3.6: Names and Formulas of Inorganic Compounds
- Page ID
- 13791
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- To name ionic compounds.
The empirical and molecular formulas discussed in the preceding section are precise and informative, but they have some disadvantages. First, they are inconvenient for routine verbal communication. For example, saying “C-A-three-P-O-four-two” for Ca3(PO4)2 is much more difficult than saying “calcium phosphate.” In addition, many compounds have the same empirical and molecular formulas but different arrangements of atoms, which differences result in very different chemical and physical properties. In such cases, it is necessary for the compounds to have different names that distinguish among the possible arrangements.
Many compounds, particularly those that have been known for a relatively long time, have more than one name: a common name (sometimes several), and a systematic name, which is the name assigned by adhering to specific rules. Like the names of most elements, the common names of chemical compounds generally have historical origins, although they often appear to be unrelated to the compounds of interest. For example, the systematic name for KNO3 is potassium nitrate, but its common name is saltpeter.
In this text, a systematic nomenclature is used to assign meaningful names to the millions of known substances. Unfortunately, some chemicals that are widely used in commerce and industry are still known almost exclusively by their common names; in such cases, familiarity with the common name as well as the systematic one is required. The objective of this and the next two sections is to teach how to write the formula for a simple inorganic compound from its name—and vice versa—and introduce some frequently-encountered common names.
Binary ionic compounds contain only two elements. The procedure for naming such compounds is outlined in Figure \(\PageIndex{1}\) and uses the following steps:
