7.8: Acids and Bases in Industry and in Daily Life
- Page ID
- 153824
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Know the many uses of common acids and bases.
Antacids: A Basic Remedy
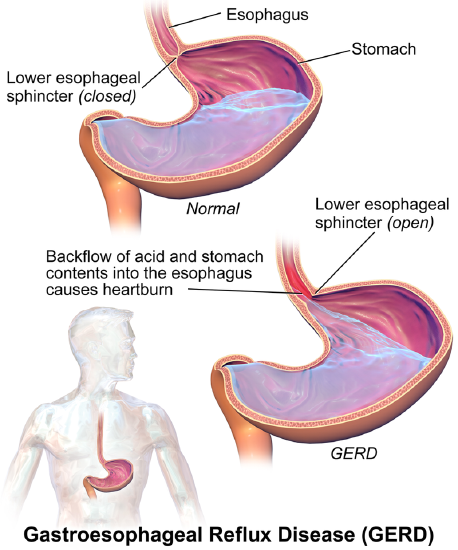
Antacids contain alkaline ions that chemically neutralize stomach gastric acid, reducing damage and relieving pain. For example, Tums is a very commonly used antacid that can be bought over the counter. Other antacids, such as Alka Seltzer, work in similar ways. Below, show the mechanism and products for reaction in which calcium carbonate (the active ingredient in Tums) neutralizes HCl in a simple proton transfer reaction.
CaCO3 (s) + HCl (aq) → H2CO3 (aq) + CaCl2 (aq)
Following this acid-base reaction, carbonic acid quickly degrades into CO2 and H2O.
H2CO3 (aq) → CO2(g) + H2O(l)
Figure \(\PageIndex{1}\) A comparison of a healthy condition to GERD. I
mage used with permission (Cc BY-SA 4.0; BruceBlaus .
Under the generic name algeldrate, aluminium hydroxide is used as an antacid. Aluminium hydroxide is preferred over other alternatives such as sodium bicarbonate because Al(OH)3, being insoluble, does not increase the pH of stomach above 7 and hence, does not trigger secretion of excess stomach acid. Brand names include Alu-Cap, Aludrox, Gaviscon, and Pepsamar. In 2016 Gaviscon was one of the biggest selling branded over-the-counter medications sold in Great Britain, with sales of £62 million. It reacts with excess acid in the stomach, reducing the acidity of the stomach content, which may relieve the symptoms of ulcers, heartburn or dyspepsia. Such products can cause constipation, because the aluminum ions inhibit the contractions of smooth muscle cells in the gastrointestinal tract, slowing peristalsis and lengthening the time needed for stool to pass through the colon. Some such products are formulated to minimize such effects through the inclusion of equal concentrations of magnesium hydroxide or magnesium carbonate, which have counterbalancing laxative effects.
Antacids are distinct from acid-reducing drugs like H2-receptor antagonists or proton pump inhibitors and they do not kill the bacteria Helicobacter pylori, which causes most ulcers. Ranitidine (Zantac), famotidine (Pepcid AC), and cimetidine (Tagamet) are in a class of medications called H2 blockers. It decreases the amount of acid made in the stomach and are used to treat ulcers; gastroesophageal reflux disease (GERD), a condition in which backward flow of acid from the stomach causes heartburn and injury of the food pipe (esophagus); and conditions where the stomach produces too much acid, such as Zollinger-Ellison syndrome. These over-the-counter medications are used to prevent and treat symptoms of heartburn associated with acid indigestion and sour stomach. Omeprazole (Prilosec) also works by decreasing the amount of acid made in the stomach and is in a class of medications called proton-pump inhibitors.
Acids and Bases in Industry and at Home
Common Acids
It should not be hard for you to name several common acids, but you might find that listing bases is just a little more difficult. Here's a partial list of some common acids, along with some chemical formulas:
|
Chemist Name
|
Common Name | Uses |
|---|---|---|
| hydrochloric acid, HCl | muriatic acid (used in pools) and stomach acid | Used in cleaning (refining) metals, in maintenance of swimming pools, and for household cleaning. |
| sulfuric acid, H2SO4 | Used in car batteries, and in the manufacture of fertilizers. | |
| nitric acid, HNO3 | Used in the manufacture of fertilizers, explosives and in extraction of gold. | |
| acetic acid, HC2H3O2 | vinegar | Main ingredient in vinegar. |
| carbonic acid, H2CO3 | responsible for the "fiz" in carbonated drinks | As an ingredient in carbonated drinks. |
| citric acid, C6H8O7 | Used in food and dietary supplements. Also added as an acidulant in creams, gels, liquids, and lotions. | |
| acetylsalicylic acid, C6H4(OCOCH3)CO2H | aspirin | The active ingredient in aspirin. |
Hydrochloric Acid
Hydrochloric acid is a corrosive, strong mineral acid with many industrial uses. One of the most important applications of hydrochloric acid is in the pickling of steel, to remove rust or iron oxide scale from iron or steel before subsequent processing. Another major use of hydrochloric acid is in the production of organic compounds, such as vinyl chloride and dichloroethane for PVC. Other organic compounds produced with hydrochloric acid include bisphenol A for polycarbonate, activated carbon, and ascorbic acid, as well as numerous pharmaceutical products. Other inorganic compounds produced with hydrochloric acid include road application salt calcium chloride, nickel(II) chloride for electroplating, and zinc chloride for the galvanizing industry and battery production.
