Origin of Color in Complex Ions
- Page ID
- 3707
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)This page is going to take a simple look at the origin of color in complex ions - in particular, why so many transition metal ions are colored.
White light and Colors
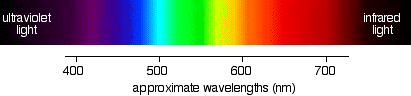
If you pass white light through a prism it splits into all the colors of the rainbow. Visible light is simply a small part of an electromagnetic spectrum most of which we cannot see - gamma rays, X-rays, infra-red, radio waves and so on. Each of these has a particular wavelength, ranging from 10-16 meters for gamma rays to several hundred metersfor radio waves. Visible light has wavelengths from about 400 to 750 nm. (1 nanometer = 10-9 meters.)
If white light (ordinary sunlight, for example) passes through copper(II) sulfate solution, some wavelengths in the light are absorbed by the solution. Copper(II) ions in solution absorb light in the red region of the spectrum. The light which passes through the solution and out the other side will have all the colors in it except for the red. We see this mixture of wavelengths as pale blue (cyan). The diagram gives an impression of what happens if you pass white light through copper(II) sulfate solution.
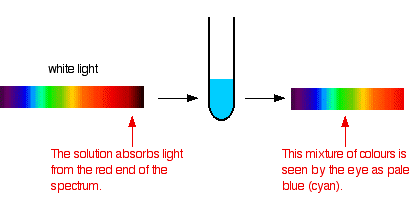
Working out what color you will see is not easy if you try to do it by imagining "mixing up" the remaining colors. You wouldn't have thought that all the other colors apart from some red would look cyan, for example. Sometimes what you actually see is quite unexpected. Mixing different wavelengths of light doesn't give you the same result as mixing paints or other pigments. You can, however, sometimes get some estimate of the color you would see using the idea of complementary colors.
If you arrange some colors in a circle, you get a "color wheel". The diagram shows one possible version of this.
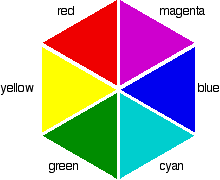
colors directly opposite each other on the color wheel are said to be complementary colors. Blue and yellow are complementary colors; red and cyan are complementary; and so are green and magenta. Mixing together two complementary colors of light will give you white light. What this all means is that if a particular color is absorbed from white light, what your eye detects by mixing up all the other wavelengths of light is its complementary color. Copper(II) sulfate solution is pale blue (cyan) because it absorbs light in the red region of the spectrum. Cyan is the complementary color of red.
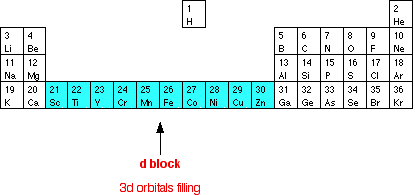
We often casually talk about the transition metals as being those in the middle of the Periodic Table where d orbitals are being filled, but these should really be called d block elements rather than transition elements (or metals). This shortened version of the Periodic Table shows the first row of the d block, where the 3d orbitals are being filled.
The usual definition of a transition metal is one which forms one or more stable ions which have incompletely filled d orbitals. Zinc with the electronic structure [Ar] 3d104s2 does not count as a transition metal whichever definition you use. In the metal, it has a full 3d level. When it forms an ion, the 4s electrons are lost - again leaving a completely full 3d level. At the other end of the row, scandium ( [Ar] 3d14s2 ) does not really counts as a transition metal either. Although there is a partially filled d level in the metal, when it forms its ion, it loses all three outer electrons. Technically, the Sc3+ ion does not count as a transition metal ion because its 3d level is empty.
The diagrams show the approximate colors of some typical hexaaqua metal ions, with the formula [ M(H2O)6 ] n+. The charge on these ions is typically 2+ or 3+.
Non-transition metal ions
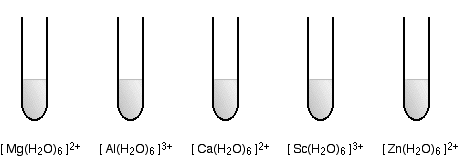
These ions are all colorless.
Transition metal ions
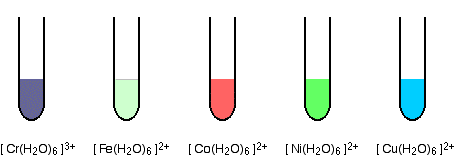
The corresponding transition metal ions are colored. Some, like the hexaaquamanganese(II) ion (not shown) and the hexaaquairon(II) ion, are quite faintly colored - but they are colored.
So, what causes transition metal ions to absorb wavelengths from visible light (causing color) whereas non-transition metal ions do not? And why does the color vary so much from ion to ion?
The Origin of Color in Complex Ions containing transition metals
Complex ions containing transition metals are usually colored, whereas the similar ions from non-transition metals are not. That suggests that the partly filled d orbitals must be involved in generating the color in some way. Remember that transition metals are defined as having partly filled d orbitals.
Octahedral Complexes
For simplicity we are going to look at the octahedral complexes which have six simple ligands arranged around the central metal ion. The argument is not really any different if you have multidentate ligands. When the ligands bond with the transition metal ion, there is repulsion between the electrons in the ligands and the electrons in the d orbitals of the metal ion. That raises the energy of the d orbitals. However, because of the way the d orbitals are arranged in space, it doesn't raise all their energies by the same amount. Instead, it splits them into two groups. The diagram shows the arrangement of the d electrons in a Cu2+ ion before and after six water molecules bond with it.
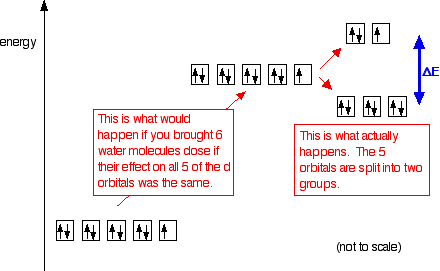
Whenever 6 ligands are arranged around a transition metal ion, the d orbitals are always split into 2 groups in this way - 2 with a higher energy than the other 3. The size of the energy gap between them (shown by the blue arrows on the diagram) varies with the nature of the transition metal ion, its oxidation state (whether it is 3+ or 2+, for example), and the nature of the ligands. When white light is passed through a solution of this ion, some of the energy in the light is used to promote an electron from the lower set of orbitals into a space in the upper set.