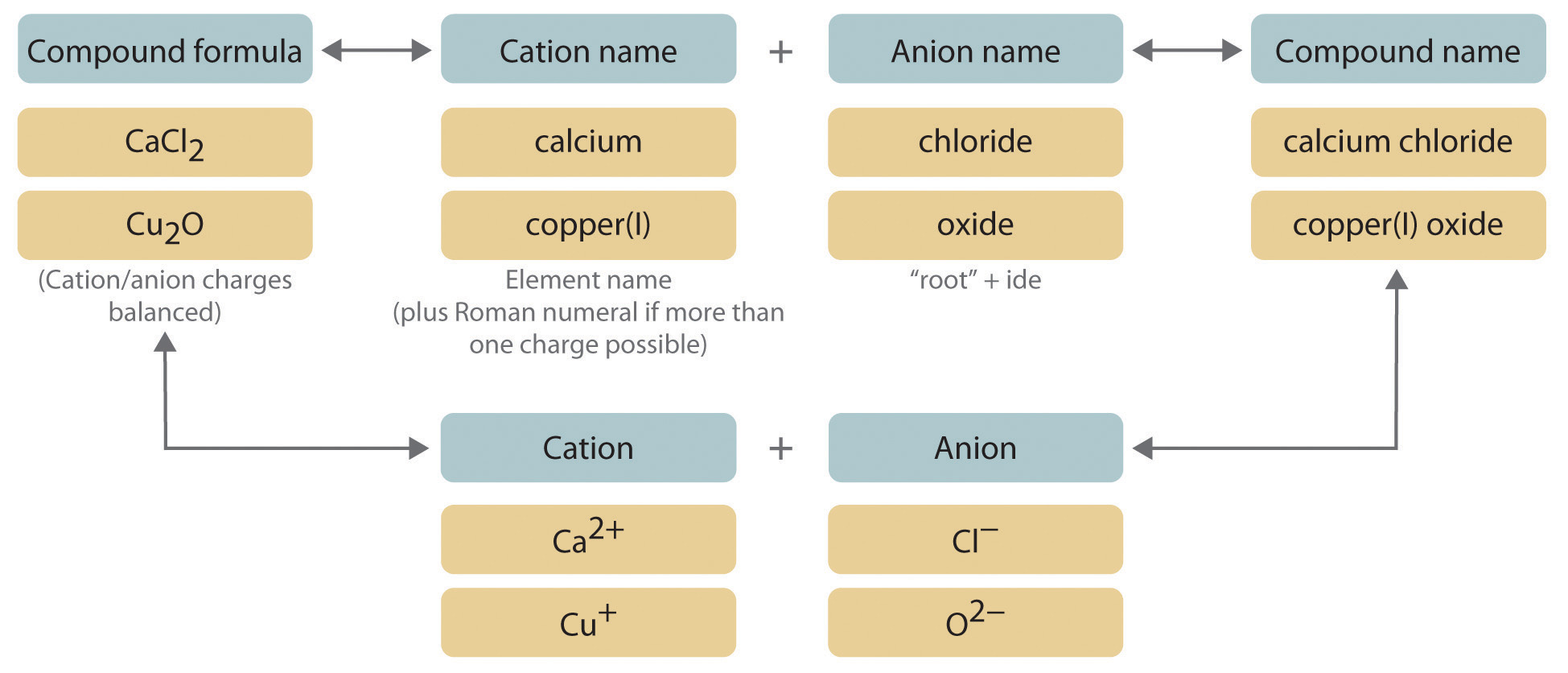
1. Place the ions in their proper order: cation and then anion.
2. Name the cation.
a. Metals that form only one cation. As noted previosuly, these metals are usually in Groups 1–3, 12, and 13. The name of the cation of a metal that forms only one cation is the same as the name of the metal (with the word ion added if the cation is by itself). For example, Na+ is the sodium ion, Ca2+ is the calcium ion, and Al3+ is the aluminum ion.
b. Metals that form more than one cation. As shown in Figure \(\PageIndex{2}\), many metals can form more than one cation. This behavior is observed for most transition metals, many actinides, and the heaviest elements of Groups 13–15. In such cases, the positive charge on the metal is indicated by a roman numeral in parentheses immediately following the name of the metal. Thus Cu+ is copper(I) (read as “copper one”), Fe2+ is iron(II), Fe3+ is iron(III), Sn2+ is tin(II), and Sn4+ is tin(IV).
An older system of nomenclature for such cations is still widely used, however. The name of the cation with the higher charge is formed from the root of the element’s Latin name with the suffix -ic attached, and the name of the cation with the lower charge has the same root with the suffix -ous. The names of Fe3+, Fe2+, Sn4+, and Sn2+ are therefore ferric, ferrous, stannic, and stannous, respectively. Even though this text uses the systematic names with roman numerals, it is important to recognize these common names because they are still often used. For example, on the label of dental fluoride rinse, the compound chemists call tin(II) fluoride is usually listed as stannous fluoride.
Some examples of metals that form more than one cation are listed in Table \(\PageIndex{1}\), along with the names of the ions. Note that the simple Hg+ cation does not occur in chemical compounds. Instead, all compounds of mercury(I) contain a dimeric cation, Hg22+, in which the two Hg atoms are bonded together.
| Cation | Systematic Name | Common Name | Cation | Systematic Name | Common Name |
|---|---|---|---|---|---|
| * Not widely used. | |||||
| †The isolated mercury(I) ion exists only as the gaseous ion. | |||||
| Co2+ | cobalt(II) | cobaltous* | Pb4+ | lead(IV) | plumbic* |
| Co3+ | cobalt(III) | cobaltic* | Pb2+ | lead(II) | plumbous* |
| Cr2+ | chromium(II) | chromous | Cu2+ | copper(II) | cupric |
| Cr3+ | chromium(III) | chromic | Cu+ | copper(I) | cuprous |
| Fe2+ | iron(II) | ferrous | Sn4+ | tin(IV) | stannic |
| Fe3+ | iron(III) | ferric | Sn2+ | tin(II) | stannous |
| Mn2+ | manganese(II) | manganous* | Hg2+ | mercury(II) | mercuric |
| Mn3+ | manganese(III) | manganic* | Hg22+ | mercury(I) | mercurous† |
c. Polyatomic cations. The names of the common polyatomic cations that are relatively important in ionic compounds (such as, the ammonium ion) are in Table 2.4
3. Name the anion.
a. Monatomic anions. Monatomic anions are named by adding the suffix -ide to the root of the name of the parent element; thus, Cl− is chloride, O2− is oxide, P3− is phosphide, N3− is nitride (also called azide), and C4− is carbide. Because the charges on these ions can be predicted from their position in the periodic table, it is not necessary to specify the charge in the name. Examples of monatomic anions are in Table 2.2
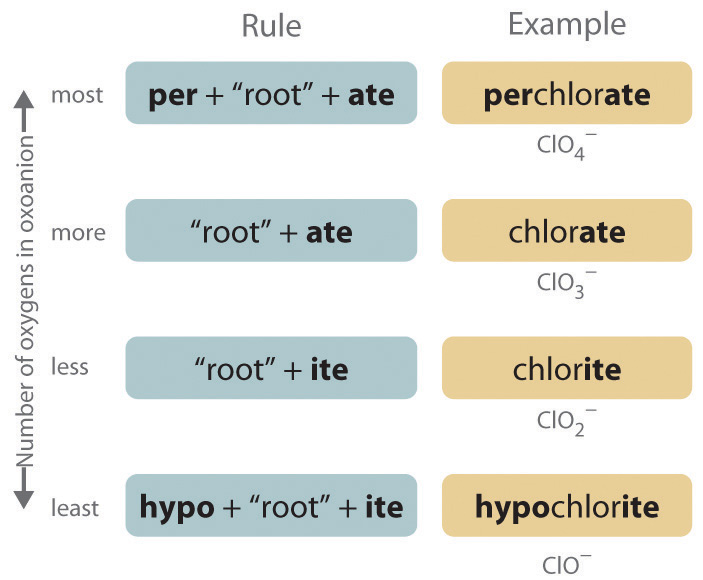
b. Polyatomic anions. Polyatomic anions typically have common names that must be memorized; some examples are in Table 2.4. Polyatomic anions that contain a single metal or nonmetal atom plus one or more oxygen atoms are called oxoanions (or oxyanions). In cases where only two oxoanions are known for an element, the name of the oxoanion with more oxygen atoms ends in -ate, and the name of the oxoanion with fewer oxygen atoms ends in -ite. For example, NO3− is nitrate and NO2− is nitrite.
