14.4.1: Ziegler-Natta Polymerizations
- Page ID
- 385633
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Another example of an organometallic catalytic reaction is the Ziegler-Natta olefin polymerization. This reaction is of high industrial importance for the production of olefins like polyethylene. There are both heterogeneous and homogeneous Ziegler-Natta catalysts. The mechanism for the homogeneous catalysts is generally well understood. Homogeneous catalysts are typically metallocene catalysts.
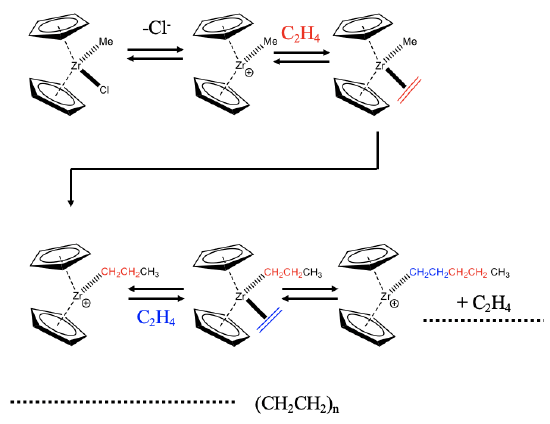
An example of a zirconium-based catalyst is shown (Fig. \(\PageIndex{1}\)). The catalyst is a coordinatively unsaturated complex cation with two cyclopentadienyl rings and a methyl group. The catalyst is formed from its precatalyst, a neutral molecule with an additional chloro ligand. The catalyst oxidatively adds an olefin like an ethylene molecule to the coordinatively unsaturated site. This step is followed an olefin insertion step that produces a propyl group. The migratory insertion leads to the formation of a vacant site, that can be re-oocupied by another ethylene molecule. This molecule can insert into the propyl chain thereby prolonging the propyl chain to a pentyl chain. The olefin insertion step generates another vacant site that can be reoccupied by a new ethylene molecule. Repeating the catalytic cycle many times eventually leads to polyethylene.
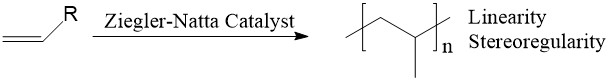
The Ziegler-Natta (ZN) catalyst, named after two chemists: Karl Ziegler and Giulio Natta, is a powerful tool to polymerize α-olefins with high linearity and stereoselectivity (Figure 1). A typical ZN catalyst system usually contains two parts: a transition metal (Group IV metals, like Ti, Zr, Hf) compound and an organoaluminum compound (co-catalyst). The common examples of ZN catalyst systems include TiCl4 + Et3Al and TiCl3 + AlEt2Cl.
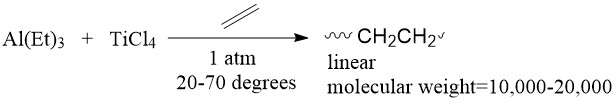
In 1953, German chemist Karl Ziegler discovered a catalytic system able to polymerize ethylene into linear, high molecular weight polyethylene which conventional polymerization techniques could not make.1 The system contained a transition metal halide with a main group element alkyl compound (Figure \(\PageIndex{3}\)).
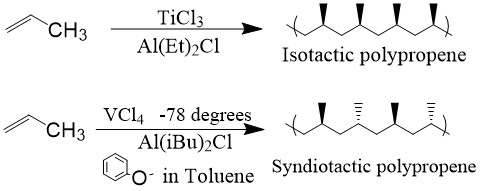
Following the catalytic design, Italian chemist Giulio Natta found that polymerization of α-olefins resulted in stereoregular structures,2 either syndiotactic or isotactic, depending on the catalyst used (Figure \(\PageIndex{4}\)). Because of these important discoveries, Karl Ziegler and Giulio Natta shared the Nobel Prize in Chemistry in 1963.
Advantages over traditional polymerization method
Traditionally, polymerization of α-olefins was done by radical polymerization (Figure \(\PageIndex{5}\)). Problem with this technique was that the formation of undesired allylic radicals leaded to branched polymers.3 For example, radical polymerization of propene gived branched polymers with large molecular weight distribution. Also, radical polymerization had no control over stereochemistry. Linear unbranched polyethylene and stereoregulated polypropylene could not be fabricated by free radical polymerization. This technique largely limited the potential applications of these polymeric materials.
