6.4.3: The electronic spectra of charge transfer complexes illustrate the impact of frontier orbital interactions on the electronic structure of Lewis acid-base adducts
- Page ID
- 162903
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
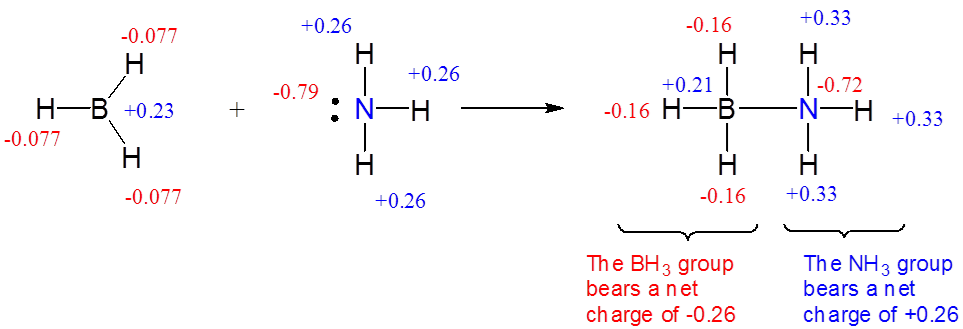
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)When a Lewis acid-base adduct is formed, electron density and negative charge are transferred from the Lewis base to the acid.
The formation of a Lewis-acid base complex involves the transfer of electron density from the base to the acid.
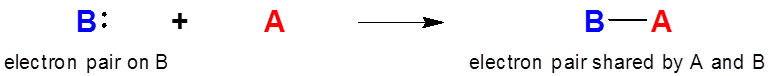 \[ \nonumber \]
\[ \nonumber \]
 \[ \nonumber \]
\[ \nonumber \]
Weakly bound Lewis acid-base adducts in which charge is incompletely transferred are called charge transfer complexes.
In many weakly bound Lewis acid-base complexes, the transfer of electron density and, consequently, charge from the base group to the acid group is only partial:
\[A~+~B~\rightleftharpoons~A^{\delta-}B^{\delta+} \nonumber \]
Such Lewis acid-base adducts are commonly called charge transfer complexes (CT complexes) or donor-acceptor complexes (DA complexes). In these
- the base is called the donor (D) since it is a net donor of electrons and, consequently, their negative charge
- the acid is called the acceptor (A) since it is a net acceptor of electrons and, consequently, their negative charge
A particularly well-known class of charge transfer complexes is the iodine charge transfer complexes. In iodine charge transfer complexes the \(I_2\) acts as a Lewis acid. This is possible since iodine is a Row 3+ element and so is capable of forming hypervalent complexes on reaction with a Lewis base. For example, \(I_2\) reacts with \(I^-\) to give the triiodide ion.
 \[ \nonumber \]
\[ \nonumber \]
Triiodide is well-known from introductory chemistry from the bright blue color that appears when the triiodide complexes with starch to give the dark blue starch-iodide complex.
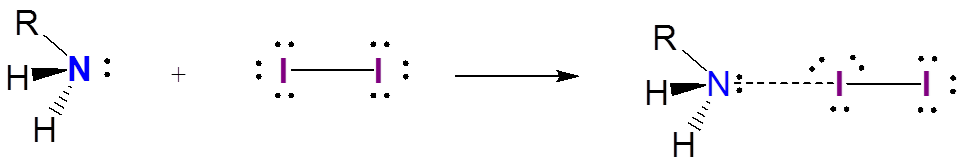
In contrast to the stable triiodide anion, iodine charge transfer complexes are only weakly associated. Complexes between iodine and amines are a well known example:
 \[ \nonumber \]
\[ \nonumber \]
Iodine also forms weakly-associated charge transfer complexes with many solvents. For instance, iodine can weakly associate with both acetone and benzene:
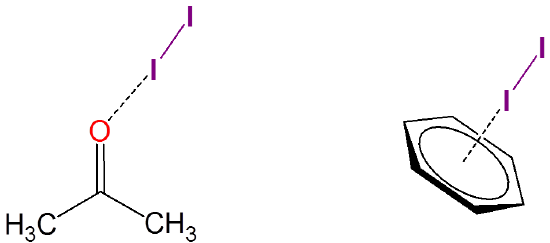
It is worth noting in passing that several organic charge transfer complex systems have been developed in which organic electron donors and acceptors weakly associate with each other. These are of considerable interest for use in molecular electronics applications, and as a result, a large variety of organic electron donors and acceptors have been developed. An example of an organic electron acceptor is tetracyanoethylene (TCNE):
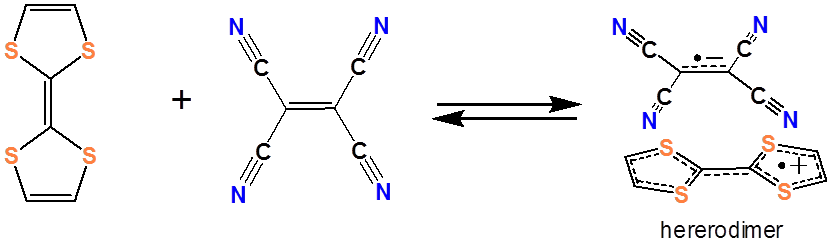
and an example of an organic electron acceptor is tetrathiofulvalene (TTF).
