4.3: Phosphines
- Page ID
- 172740
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Learning Objectives
In this lecture you will learn the following
- Classification of ligands.
- Nature of bonding in phosphines.
- Steric and electronic properties of phosphines.
- Bonding in phosphines and CO.
- Cone angle and its application in catalysis.
Classification of Ligands by donor atoms
Ligand is a molecule or an ion that has at least one electron pair that can be donated. Ligands may also be called Lewis bases; in terms of organic chemistry, they are ‘nucleophiles’.
Metal ions or molecules such as BF3 (with incomplete valence electron shells (electron deficient) are called Lewis acids or electrophiles).
Why do molecules like H2O or NH3 give complexes with ions of both main group and transition metals. E.g [Al(OH2)6]3+ or [Co(NH3)6]3+
Why other molecules such as PF3 or CO give complexes only with transition metals.
Although PF3 or CO give neutral molecules such as Ni(PF3)4 or Ni(CO)4 or Cr(CO)6.
Why do, NH3, amines, oxygen donors, and so on, not give complexes such as Ni(NH3)4.
Classical or simple donor ligands
Act as electron pair donors to acceptor ions or molecules, and form complexes of all types of Lewis acids, metal ions or molecules.
Non-classical ligands, π-bonding or π-acid ligands: Form largely with transition metal atoms.
In this case special interaction occurs between the metals and ligands
These ligands act as both σ-donors and π-acceptors due to the availability of empty orbitals of suitable symmetry, and energies comparable with those of metal t2g (non-bonding) orbitals.
e.g. Consider PR3 and NH3: Both can act as bases toward H+, but P atom differs from N in that PR3 has σ* orbitals of low energy, whereas in N the lowest energy d orbitals or σ* orbitals are far too high on energy to use.
Consider CO that do not have measurable basicity to proton, yet readily reacts with metals like Ni that have high heats of atomization to give compounds like Ni(CO)4.
Ligands may also be classified electronically depending upon how many electrons that they contribute to a central atom. Atoms or groups that can form a single covalent bond are one electron donors.
EXAMPLES: F, SH, CH3 etc.,
Compounds with an electron pair are two-electron donors
EXAMPLE: NH3, H2O, PR3 etc.,
Bonding in Metal –Carbonyl and Metal-Phosphines
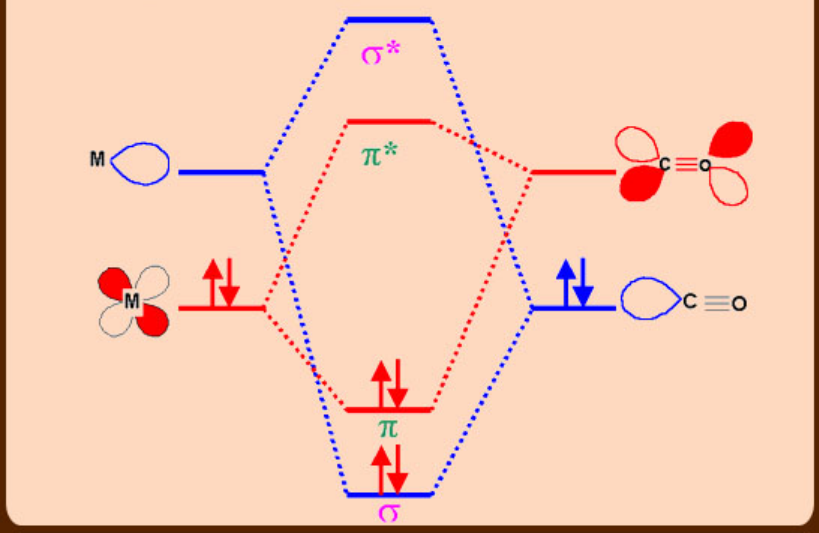