The halogens and some of the transition metals form more extensive series of oxoanions with as many as four members. In the names of these oxoanions, the prefix per- is used to identify the oxoanion with the most oxygen (so that ClO4− is perchlorate and ClO3− is chlorate), and the prefix hypo- is used to identify the anion with the fewest oxygen (ClO2− is chlorite and ClO− is hypochlorite). The relationship between the names of oxoanions and the number of oxygen atoms present is diagrammed in Figure \(\PageIndex{3}\) "The Relationship between the Names of Oxoanions and the Number of Oxygen Atoms Present." Differentiating the oxoanions in such a series is no trivial matter; for example, the hypochlorite ion is the active ingredient in laundry bleach and swimming pool disinfectant, but compounds that contain the perchlorate ion can explode if they come into contact with organic substances.
4. Write the name of the compound as the name of the cation followed by the name of the anion.

It is not necessary to indicate the number of cations or anions present per formula unit in the name of an ionic compound because this information is implied by the charges on the ions. The charge of the ions must be considered when writing the formula for an ionic compound from its name, however. Because the charge on the chloride ion is −1 and the charge on the calcium ion is +2, for example, consistent with their positions in the periodic table, arithmetic indicates that calcium chloride must contain twice as many chloride ions as calcium ions to maintain electrical neutrality. Thus, the formula is CaCl2. Similarly, calcium phosphate must be Ca3(PO4)2 because the cation and the anion have charges of +2 and −3, respectively. The best way to learn how to name ionic compounds is to work through a few examples, referring to Figure \(\PageIndex{1}\), Table 2.2, Table 2.4, and Table \(\PageIndex{1}\) as needed.
With only a few exceptions, these metals are usually transition metals or actinides.
Cations are always named before anions.
Most transition metals, many actinides, and the heaviest elements of groups 13–15 can form more than one cation.
Write the systematic name (and the common name if applicable) for each ionic compound.
- LiCl
- MgSO4
- (NH4)3PO4
- Cu2O
Given: empirical formula
Asked for: name
Strategy:
A If only one charge is possible for the cation, give its name, consulting Table 2.2 or Table 2.4 if necessary. If the cation can have more than one charge (Table \(\PageIndex{1}\)), specify the charge using roman numerals.
B If the anion does not contain oxygen, name it according to step 3a, using Table 2.2 and Table 2.4 if necessary. For polyatomic anions that contain oxygen, use Table 2.4 and the appropriate prefix and suffix listed in step 3b.
C Beginning with the cation, write the name of the compound.
Solution:
a. A B Lithium is in group 1, so we know that it forms only the Li+ cation, which is the lithium ion. Similarly, chlorine is in group 7, so it forms the Cl− anion, which is the chloride ion. C Because we begin with the name of the cation, the name of this compound is lithium chloride, which is used medically as an antidepressant drug.
b. A B The cation is the magnesium ion, and the anion, which contains oxygen, is sulfate. C Because we list the cation first, the name of this compound is magnesium sulfate. A hydrated form of magnesium sulfate (MgSO4·7H2O) is sold in drugstores as Epsom salts, a harsh but effective laxative.
c. A B The cation is the ammonium ion (from Table 2.4), and the anion is phosphate. C The compound is therefore ammonium phosphate, which is widely used as a fertilizer. It is not necessary to specify that the formula unit contains three ammonium ions because three are required to balance the negative charge on phosphate.
d. A B The cation is a transition metal that often forms more than one cation (Table \(\PageIndex{1}\)). We must therefore specify the positive charge on the cation in the name: copper(I) or, according to the older system, cuprous. The anion is oxide. C The name of this compound is copper(I) oxide or, in the older system, cuprous oxide. Copper(I) oxide is used as a red glaze on ceramics and in antifouling paints to prevent organisms from growing on the bottoms of boats.
Cu2O. The bottom of a boat is protected with a red antifouling paint containing copper(I) oxide, Cu2O.