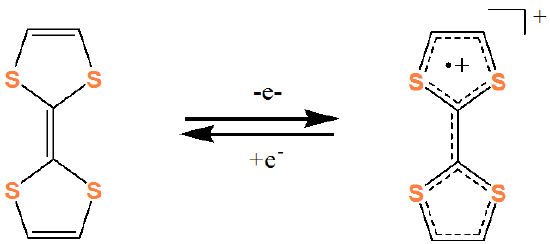
Together they form a charge transfer complex:
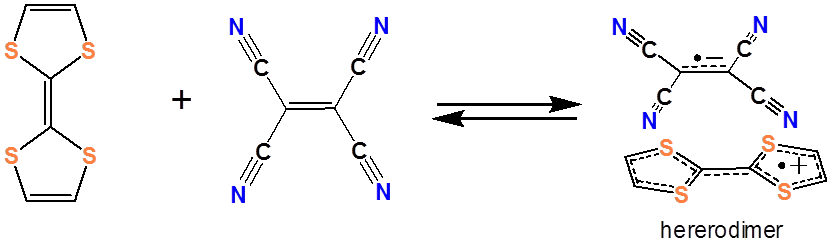
Similar organic charge transfer systems have been investigated for use in organic photovoltaic cells. There is considerable interest in developing such organic photovoltaic devices owing to the ease with which organic materials may be fabricated from organic solutions and suspensions.
Charge transfer complexes exhibit charge transfer transitions in which absorption triggers the transfer of an electron from the donor to the acceptor.
When iodine is dissolved in solutions of donor solvents, the striking purple color of molecular iodine is replaced by a yellow-brown color. This is because charge transfer complexes like those formed by \(I_2\) can absorb light in ways that neither the donor nor the acceptor can on their own. Specifically, charge transfer complexes exhibit charge transfer bands (CT bands) in their absorption spectra. In the charge transfer transition the initial partial transfer of charge from the donor Lewis base to the acceptor Lewis acid in the charge transfer complex is pushed further by photoexcitation.
\[D^{\delta+}-A^{\delta-}~\overset{h\nu_{CT}}{~\longrightarrow}~D^+-A^- \nonumber \]
The nature of these charge transfer transitions is seen from the orbital description of binding for iodine charge transfer complexes. When a donor-I2 complex forms, the formation of donor-I2 bonding and antibonding orbitals results in a shift in the \(I2 \sigma \rightarrow \sigma*\) transition to higher energy, as a new charge transfer band is formed, associated with the excitation of an electron from the largely amine-centered amine-I2 \(\sigma \) orbital to the largely I2-centered amine-I2 \(\sigma *\) orbital.
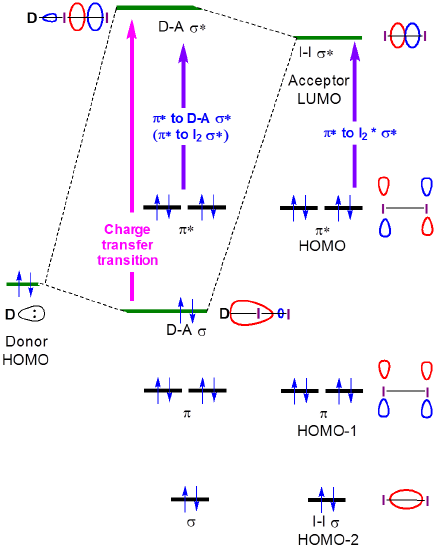
Solutions of I2 as mixtures with Lewis bases such as amines and in donor solvents clearly have charge transfer bands in their absorption spectra. Several such spectra are given in Figure 6.4.2.3.