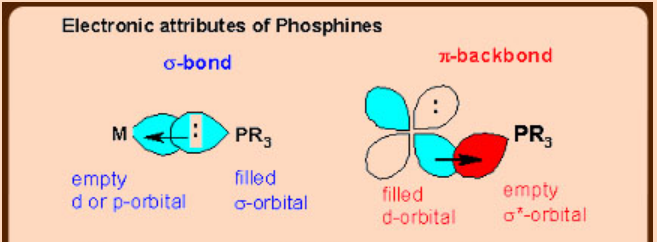
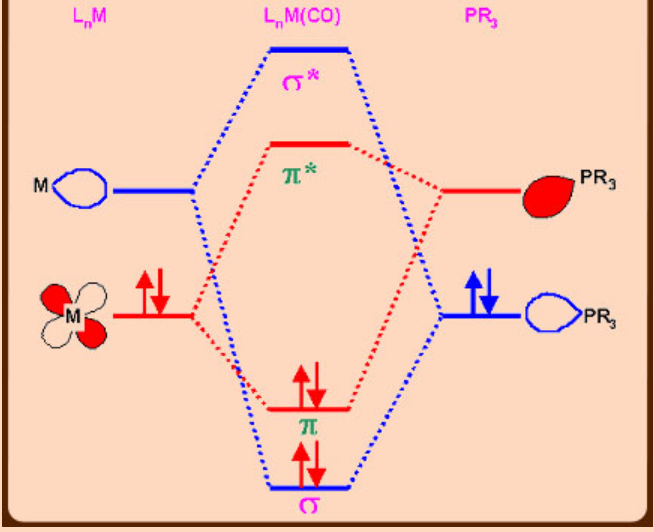
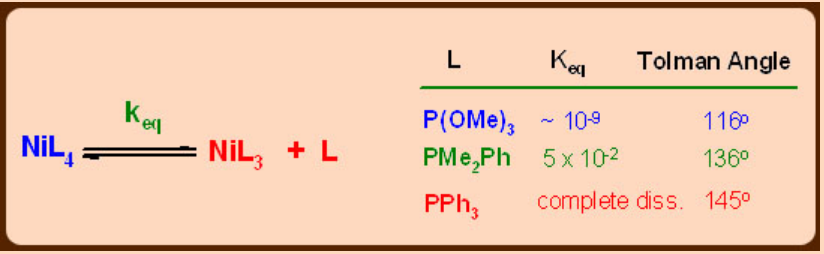
Steric factors in phosphines (Tolman’s cone angle)
Cone angle is very useful in assessing the steric properties of phosphines and their coordination behavior.
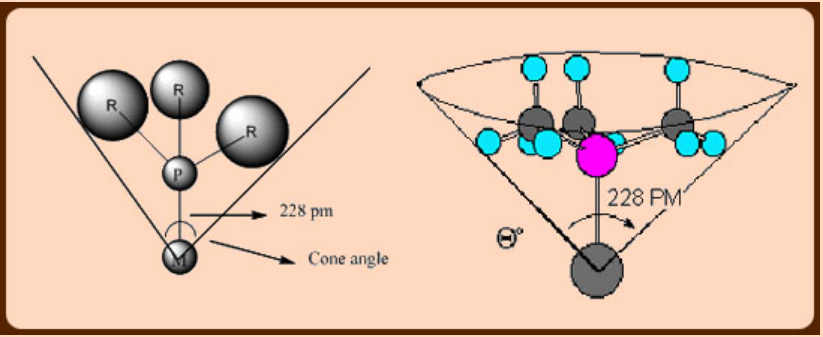
The electronic effect of phosphines can be assessed by IR and NMR spectroscopic data especially when carbonyls are co-ligands. In a metal complex containing both phosphines and carbonyl, the ν(CO) frequencies would reveal the σ-donor or π–acceptor abilities of phosphines. If the phosphines employed are strong σ-donors, then more electron density would move from M (t2g orbitals)- π*(CO) and as a result, a lowering in the ν(CO) is observed. In contrast, if a given phosphine is a poor σ-donor but strong π -acceptor, then phosphine(σ*-orbitals) also compete with CO for back bonding which results in less lowering in ν(CO) frequency.
Another important aspect is the steric size of PR3 ligands, unlike in the case of carbonyls, which can be readily tuned by changing R group. This is of great advantage in transition metal chemistry, especially in metal mediated catalysis, where stabilizing the metals in low coordination states is very important besides low oxidation states. This condition can promote oxidative addition at the metal centre which is an important step in homogeneous catalysis. The steric effects of phosphines can be quantified with Tolman’s cone angle.
Cone angle can be defined as a solid angle at metal at a M—P distance of 228 pm which encloses the van der Waal’s surfaces of all ligand atoms or substituents over all rotational orientations. The cone angles for most commonly used phosphines are listed in the following table.
|
Phosphine |
Cone Angle (°) |
|
PH3 |
87 |
|
PF3 |
104 |
|
P(OMe)3 |
107 |
|
PMe3 |
118 |
|
PMe2Ph |
122 |
|
PEt3 |
132 |
|
PPh3 |
145 |
|
PCy3 |
170 |
|
P(But)3 |
182 |
|
P(mesityl)3 |
212 |
Phosphines with different cone angles versus coordination number for group 8 metals:
|
ML4 |
ML3 |
ML2 |
|
(Me3P)4Ni |
||
|
(Me3P)4Pd |
||
|
(Me3P)4Pt |
(Ph3P)3Pt |
(tert-Bu3P)2Pt |
Tolman Angle and Catalysis
Sterically demanding phosphine ligands can be used to create an empty coordination site (16 VE complexes) which is an important trick to fine tune the catalytic activity of phosphine complexes.