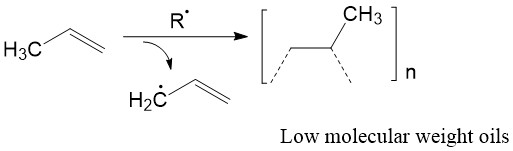
The invention of ZN catalyst successfully addressed these two problems. The catalyst can give linear α-olefin polymers with high and controllable molecular weights. Moreover, it makes the fabrication of polymers with specific tacticity possible. By controlling the stereochemistry of products, either syndiotactic or isotactic polymers can be achieved.
Mechanism of Ziegler-Natta catalytic polymerization
Activation of Ziegler-Natta catalyst
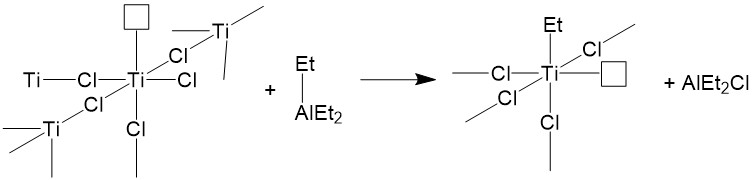
It is necessary to understand the catalyst’s structure before understanding how this catalyst system works. Herein, TiCl4+AlEt3 catalyst system is taken as an example. The titanium chloride compound has a crystal structure in which each Ti atom is coordinated to 6 chlorine atoms. On the crystal surface, a Ti atom is surrounded by 5 chlorine atoms with one empty orbital to be filled. When Et3Al comes in, it donates an ethyl group to Ti atom and the Al atom is coordinated to one of the chlorine atoms. Meanwhile, one chlorine atom from titanium is kicked out during this process. Thus, the catalyst system still has an empty orbital (Figure \(\PageIndex{6}\)). The catalyst is activated by the coordination of AlEt3 to Ti atom.
Initiation step
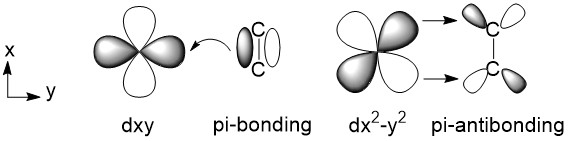
The polymerization reaction is initiated by forming alkene-metal complex. When a vinyl monomer like propylene comes to the active metal center, it can be coordinated to Ti atom by overlapping their orbitals. As shown in Figure \(\PageIndex{7}\), there is an empty dxy orbital and a filled dx2-y2 orbital in Ti’s outermost shell (the other four orbitals are not shown here). The carbon-carbon double bond of alkene has a pi bond, which consists of a filled pi-bonding orbital and an empty pi-antibonding orbital. So, the alkene's pi-bonding orbital and the Ti's dxy orbital come together and share a pair of electrons. Once they're together, that Ti’s dx2-y2 orbital comes mighty close to the pi-antibonding orbital, sharing another pair of electrons.
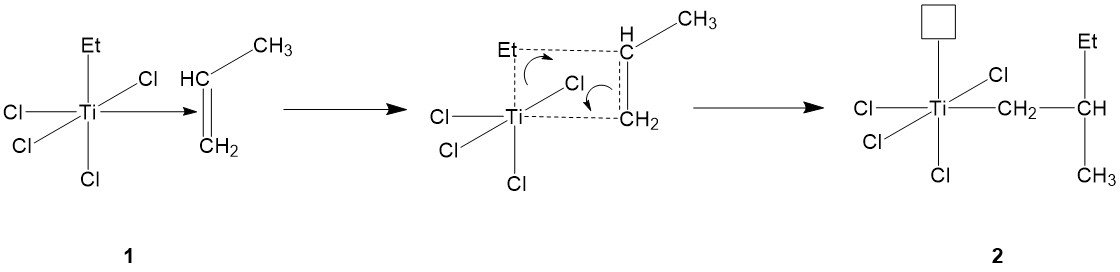
The formed alkene-metal complex (1) then goes through electron shuffling, with several pairs of electrons shifting their positions: The pair of electrons from the carbon-carbon pi-bond shifts to form Ti-carbon bond, while the pair of electrons from the bond between Ti and AlEt3’ ethyl group shifts to form a bond between the ethyl group and the methyl-substituted carbon of propylene (Figure \(\PageIndex{8}\)). After electron shuffling, Ti is back with an empty orbital again, needing electrons to fill it (2).
Propagation step
When other propylene molecules come in, this process starts over and over, giving linear polypropylene (Figure \(\PageIndex{9}\)).