Sulfuric Acid
Sulfuric acid is a very important commodity chemical, and indeed, a nation's sulfuric acid production is a good indicator of its industrial strength. World production in the year 2004 was about 180 million tonnes, with the following geographic distribution: Asia 35%, North America (including Mexico) 24%, Africa 11%, Western Europe 10%, Eastern Europe and Russia 10%, Australia and Oceania 7%, South America 7%. Most of this amount (≈60%) is consumed for fertilizers, particularly superphosphates, ammonium phosphate and ammonium sulfates. About 20% is used in chemical industry for production of detergents, synthetic resins, dyestuffs, pharmaceuticals, petroleum catalysts, insecticides and antifreeze, as well as in various processes such as oil well acidicizing, aluminium reduction, paper sizing, water treatment. About 6% of uses are related to pigments and include paints, enamels, printing inks, coated fabrics and paper, and the rest is dispersed into a multitude of applications such as production of explosives, cellophane, acetate and viscose textiles, lubricants, non-ferrous metals, and batteries.

Because the hydration of sulfuric acid is thermodynamically favorable (and is highly exothermic) and the affinity of it for water is sufficiently strong, sulfuric acid is an excellent dehydrating agent. Concentrated sulfuric acid has a very powerful dehydrating property, removing water (\(\ce{H2O}\)) from other compounds including sugar and other carbohydrates and producing carbon, heat, steam. Sulfuric acid behaves as a typical acid in its reaction with most metals by generating hydrogen gas (Equation \ref{Eq1}).
Nitric Acid
Nitric acid (\(\ce{HNO3}\)) is a highly corrosive mineral acid and is also commonly used as a strong oxidizing agent. Nitric acid is normally considered to be a strong acid at ambient temperatures. The main industrial use of nitric acid is for the production of fertilizers. Nitric acid is neutralized with ammonia to give ammonium nitrate. This application consumes 75–80% of the 26 million tonnes produced annually (1987). The other main applications are for the production of explosives, nylon precursors, and specialty organic compounds.
|
Some Common Bases
|
Uses |
|---|---|
| sodium hydroxide, NaOH (lye or caustic soda) |
Used in the manufacture of soaps and detergents and as the main ingredient in oven and drain cleaners. |
| potassium hydroxide, KOH (lye or caustic potash) |
Used in the production of liquid soaps and soft soaps. Used in alkaline batteries. |
| magnesium hydroxide, Mg(OH)2 (milk of magnesia) |
Used as an ingredient in laxatives, antacids, and deodorants. Also used in the neutralization of acidic wastewater. |
| calcium hydroxide, Ca(OH)2 (slaked lime) |
Used in the manufacture of cement and lime water. Also, added to neutralize acidic soil. |
| aluminum hydroxide | Used in water purification and as an ingredient in antacids. |
| ammonia, NH3 | Used as a building block for the synthesis of many pharmaceutical products and in many commercial cleaning products. Used in the manufacture of fertilizers. |
Sodium Hydroxide
Sodium hydroxide, also known as lye and caustic soda, is an inorganic compound with formula \(\ce{NaOH}\).
It is a popular strong base used in industry. Around 56% of sodium hydroxide produced is used by industry, 25% of which is used in the paper industry. Sodium hydroxide is also used in the manufacture of sodium salts and detergents, pH regulation, and organic synthesis. It is used in the Bayer process of aluminium production. In bulk, it is most often handled as an aqueous solution, since solutions are cheaper and easier to handle.
Sodium hydroxide is used in many scenarios where it is desirable to increase the alkalinity of a mixture, or to neutralize acids.

Calcium Hydroxide
Calcium hydroxide (traditionally called slaked lime) is an inorganic compound with the chemical formula \(\ce{Ca(OH)2}\). It is a colorless crystal or white powder. It has many names including hydrated lime, caustic lime, builders' lime, slaked lime, cal, or pickling lime.
One significant application of calcium hydroxide is as a flocculant, in water and sewage treatment. It is also used in fresh water treatment for raising the pH of the water so that pipes will not corrode where the base water is acidic, because it is self-regulating and does not raise the pH too much.
It is also used in the preparation of ammonia gas (NH3), using the following reaction:
- Ca(OH)2 + 2NH4Cl → 2NH3 + CaCl2 + 2H2O
Another large application is in the paper industry, where it is an intermediate in the reaction in the production of sodium hydroxide. Because of its low toxicity and the mildness of its basic properties, slaked lime is widely used in the food industry:
Ammonia
Ammonia is a compound of nitrogen and hydrogen with the formula \(\ce{NH3}\) and is a colorless gas with a characteristic pungent smell. It is the active product of “smelling salts,” and can quickly revive the faint of heart and light of head. Although common in nature and in wide use, ammonia is both caustic and hazardous in its concentrated form.
Globally, approximately 88% (as of 2014) of ammonia is used as fertilizers either as its salts, solutions or anhydrously. When applied to soil, it helps provide increased yields of crops such as maize and wheat. 30% of agricultural nitrogen applied in the US is in the form of anhydrous ammonia and worldwide 110 million tonnes are applied each year. Ammonia is also a building block for the synthesis of many pharmaceutical products and is used in many commercial cleaning products.
Summary
Common acids and bases have many and various uses in industry and in daily life.
Contributors and Attributions
Peggy Lawson (Oxbow Prairie Heights School). Funded by Saskatchewan Educational Technology Consortium.
- Wikipedia
- TextMap: Chemistry-A Molecular Approach (Tro)
Henry Agnew (UC Davis)
- MedlinePlus

