used as hydrophobic solvents in chemistry. They were formerly very common in industry; however, their use has been greatly curtailed due to their toxicity and harmful environmental effects.
Some specific compounds are still used. Halothane (2-bromo-2-chloro-1,1,1-trifluoroethane) is still used in some situations as an inhalation anesthetic. The compound DDT is a very effective pesticide, but is only used when nothing else works because of its harmful effects on the environment.
IUPAC System: The rules for naming simple alkyl halides are listed below.
- Name the parent compound by finding the longest continuous carbon atom chain that also contains the halogen. Add a prefix for the particular halogen atom. The prefixes for each of the four halogens are fluoro-, chloro-, bromo-, and iodo-. If more than one kind of halogen atom is present, put them in alphabetical order. If there is more than one of the same halogen on a given carbon atom, use the prefixes di-, tri-, or tetra- before the prefix for the halogen.
- As with hydrocarbons, number the carbon chain in a way that makes the sum of halogen numbers as low as possible. If different halogens are in equivalent positions, give the lower number to the one that comes first in alphabetical order.
- Add the numerical prefix into the name before the halogen prefix.
- Separate numbers with commas and separate numbers from names or prefixes with a hyphen. There are no spaces in the name.
Listed below are some examples of names and structural formulas of a few alkyl halides.
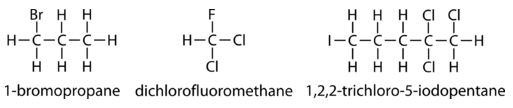
Note that for the structure based on methane, no number needs to be used since there is only one carbon atom. In the third example, the chloro- is listed first alphabetically and the chain is numbered so that the sum of the numbers is as low as possible.
Common Names: The common names of alkyl halides consist of two parts: the name of the alkyl group plus the stem of the name of the halogen, with the ending -ide.
Alkyl halides with simple alkyl groups (one to four carbon atoms) are often called by common names. Those with a larger number of carbon atoms are usually given IUPAC names.
Give the common and IUPAC names for each compound.
- CH3CH2CH2Br
- (CH3)2CHCl
Solution
- The alkyl group (CH3CH2CH2–) is a propyl group, and the halogen is bromine (Br). The common name is therefore propyl bromide. For the IUPAC name, the prefix for bromine (bromo) is combined with the name for a three-carbon chain (propane), preceded by a number identifying the carbon atom to which the Br atom is attached, so the IUPAC name is 1-bromopropane.
- The alkyl group [(CH3)2CH–] has three carbon atoms, with a chlorine (Cl) atom attached to the middle carbon atom. The alkyl group is therefore isopropyl, and the common name of the compound is isopropyl chloride. For the IUPAC name, the Cl atom (prefix chloro-) attached to the middle (second) carbon atom of a propane chain results in 2-chloropropane.
Give common and IUPAC names for each compound.
- CH3CH2I
- CH3CH2CH2CH2F
Answers
a. 1-iodoethane, ethyl iodide
b. 1-fluorobutane, butyl fluoride
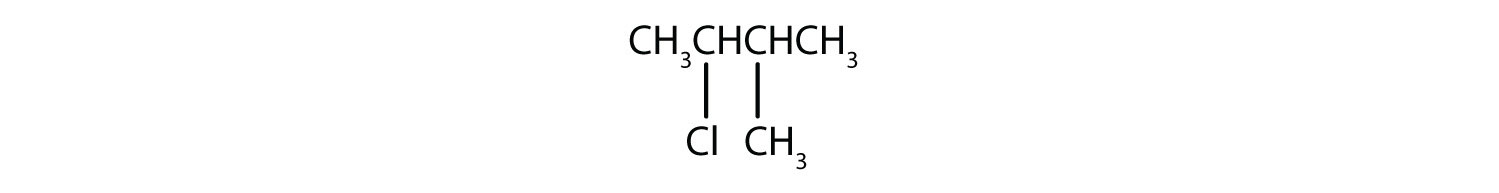
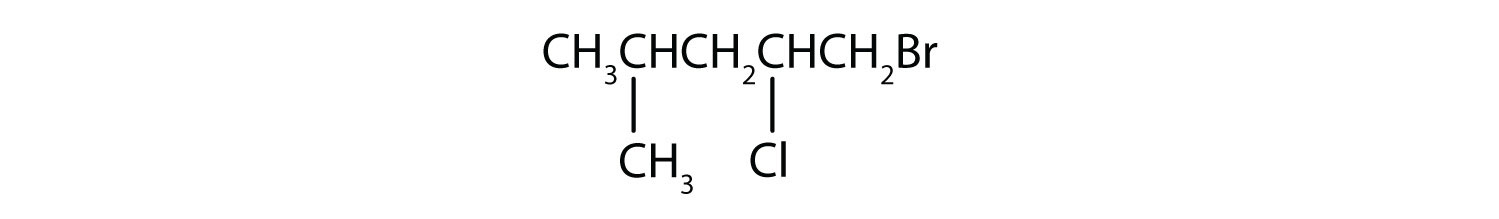
Give the IUPAC name for each compound.