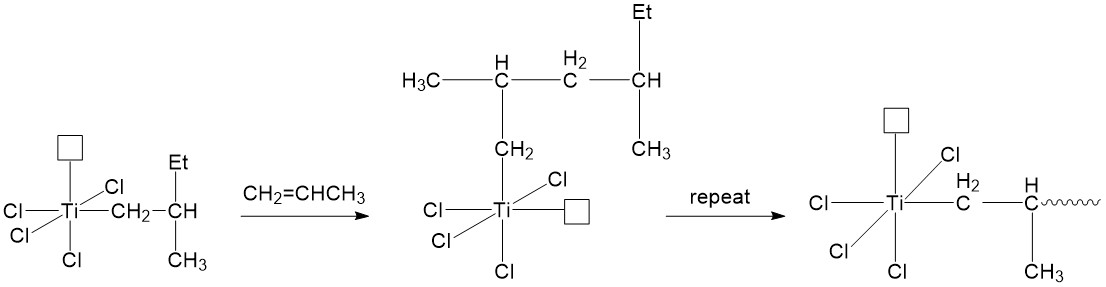
Termination step
Termination is the final step of a chain-growth polymerization, forming “dead” polymers (desired products). Figure \(\PageIndex{10}\) illustrates several termination approaches developed with the aid of co-catalyst AlEt3.4
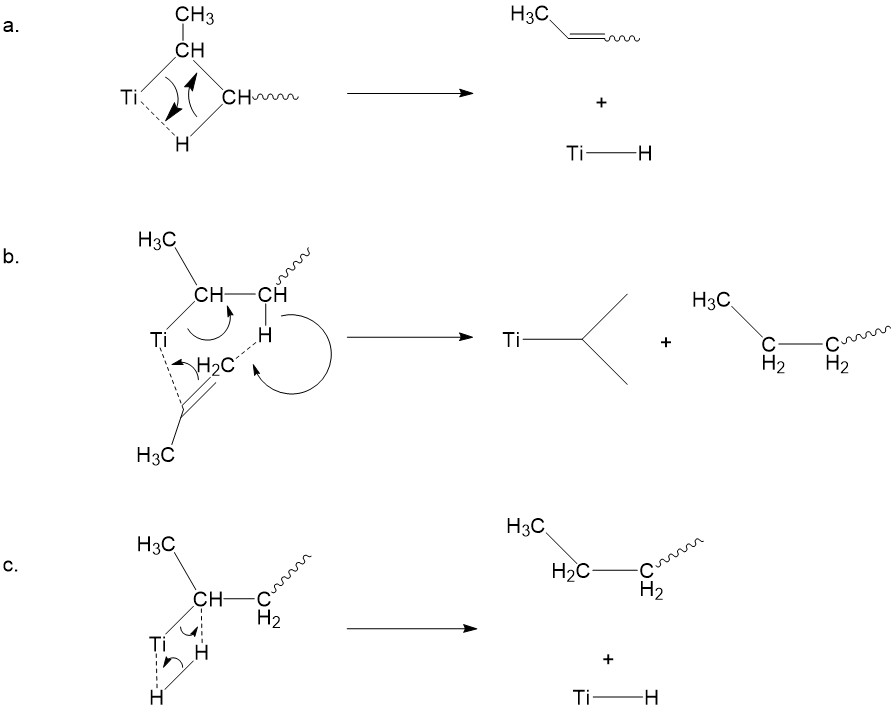
Mechanistic study: kinetic isotope effect experiments
Unlike the mechanism discussed above, there is also a competing mechanism proposed by Ivin and coworkers.5 They proposed that a 1,2-hydrogen shift occurs prior to monomer association, giving a carbene intermediate (Figure \(\PageIndex{11}\)).
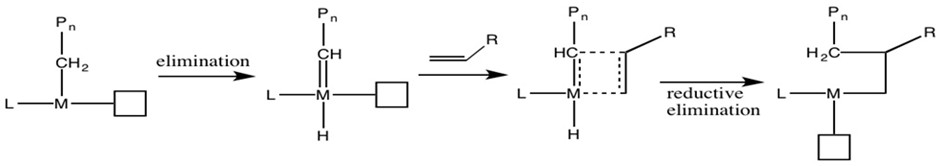
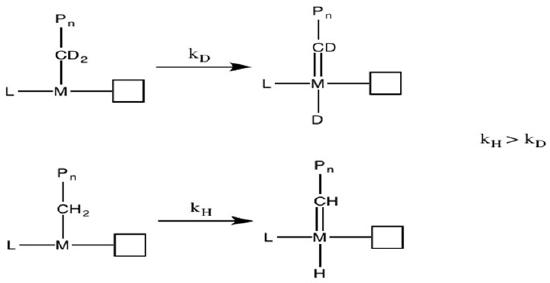
To determine the actual mechanism, Grubbs6 conducted kinetic isotope effect (KIE) experiments. Due to their different weights, carbon-deuterium bond reacts slower than carbon-hydrogen bond (Figure \(\PageIndex{12}\)). If such bonds are involved in the rate-determining step, the isotopic species should proceed in lower rate.