Exercise
Write the systematic name (and the common name if applicable) for each ionic compound.
- CuCl2
- MgCO3
- FePO4
Answer:
- copper(II) chloride (or cupric chloride)
- magnesium carbonate
- iron(III) phosphate (or ferric phosphate)
Write the formula for each compound.
- calcium dihydrogen phosphate
- aluminum sulfate
- chromium(III) oxide
Given: systematic name
Asked for: formula
Strategy:
A Identify the cation and its charge using the location of the element in the periodic table and Table 2.2 and form cations with different charges, use the appropriate roman numeral or suffix to indicate its charge.
B Identify the anion using Table 2.2 and Table 2.4. Beginning with the cation, write the compound’s formula and then determine the number of cations and anions needed to achieve electrical neutrality.
Solution:
a. A Calcium is in group 2, so it forms only the Ca2+ ion. B Dihydrogen phosphate is the H2PO4− ion (Table 2.4). Two H2PO4− ions are needed to balance the positive charge on Ca2+, to give Ca(H2PO4)2. A hydrate of calcium dihydrogen phosphate, Ca(H2PO4)2·H2O, is the active ingredient in baking powder.
b. A Aluminum, near the top of group 13 in the periodic table, forms only one cation, Al3+ (Figure \(\PageIndex{2}\) "Metals That Form More Than One Cation and Their Locations in the Periodic Table"). B Sulfate is SO42− (Table 2.4). To balance the electrical charges, we need two Al3+ cations and three SO42− anions, giving Al2(SO4)3. Aluminum sulfate is used to tan leather and purify drinking water.
c. A Because chromium is a transition metal, it can form cations with different charges. The roman numeral tells us that the positive charge in this case is +3, so the cation is Cr3+. B Oxide is O2−. Thus two cations (Cr3+) and three anions (O2−) are required to give an electrically neutral compound, Cr2O3. This compound is a common green pigment that has many uses, including camouflage coatings.
Cr2O3. Chromium(III) oxide (Cr2O3) is a common pigment in dark green paints, such as camouflage paint.
Write the formula for each compound.
- barium chloride
- sodium carbonate
- iron(III) hydroxide
Answer:
- BaCl2
- Na2CO3
- Fe(OH)3
Summary
- There is a systematic method used to name ionic compounds.
Ionic compounds are named according to systematic procedures, although common names are widely used. Systematic nomenclature enables chemists to write the structure of any compound from its name and vice versa. Ionic compounds are named by writing the cation first, followed by the anion. If a metal can form cations with more than one charge, the charge is indicated by roman numerals in parentheses following the name of the metal. Oxoanions are polyatomic anions that contain a single metal or nonmetal atom and one or more oxygen atoms.
- To describe the composition of a chemical compound.
- To name covalent compounds that contain up to three elements.
As with ionic compounds, the system for naming covalent compounds enables chemists to write the molecular formula from the name and vice versa. This and the following section describe the rules for naming simple covalent compounds, beginning with inorganic compounds and then turning to simple organic compounds that contain only carbon and hydrogen.
When chemists synthesize a new compound, they may not yet know its molecular or structural formula. In such cases, they usually begin by determining its empirical formula, the relative numbers of atoms of the elements in a compound, reduced to the smallest whole numbers. Because the empirical formula is based on experimental measurements of the numbers of atoms in a sample of the compound, it shows only the ratios of the numbers of the elements present. The difference between empirical and molecular formulas can be illustrated with butane, a covalent compound used as the fuel in disposable lighters. The molecular formula for butane is C4H10. The ratio of carbon atoms to hydrogen atoms in butane is 4:10, which can be reduced to 2:5. The empirical formula for butane is therefore C2H5. The formula unit is the absolute grouping of atoms or ions represented by the empirical formula of a compound, either ionic or covalent. Butane has the empirical formula C2H5, but it contains two C2H5 formula units, giving a molecular formula of C4H10.
Because ionic compounds do not contain discrete molecules, empirical formulas are used to indicate their compositions. All compounds, whether ionic or covalent, must be electrically neutral. Consequently, the positive and negative charges in a formula unit must exactly cancel each other. If the cation and the anion have charges of equal magnitude, such as Na+ and Cl−, then the compound must have a 1:1 ratio of cations to anions, and the empirical formula must be NaCl. If the charges are not the same magnitude, then a cation:anion ratio other than 1:1 is needed to produce a neutral compound. In the case of Mg2+ and Cl−, for example, two Cl− ions are needed to balance the two positive charges on each Mg2+ ion, giving an empirical formula of MgCl2. Similarly, the formula for the ionic compound that contains Na+ and O2− ions is Na2O.