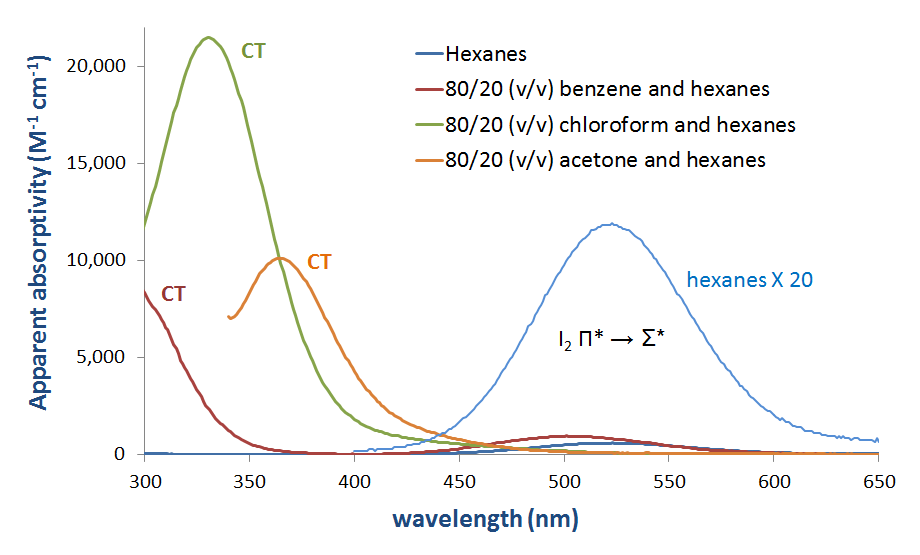
In principle, the energies of both the charge transfer and \(I_2~\pi *~\rightarrow ~D-A~ \sigma *\) transitions both increase with donor strength, as shown in Figure 6.4.2.4.
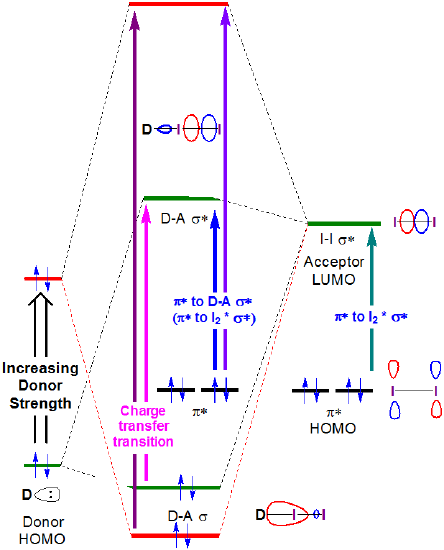
As can be seen in Figure 6.4.2, the charge transfer band energy might be expected to increase as the donor HOMO increases in energy to become closer in energy to the acceptor LUMO. Although care should be taken when interpreting the solution phase spectra of I2, this expectation is borne out by a cursory and qualitative analysis of the spectra in Figure 6.4.2.3. The CT transition energy shifts towards lower wavelengths (and thus higher energy) as the highest occupied atomic orbital energy for the donor atom increases on going from acetone (oxygen, -15.85 eV) to chloroform (chlorine, -13.67eV) and finally benzene (carbon, -10.66 eV).**
Charge transfer transitions are responsible for the intense color of many transition metal complexes. In these cases, however, the weak Lewis acid-base interaction involves incomplete electron donation and acceptance in a \(p \pi -d \pi\) (or \(p\pi-d\pi*\))-bond between a metal and ligand. The charge transfer bands in the absorption spectra of these complexes involve the transfer of electrons between the metal and ligand. In particular,
- Metal to ligand charge transfer (MLCT or CTTL) bands involve the transfer of an electron from a filled or partly filled metal d orbital to a ligand \(\pi*\)-type orbital.

- Ligand to metal charge transfer (LMCT or CTTM) bands involve the transfer of an electron from a filled or partly filled ligand orbital to a metal d-orbital.

- Metal to metal charge transfer bands can be observed in some bimetallic complexes. However, these are usually thought of only as an electron transfer rather than as a shift in the status of a Lewis acid-base interaction.
Because metal-ligand charge transfer bands involve intermolecular electron transfer between the metal and ligand to generate a high energy redox state, the CT excited state is both a better oxidant and reductant than the ground state. Consequently there has been intense research into the development of metal complexes whose charge transfer excited states are powerful oxidants and reductants in the expectation that they will be able to drive the photocatalytic oxidation and reduction of substrates.
* The apparent absorptivity of I2 in hexanes was calculated from the absorption spectrum of 215 µM I2 in hexanes. All other apparent absorptivities were calculated from absorption spectra of solutions that were 44 µM in I2.
** The band positions are not the CT band energies, and the HOMO energies given are atomic energy levels and do not necessarily correspond to the HOMO of the donor in solution. Because of this and other simplifications this analysis is not intended to replace a rigorous computational analysis of the factors that give rise to CT band positions.
References
- Meyerstein, D.; Treinin, A., Charge-transfer complexes of iodine and inorganic anions in solution. Transactions of the Faraday Society 1963, 59 (0), 1114-1120.
- Baskar, A. J. A.; Rajpurohit, A. S.; Panneerselvam, M.; Jaccobb, M.; RoopSingh, D.; Kannappan, V., Experimental and theoretical analysis of substituent effect on charge transfer complexes of iodine and some alkylbenzenes in n-hexane solution at 303K. Chemical Data Collections 2017, 7-8, 80-92.

