2.5: Mendeleev and Periodic Table
- Page ID
- 152144
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Explain how elements are organized into the periodic table.
- Describe how some characteristics of elements relate to their positions on the periodic table.
The Periodic Table is the symbol of Chemistry for many. It is a single image that contains all of the known elements in the universe combined into an easily legible table. There are many patterns present in the table as well. All of the elements seem to fit together, connecting to form the "image of chemistry". The idea of elements first came about in 3000 B.C. The great Greek philosopher Aristotle conceived an idea that everything on earth was made up of these elements. In ancient times, elements like gold and silver were readily accessible, however, the elements that Aristotle chose were Earth, Water, Fire, and Air.
Emerging Patterns and Periodicity within the Known Elements
The modern periodic table has evolved through a long history of attempts by chemists to arrange the elements according to their properties, as an aid in predicting chemical behavior. One of the first to suggest such an arrangement was the German chemist Johannes Dobereiner (1780–1849), who noticed that many of the known elements could be grouped into triads. A triad is a set of three elements that have similar properties—for example, chlorine, bromine, and iodine; copper, silver, and gold. Dobereiner proposed that all elements could be grouped in such triads, but subsequent attempts to expand his concept were unsuccessful.
By the mid-19th century, the atomic masses of many of the elements had been determined. Scientists like John Newlands and Alexandre-Emile Béguyer de Chancourtois formed their own versions of periodic tables. The English chemist John Newlands (1838–1898), who hypothesized that the chemistry of the elements might be related to their masses, arranged the known elements in order of increasing atomic mass and discovered that every seventh element had similar properties. Newlands therefore suggested that the elements could be classified into octaves. He described octaves as a group of seven elements, which correspond to the horizontal rows in the main groups of today's periodic table. There were seven elements, because the noble gases were not known at the time. Unfortunately, Newlands’s “law of octaves” did not seem to work for elements heavier than calcium, and his idea was publicly ridiculed.
The Periodic Table: Mendeleev and Meyer
The periodic table achieved its modern form through the work of the German chemist Julius Lothar Meyer (1830–1895) and the Russian chemist Dimitri Mendeleev (1834–1907), both of whom focused on the relationships between atomic mass and various physical and chemical properties. In 1869, they independently proposed essentially identical arrangements of the elements. Meyer aligned the elements in his table according to periodic variations in simple atomic properties, such as “atomic volume”, which he obtained by dividing the atomic mass (molar mass) in grams per mole by the density of the element in grams per cubic centimeter. This property is equivalent to what is today defined as molar volume—the molar mass of an element divided by its density (measured in cubic centimeters per mole):
\[ \frac{molar\; mass\left ( \cancel{g}/mol \right )}{density\left ( \cancel{g}/cm^{3} \right )}=molar\; volume\left ( cm^{3}/mol \right ) \tag{3.1.1} \]
Mendeleev, who first published his periodic table in 1869 (Figure \(\PageIndex{1}\) ), is usually credited with the origin of the modern periodic table. The key difference between his arrangement of the elements, and that of Meyer and others, is that Mendeleev did not assume that all the elements had been discovered (in fact, only about two-thirds of the naturally occurring elements were known at the time). Instead, he deliberately left blanks in his table at atomic masses 44, 68, 72, and 100—in the expectation that elements with those atomic masses would be discovered. Those blanks correspond to the elements we now know as scandium, gallium, germanium, and technetium.

The groups in Mendeleev's table are determined by how many oxygen or hydrogen atoms are needed to form compounds with each element. For example, in Group I, two atoms of hydrogen (H), lithium (Li), sodium (Na), and potassium (K) form compounds with one atom of oxygen. In Group VII, one atom of fluorine (F), chlorine (Cl), and bromine (Br), react with one atom of hydrogen. Notice how this approach has trouble with the transition metals. Until roughly 1960, a rectangular table based on reactivity—and developed from Mendeleev's table—was standard at the front of chemistry lecture halls.
The most convincing evidence in support of Mendeleev’s arrangement of the elements was the discovery of two previously unknown elements whose properties closely corresponded with his predictions (Table \(\PageIndex{1}\) ). Two of the blanks Mendeleev had left in his original table were below aluminum and silicon, awaiting the discovery of two as-yet-unknown elements, eka-aluminum and eka-silicon (from the Sanskrit eka, meaning “one,” as in “one beyond aluminum”). The observed properties of gallium and germanium matched those of eka-aluminum and eka-silicon so well that once they were discovered, Mendeleev’s periodic table rapidly gained acceptance.