Ionic compounds do not contain discrete molecules, so empirical formulas are used to indicate their compositions.
Nomenclature of Metals: https://youtu.be/zVhGxYTgRk0
Nomenclature of Transition Metals: https://youtu.be/gIaRpko0A_A
Binary Ionic Compounds
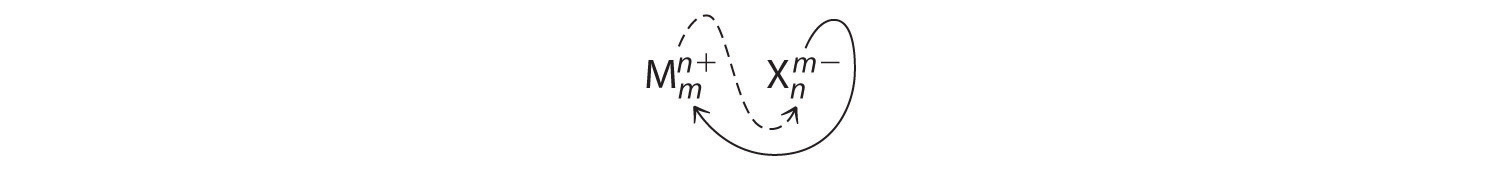
An ionic compound that contains only two elements, one present as a cation and one as an anion, is called a binary ionic compound. One example is MgCl2, a coagulant used in the preparation of tofu from soybeans. For binary ionic compounds, the subscripts in the empirical formula can also be obtained by crossing charges: use the absolute value of the charge on one ion as the subscript for the other ion. This method is shown schematically as follows:
Crossing charges. One method for obtaining subscripts in the empirical formula is by crossing charges.
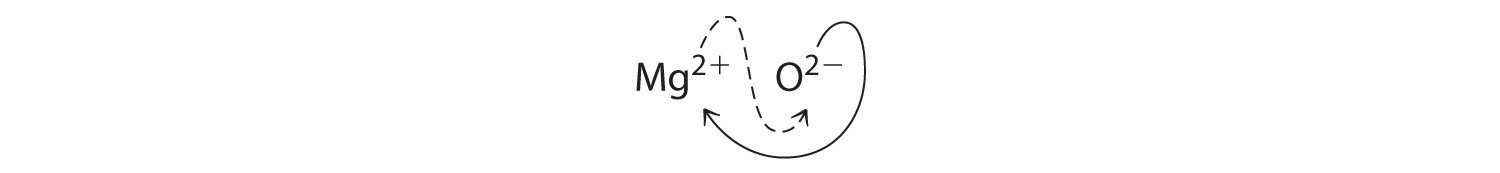
When crossing charges, it is sometimes necessary to reduce the subscripts to their simplest ratio to write the empirical formula. Consider, for example, the compound formed by Mg2+ and O2−. Using the absolute values of the charges on the ions as subscripts gives the formula Mg2O2:
This simplifies to its correct empirical formula MgO. The empirical formula has one Mg2+ ion and one O2− ion.
Write the empirical formula for the simplest binary ionic compound formed from each ion or element pair.
- Ga3+ and As3−
- Eu3+ and O2−
- calcium and chlorine
Given: ions or elements
Asked for: empirical formula for binary ionic compound
Strategy:
- If not given, determine the ionic charges based on the location of the elements in the periodic table.
- Use the absolute value of the charge on each ion as the subscript for the other ion. Reduce the subscripts to the lowest numbers
to write the empirical formula. Check to make sure the empirical formula is electrically neutral.
Solution
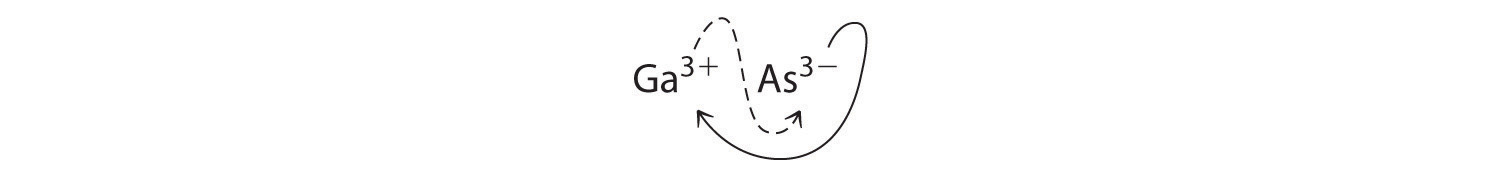
a. B Using the absolute values of the charges on the ions as the subscripts gives Ga3As3:
Reducing the subscripts to the smallest whole numbers gives the empirical formula GaAs, which is electrically neutral [+3 + (−3) = 0]. Alternatively, we could recognize that Ga3+ and As3− have charges of equal magnitude but opposite signs. One Ga3+ ion balances the charge on one As3− ion, and a 1:1 compound will have no net charge. Because we write subscripts only if the number is greater than 1, the empirical formula is GaAs. GaAs is gallium arsenide, which is widely used in the electronics industry in transistors and other devices.