Solution
- The parent alkane has five carbon atoms in the longest continuous chain; it is pentane. A bromo (Br) group is attached to the second carbon atom of the chain. The IUPAC name is 2-bromopentane.
- The parent alkane is hexane. Methyl (CH3) and bromo (Br) groups are attached to the second and fourth carbon atoms, respectively. Listing the substituents in alphabetical order gives the name 4-bromo-2-methylhexane.
Give the IUPAC name for each compound.
A wide variety of interesting and often useful compounds have one or more halogen atoms per molecule. For example, methane (CH4) can react with chlorine (Cl2), replacing one, two, three, or all four hydrogen atoms with Cl atoms. Several halogenated products derived from methane and ethane (CH3CH3) are listed in Table \(\PageIndex{1}\), along with some of their uses.
| Formula | Common Name | IUPAC Name | Some Important Uses |
|---|---|---|---|
| Derived from CH4 | |||
| CH3Cl | methyl chloride | chloromethane | refrigerant; the manufacture of silicones, methyl cellulose, and synthetic rubber |
| CH2Cl2 | methylene chloride | dichloromethane | laboratory and industrial solvent |
| CHCl3 | chloroform | trichloromethane | industrial solvent |
| CCl4 | carbon tetrachloride | tetrachloromethane | dry-cleaning solvent and fire extinguishers (but no longer recommended for use) |
| CBrF3 | halon-1301 | bromotrifluoromethane | fire extinguisher systems |
| CCl3F | chlorofluorocarbon-11 (CFC-11) | trichlorofluoromethane | foaming plastics |
| CCl2F2 | chlorofluorocarbon-12 (CFC-12) | dichlorodifluoromethane | refrigerant |
| Derived from CH3CH3 | |||
| CH3CH2Cl | ethyl chloride | chloroethane | local anesthetic |
| ClCH2CH2Cl | ethylene dichloride | 1,2-dichloroethane | solvent for rubber |
| CCl3CH3 | methylchloroform | 1,1,1-trichloroethane | solvent for cleaning computer chips and molds for shaping plastics |
Chlorofluorocarbons and Fluorocarbons
Chlorofluorocarbons (CFCs) and hydrochlorofluorocarbons (HCFCs) are fully or partly halogenated paraffin hydrocarbons that contain only carbon (C), hydrogen (H), chlorine (Cl), and fluorine (F), produced as volatile derivative of methane, ethane, and propane. They are also commonly known by the DuPont brand name Freon. The most common representative is dichlorodifluoromethane (R-12 or Freon-12). Many CFCs have been widely used as refrigerants, propellants (in aerosol applications), and solvents. Because CFCs contribute to ozone depletion in the upper atmosphere, the manufacture of such compounds has been phased out under the Montreal Protocol, and they are being replaced with other products such as hydrofluorocarbons (HFCs)
Chlorinated or fluorinated alkenes undergo polymerization. Important halogenated polymers include polyvinyl chloride (PVC), and polytetrafluoroethene (PTFE, or Teflon). Billions of kilograms of chlorodifluoromethane are produced annually as precursor to tetrafluoroethylene, the monomer that is converted into Teflon. The production of these materials releases substantial amounts of wastes. PVC and PTFE will be discussed in more detail in Chapter 10.
Summary
- Haloalkanes also known as alkyl halides or halogenoalkanes, are a group of organic compounds in which one or more halogen atoms are substituted for one or more hydrogen atoms in a hydrocarbon.
- These compounds are used as dry cleaning, industrial, and laboratory solvents, coolants in refrigerators and air conditioners, and propellants in hairsprays and deodorants
- The use of haloalkanes has been greatly curtailed due to their toxicity and harmful environmental effects.
- Naming of simple haloalkanes is described.
Contributors and Attributions
- Libretext: The Basics of GOB Chemistry (Ball et al.)
- Wikipedia