Video \(\PageIndex{1}\): The genius of Mendeleev's periodic table.
| Property | eka-Aluminum (predicted) | Gallium (observed) | eka-Silicon (predicted) | Germanium (observed) |
|---|---|---|---|---|
| atomic mass | 68 | 69.723 | 72 | 72.64 |
| element | metal | metal | dirty-gray metal | gray-white metal |
| low mp* | mp = 29.8°C | high mp | mp = 938°C | |
| d = 5.9 g/cm3 | d = 5.91 g/cm3 | d = 5.5 g/cm3 | d = 5.323 g/cm3 | |
| oxide | E2O3 | Ga2O3 | EO2 | GeO2 |
| d = 5.5 g/cm3 | d = 6.0 g/cm3 | d = 4.7 g/cm3 | d = 4.25 g/cm3 | |
| chloride | ECl3 | GaCl3 | ECl4 | GeCl4 |
| volatile |
mp = 78°C bp* = 201°C |
bp < 100°C | bp = 87°C | |
| *mp = melting point; bp = boiling point. | ||||
When the chemical properties of an element suggested that it might have been assigned the wrong place in earlier tables, Mendeleev carefully reexamined its atomic mass. He discovered, for example, that the atomic masses previously reported for beryllium, indium, and uranium were incorrect. The atomic mass of indium had originally been reported as 75.6, based on an assumed stoichiometry of InO for its oxide. If this atomic mass were correct, then indium would have to be placed in the middle of the nonmetals, between arsenic (atomic mass 75) and selenium (atomic mass 78). Because elemental indium is a silvery-white metal, however, Mendeleev postulated that the stoichiometry of its oxide was really In2O3 rather than InO. This would mean that indium’s atomic mass was actually 113, placing the element between two other metals, cadmium and tin.
One group of elements that was absent from Mendeleev’s table is the noble gases, all of which were discovered more than 20 years later—between 1894 and 1898—by Sir William Ramsay (1852–1916; Nobel Prize in Chemistry 1904). Initially, Ramsay did not know where to place these elements in the periodic table. Argon, the first to be discovered, had an atomic mass of 40. This was greater than chlorine’s and comparable to that of potassium; so Ramsay, using the same kind of reasoning as Mendeleev, decided to place the noble gases between the halogens and the alkali metals. In 1913, however, young British physicist H. G. J. Moseley (1887–1915) analyzed the frequencies of x-rays emitted by the elements, and discovered that the underlying foundation of the order of the elements was by the atomic number—not the atomic mass. Moseley hypothesized that the placement of each element in his series corresponded to its atomic number Z, which is the number of positive charges (protons) in its nucleus. Moseley left his research work at the University of Oxford to join the British army as a telecommunications officer during World War I. He was killed during the Battle of Gallipoli in Turkey. Finally, in 1945, the Manhattan Project yielded the discovery of many new radioactive elements. Glenn T. Seaborg suggested an addition of the actinide and lanthanide series at the bottom of the table. This idea came with the discovery of Americium and Curium, and their unique properties. The change was not accepted at first, but is now included in all periodic tables. Figure \(\PageIndex{2}\) shows the time period for the discovery of the different elements.
- Before 1800 (36 elements): Discoveries during and before the Age of Enlightenment.
- 1800-1849 (+22 elements): Impulse from Scientific Revolution and Atomic theory and Industrial Revolution.
- 1850-1899 (+23 elements): The age of Classifying Elements received an impulse from the Spectrum analysis.
- 1900-1949 (+13 elements): Impulse from the old quantum theory, the Refinements to the periodic table, and quantum mechanics.
- 1950-1999 (+15 elements): Manhattan Project and Particle physics issues, for atomic numbers 97 and above.
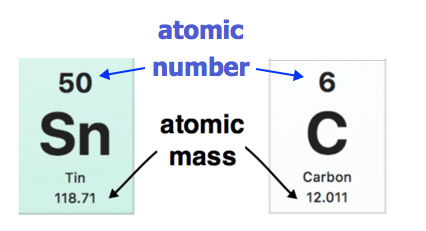
Each element on the modern periodic table is represented by its atomic number and atomic mass (Figure \(\PageIndex{3}\)). The atomic mass of each element is found under the element symbol in the periodic table. Examples are shown below. The atomic mass of tin (Sn) is 118.71 u while the atomic mass of carbon (C) is 12.011 u. On the other hand, the atomic number (Z) of each element is found above the atomic symbol.
The periodic table is found vis this link:
Summary
- The modern periodic table was based on empirical correlations of properties such as atomic mass; early models using limited data noted the existence of triads and octaves of elements with similar properties.
- The periodic table achieved its current form through the work of Dimitri Mendeleev and Julius Lothar Meyer, both of whom focused on the relationship between atomic mass and chemical properties.
- The correlation with the electronic structure of atoms was discovered when H. G. J. Moseley showed that the periodic arrangement of the elements was determined by atomic number, not atomic mass.
Contributors and Attributions
- Anonymous
Modified by Joshua Halpern (Howard University)
- Genius of Mendelev by TED Ed on YouTube
Ed Vitz (Kutztown University), John W. Moore (UW-Madison), Justin Shorb (Hope College), Xavier Prat-Resina (University of Minnesota Rochester), Tim Wendorff, and Adam Hahn.