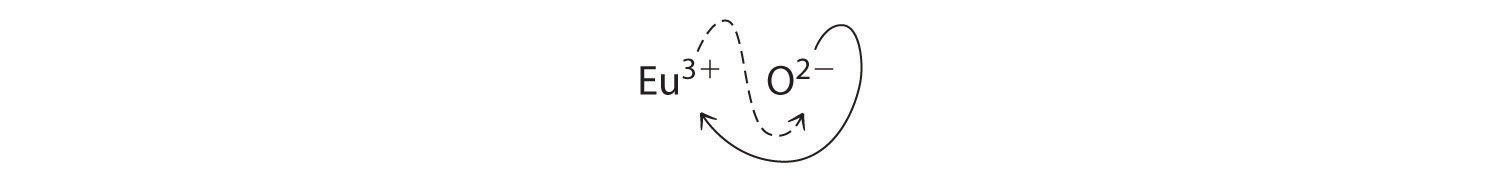
b. B Because Eu3+ has a charge of +3 and O2− has a charge of −2, a 1:1 compound would have a net charge of +1. We must therefore find multiples of the charges that cancel. We cross charges, using the absolute value of the charge on one ion as the subscript for the other ion:
The subscript for Eu3+ is 2 (from O2−), and the subscript for O2− is 3 (from Eu3+), giving Eu2O3; the subscripts cannot be reduced further. The empirical formula contains a positive charge of 2(+3) = +6 and a negative charge of 3(−2) = −6, for a net charge of 0. The compound Eu2O3 is neutral. Europium oxide is responsible for the red color in television and computer screens.
c. A Because the charges on the ions are not given, we must first determine the charges expected for the most common ions derived from calcium and chlorine. Calcium lies in group 2, so it should lose two electrons to form Ca2+. Chlorine lies in group 17, so it should gain one electron to form Cl−.
B Two Cl− ions are needed to balance the charge on one Ca2+ ion, which leads to the empirical formula CaCl2. We could also cross charges, using the absolute value of the charge on Ca2+ as the subscript for Cl and the absolute value of the charge on Cl− as the subscript for Ca:
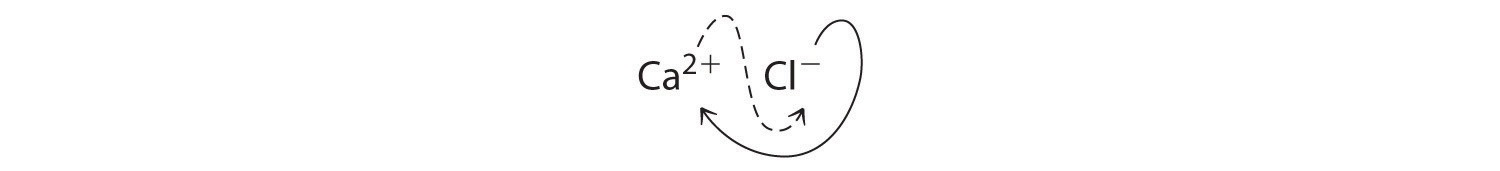
The subscripts in CaCl2 cannot be reduced further. The empirical formula is electrically neutral [+2 + 2(−1) = 0]. This compound is calcium chloride, one of the substances used as “salt” to melt ice on roads and sidewalks in winter.
Write the empirical formula for the simplest binary ionic compound formed from each ion or element pair.
- Li+ and N3−
- Al3+ and O2−
- lithium and oxygen
Answer:
- Li3N
- Al2O3
- Li2O
Polyatomic Ions
Polyatomic ions are groups of atoms that bear net electrical charges, although the atoms in a polyatomic ion are held together by the same covalent bonds that hold atoms together in molecules. Just as there are many more kinds of molecules than simple elements, there are many more kinds of polyatomic ions than monatomic ions. Two examples of polyatomic cations are the ammonium (NH4+) and the methylammonium (CH3NH3+) ions. Polyatomic anions are much more numerous than polyatomic cations; some common examples are in Table \(\PageIndex{1}\).