In Grubbs’s experiment, deuterated ethylene and normal ethylene were catalyzed by Ziegler-Natta catalyst with 1:1 ratio (Figure \(\PageIndex{13}\)). The result showed that H/D ratio in the resulting polymers was still 1:1.6 This indicated that no carbon-hydrogen bond cleavage or formation is involved in the rate determining step. Therefore, the mechanism proposed by Ivin was excluded.

Regio- and stero-selectivity
Regio-selectivity
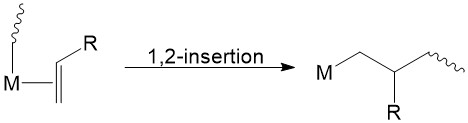
For propene polymerization, most ZN catalysts are highly regioselective, favoring 1,2-primary insertion [M-R + CH2=CH(Me)=M-CH2-CH(Me)(R)] (Figure \(\PageIndex{14}\)), due to electronic and steric effects.7
Stereo-selectivity
Stereochemistry of polymers made from ZN-catalyst can be well regulated by rational design of ligands. By using different ligand system, either syndiotactic or isotactic polymers can be obtained (Figure \(\PageIndex{15}\)).
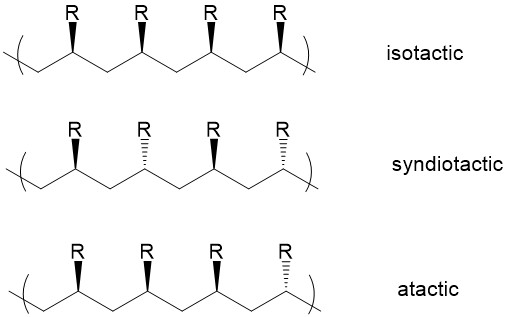
The relative stereochemistry of adjacent chiral centers within a macromolecule is defined as tacticity. Three kinds of stereochemistry are possible: isotactic, syndiotactic and atactic. In isotactic polymers, substituents are located on the same side of the polymer backbone, while substituents on syodiotactic polymers have alternative positions. In atactic polymers, substituents are placed randomly along the chain.
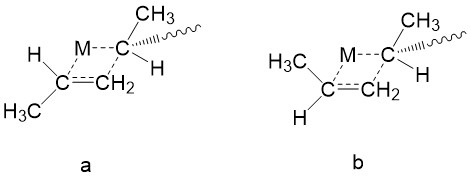
Choice of ZN catalyst regulates the stereochemistry. We use propylene polymerization as an example here. Recall the mechanism section, a monomer approaches the metal center and forms a four-membered ring intermediate. The binding of a monomer to the reactive metal-carbon bond should occur in from the least hindered site.8 As shown in Figure \(\PageIndex{16}\), the trans-complex in which methyl on the growing chain is trans to the methyl group on the incoming monomer, should be energetically favored than the cis-complex, as the trans-complex experience less steric effect.
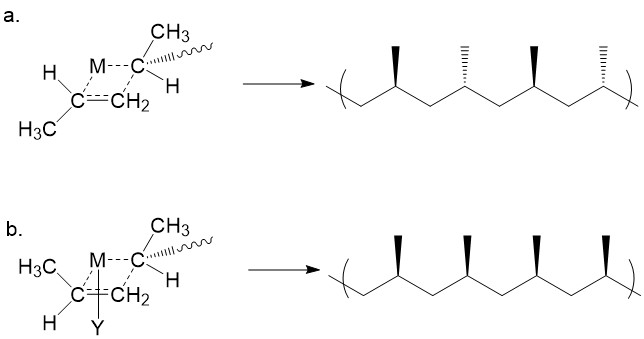
Following the trans- complex, the methyl group on the newly added monomer is trans to that on the previous monomer. The step repeats so that a syndiotactic polypropylene is obtained (Figure \(\PageIndex{17}\)a). However, if the metal center is coordinated with bulky ligands (e.g. –iBu, -Et2 groups), as denoted by Y in Figure \(\PageIndex{17}\)b, the incoming monomer will adopt a cis-conformation to avoid serious steric effect with the bulky ligand. Thus, at the presence of bulky ligand, the propylene monomer is cis to the growing chain. This results in a isotactic product.