Each wavelength of light has a particular energy associated with it. Red light has the lowest energy in the visible region. Violet light has the greatest energy. Suppose that the energy gap in the d orbitals of the complex ion corresponded to the energy of yellow light.
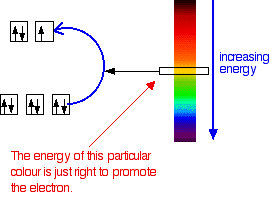
The yellow light would be absorbed because its energy would be used in promoting the electron. That leaves the other colors. Your eye would see the light passing through as a dark blue, because blue is the complementary color of yellow.
Tetrahedral Complexes
Simple tetrahedral complexes have four ligands arranged around the central metal ion. Again the ligands have an effect on the energy of the d electrons in the metal ion. This time, of course, the ligands are arranged differently in space relative to the shapes of the d orbitals. The net effect is that when the d orbitals split into two groups, three of them have a greater energy, and the other two a lesser energy (the opposite of the arrangement in an octahedral complex). Apart from this difference of detail, the explanation for the origin of color in terms of the absorption of particular wavelengths of light is exactly the same as for octahedral complexes.
Non-transition metals do not have partly filled d orbitals. Visible light is only absorbed if some energy from the light is used to promote an electron over exactly the right energy gap. Non-transition metals do not have any electron transitions which can absorb wavelengths from visible light. For example, although scandium is a member of the d block, its ion (Sc3+) hasn't got any d electrons left to move around. This is no different from an ion based on Mg2+ or Al3+. Scandium(III) complexes are colorless because no visible light is absorbed. In the zinc case, the 3d level is completely full - there are not any gaps to promote an electron in to. Zinc complexes are also colorless.
Factors Affecting the Color of Transition Metal complexes
In each case we are going to choose a particular metal ion for the center of the complex, and change other factors. color changes in a fairly haphazard way from metal to metal across a transition series.
The nature of the ligand
Different ligands have different effects on the energies of the d orbitals of the central ion. Some ligands have strong electrical fields which cause a large energy gap when the d orbitals split into two groups. Others have much weaker fields producing much smaller gaps. Remember that the size of the gap determines what wavelength of light is going to get absorbed. The list shows some common ligands. Those at the top produce the smallest splitting; those at the bottom the largest splitting.
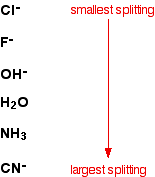
The greater the splitting, the more energy is needed to promote an electron from the lower group of orbitals to the higher ones. In terms of the color of the light absorbed, greater energy corresponds to shorter wavelengths. That means that as the splitting increases, the light absorbed will tend to shift away from the red end of the spectrum towards orange, yellow and so on.
There is a fairly clear-cut case in copper(II) chemistry. If you add an excess of ammonia solution to hexaaquacopper(II) ions in solution, the pale blue (cyan) color is replaced by a dark inky blue as some of the water molecules in the complex ion are replaced by ammonia.

The first complex must be absorbing red light in order to give the complementary color cyan. The second one must be absorbing in the yellow region in order to give the complementary color dark blue. Yellow light has a higher energy than red light. You need that higher energy because ammonia causes more splitting of the d orbitals than water does.
It is not often as simple to see as this, though! Trying to sort out what is being absorbed when you have murky colors not on the simple color wheel further up the page is much more of a problem. The diagrams show some approximate colors of some ions based on chromium(III).

It is obvious that changing the ligand is changing the color, but trying to explain the colors in terms of our simple theory is not easy.
The oxidation state of the metal
As the oxidation state of the metal increases, so also does the amount of splitting of the d orbitals. Changes of oxidation state therefore change the color of the light absorbed, and so the color of the light you see. Taking another example from chromium chemistry involving only a change of oxidation state (from +2 to +3):
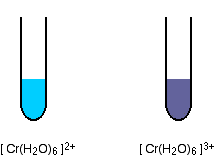
The 2+ ion is almost the same color as the hexaaquacopper(II) ion, and the 3+ ion is the hard-to-describe violet-blue-grey color.
The coordination of the Ion
Splitting is greater if the ion is octahedral than if it is tetrahedral, and therefore the color will change with a change of co-ordination. The problem is that an ion will normally only change co-ordination if you change the ligand - and changing the ligand will change the color as well. Hence, you cannot isolate out the effect of the co-ordination change. For example, a commonly quoted case comes from cobalt(II) chemistry, with the ions [Co(H2O)6]2+ and [CoCl4]2-.
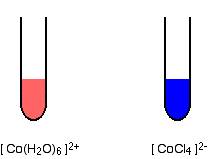
The difference in the colors is going to be a combination of the effect of the change of ligand, and the change of the number of ligands.
Contributors and Attributions
Jim Clark (Chemguide.co.uk)