| Formula | Name of Ion | Formula | Name of Ion |
|---|---|---|---|
| NH4+ | ammonium | HPO42− | hydrogen phosphate |
| CH3NH3+ | methylammonium | H2PO4− | dihydrogen phosphate |
| OH− | hydroxide | ClO− | hypochlorite |
| O22− | peroxide | ClO2− | chlorite |
| CN− | cyanide | ClO3− | chlorate |
| SCN− | thiocyanate | ClO4− | perchlorate |
| NO2− | nitrite | MnO4− | permanganate |
| NO3− | nitrate | CrO42− | chromate |
| CO32− | carbonate | Cr2O72− | dichromate |
| HCO3− | hydrogen carbonate, or bicarbonate | C2O42− | oxalate |
| SO32− | sulfite | HCO2− | formate |
| SO42− | sulfate | CH3CO2− | acetate |
| HSO4− | hydrogen sulfate, or bisulfate | C6H5CO2− | benzoate |
| PO43− | phosphate |
The method used to predict the empirical formulas for ionic compounds that contain monatomic ions can also be used for compounds that contain polyatomic ions. The overall charge on the cations must balance the overall charge on the anions in the formula unit. Thus, K+ and NO3− ions combine in a 1:1 ratio to form KNO3 (potassium nitrate or saltpeter), a major ingredient in black gunpowder. Similarly, Ca2+ and SO42− form CaSO4 (calcium sulfate), which combines with varying amounts of water to form gypsum and plaster of Paris. The polyatomic ions NH4+ and NO3− form NH4NO3 (ammonium nitrate), a widely used fertilizer and, in the wrong hands, an explosive. One example of a compound in which the ions have charges of different magnitudes is calcium phosphate, which is composed of Ca2+ and PO43− ions; it is a major component of bones. The compound is electrically neutral because the ions combine in a ratio of three Ca2+ ions [3(+2) = +6] for every two ions [2(−3) = −6], giving an empirical formula of Ca3(PO4)2; the parentheses around PO4 in the empirical formula indicate that it is a polyatomic ion. Writing the formula for calcium phosphate as Ca3P2O8 gives the correct number of each atom in the formula unit, but it obscures the fact that the compound contains readily identifiable PO43− ions.
Write the empirical formula for the compound formed from each ion pair.
- Na+ and HPO42−
- potassium cation and cyanide anion
- calcium cation and hypochlorite anion
Given: ions
Asked for: empirical formula for ionic compound
Strategy:
- If it is not given, determine the charge on a monatomic ion from its location in the periodic table. Use Table \(\PageIndex{1}\) to find the charge on a polyatomic ion.
- Use the absolute value of the charge on each ion as the subscript for the other ion. Reduce the subscripts to the smallest whole numbers when writing the empirical formula.
Solution:
- B Because HPO42− has a charge of −2 and Na+ has a charge of +1, the empirical formula requires two Na+ ions to balance the charge of the polyatomic ion, giving Na2HPO4. The subscripts are reduced to the lowest numbers, so the empirical formula is Na2HPO4. This compound is sodium hydrogen phosphate, which is used to provide texture in processed cheese, puddings, and instant breakfasts.
- A The potassium cation is K+, and the cyanide anion is CN−. B Because the magnitude of the charge on each ion is the same, the empirical formula is KCN. Potassium cyanide is highly toxic, and at one time it was used as rat poison. This use has been discontinued, however, because too many people were being poisoned accidentally.
- A The calcium cation is Ca2+, and the hypochlorite anion is ClO−. B Two ClO− ions are needed to balance the charge on one Ca2+ ion, giving Ca(ClO)2. The subscripts cannot be reduced further, so the empirical formula is Ca(ClO)2. This is calcium hypochlorite, the “chlorine” used to purify water in swimming pools.
Write the empirical formula for the compound formed from each ion pair.
- Ca2+ and H2PO4−
- sodium cation and bicarbonate anion
- ammonium cation and sulfate anion
Answer:
- Ca(H2PO4)2: calcium dihydrogen phosphate is one of the ingredients in baking powder.
- NaHCO3: sodium bicarbonate is found in antacids and baking powder; in pure form, it is sold as baking soda.
- (NH4)2SO4: ammonium sulfate is a common source of nitrogen in fertilizers.
Polyatomics: https://youtu.be/kTSPkzDcntA
Hydrates
Many ionic compounds occur as hydrates, compounds that contain specific ratios of loosely bound water molecules, called waters of hydration. Waters of hydration can often be removed simply by heating. For example, calcium dihydrogen phosphate can form a solid that contains one molecule of water per Ca(H2PO4)2 unit and is used as a leavening agent in the food industry to cause baked goods to rise. The empirical formula for the solid is Ca(H2PO4)2·H2O. In contrast, copper sulfate usually forms a blue solid that contains five waters of hydration per formula unit, with the empirical formula CuSO4·5H2O. When heated, all five water molecules are lost, giving a white solid with the empirical formula CuSO4.