Applications
ZN catalysts have provided a worldwide profitable industry with production of more than 160 billion pounds and creation of numerous positions.9 These products have been extensively applied in different areas, largely improving the quality of people’s life. They can catalyze α-olefins to produce various commercial polymers, like polyethylene, polypropylene and Polybutene-1. Polyethylene and polypropylene is reported to be the top two widely used synthetic plastic in the word.10
Among the polyethylene fabricated by ZN catalysts, there are three major classes: high density polyethylene (HDPE), linear low density polyethylene (LLDPE), and ultra-high molecular weight polyethylene (UHMWPE). HDPE, a linear homopolymer, is widely applied in garbage containers, detergent bottles and water pipes because of its high tensile strength. Compared with HDPE whose branching degree is quite low, LLDPE has many short branches. Its better toughness, flexibility and stress-cracking resistance makes it suitable for materials like cable coverings, bubble wrap and so on. UHMWPE is polyethylene with a molecular weight between 3.5 and 7.5 million. This material is extremely tough and chemically resistant. Therefore, it is often used to make gears and artificial joints.
Compared to polyethylene, polypropylene has enhanced mechanical properties and thermal resistance because of the additional methyl group. Moreover, isotactic polypropylene is stiffer and more resistant to creep than atactic polypropylene. Polypropylene has a wide range of applications in clothing, medical plastics, food packing, and building construction.
Substrate scope and limitations
ZN catalysts are effective for polymerization of α-Olefins (ethylene, propylene) and some dienes (butadiene, isoprene). However, they don’t work for some other monomers, such as 1.2 disubstituted double bonds. Vinyl chloride cannot be polymerized by ZN catalyst either, because free radical vinyl polymerization is initiated during the reaction. Another situation that ZN catalysts don’t work is when the substrate is acrylate. The reason is that ZN catalysts often initiate anionic vinyl polymerization in those monomers.11
References
- Ziegler, K.; Holzkamp, E.; Breil, H.; Martin, H., Polymerisation Von Athylen Und Anderen Olefinen. Angew Chem Int Edit 1955, 67 (16), 426-426.
- Natta, G., Une Nouvelle Classe De Polymeres D Alpha-Olefines Ayant Une Regularite De Structure Exceptionnelle. J Polym Sci 1955, 16 (82), 143-154.
- Mulhaupt, R., Catalytic polymerization and post polymerization catalysis fifty years after the discovery of Ziegler's catalysts. Macromol Chem Phys 2003, 204 (2), 289-327.
- Sinn, H.; Kaminsky, W., Ziegler-Natta Catalysis. In Advances in Organometallic Chemistry, Stone, F. G. A.; Robert, W., Eds. Academic Press: 1980; Vol. Volume 18, pp 99-149.
- Ivin, K. J.; Rooney, J. J.; Stewart, C. D.; Green, M. L.; Mahtab, R., Mechanism for the stereospecific polymerization of olefins by Ziegler–Natta catalysts. Journal of the Chemical Society, Chemical Communications 1978, (14), 604-606.
- Soto, J.; Steigerwald, M. L.; Grubbs, R. H., Concerning the Mechanism of Ziegler-Natta Polymerization - Isotope Effects on Propagation Rates. J Am Chem Soc 1982, 104 (16), 4479-4480.
- Busico, V.; Cipullo, R.; Pellecchia, R.; Ronca, S.; Roviello, G.; Talarico, G., Design of stereoselective Ziegler-Natta propene polymerization catalysts. P Natl Acad Sci USA 2006, 103 (42), 15321-15326.
- Zambelli, A., Stereoregulation of Propylene Polymerization. Abstr Pap Am Chem S 1974, 34-34.
- Ittel, S. D.; Johnson, L. K.; Brookhart, M., Late-metal catalysts for ethylene homo- and copolymerization. Chem Rev 2000, 100 (4), 1169-1203.
- Macbride, R. R., High Molecular-Weight Polyethylene Drums Show Signs of Becoming Big Business. Mod Plast 1975, 52 (12), 10-&.
- Ivan, B., Comparison of Living Polymerization Systems. Macromol Symp 1994, 88, 201-215.
Contributors and Attributions
- Fangyuan Dong and Ru Deng