Compounds that differ only in the numbers of waters of hydration can have very different properties. For example, CaSO4·½H2O is plaster of Paris, which was often used to make sturdy casts for broken arms or legs, whereas CaSO4·2H2O is the less dense, flakier gypsum, a mineral used in drywall panels for home construction. When a cast would set, a mixture of plaster of Paris and water crystallized to give solid CaSO4·2H2O. Similar processes are used in the setting of cement and concrete.
Binary Inorganic Compounds
Binary covalent compounds—covalent compounds that contain only two elements—are named using a procedure similar to that used for simple ionic compounds, but prefixes are added as needed to indicate the number of atoms of each kind. The procedure, diagrammed in Figure \(\PageIndex{2}\) consists of the following steps:
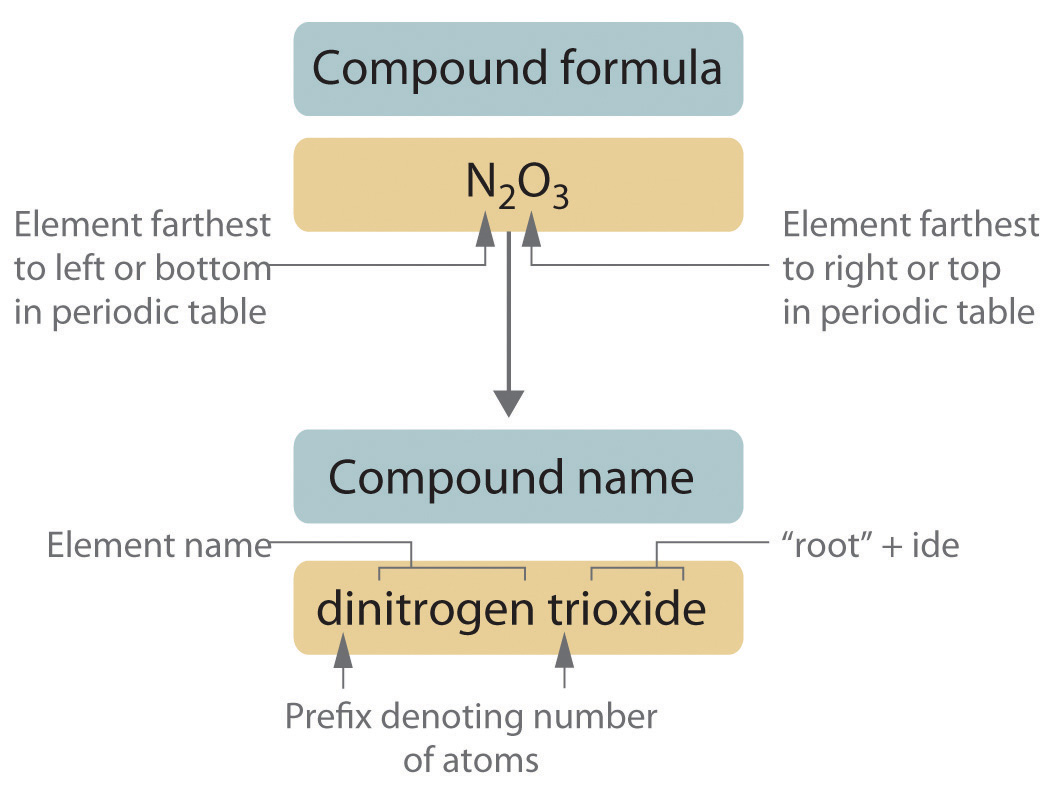
- Place the elements in their proper order.
- The element farthest to the left in the periodic table is usually named first. If both elements are in the same group, the element closer to the bottom of the column is named first.
- The second element is named as if it were a monatomic anion in an ionic compound (even though it is not), with the suffix -ide attached to the root of the element name.
- Identify the number of each type of atom present.
- Prefixes derived from Greek stems are used to indicate the number of each type of atom in the formula unit (Table \(\PageIndex{2}\)). The prefix mono- (“one”) is used only when absolutely necessary to avoid confusion, just as the subscript 1 is omitted when writing molecular formulas.
To demonstrate steps 1 and 2a, HCl is named hydrogen chloride (because hydrogen is to the left of chlorine in the periodic table), and PCl5 is phosphorus pentachloride. The order of the elements in the name of BrF3, bromine trifluoride, is determined by the fact that bromine lies below fluorine in Group 17.
Table \(\PageIndex{2}\): Prefixes for Indicating the Number of Atoms in Chemical Names Prefix Number mono- 1 di- 2 tri- 3 tetra- 4 penta- 5 hexa- 6 hepta- 7 octa- 8 nona- 9 deca- 10 undeca- 11 dodeca- 12 - If a molecule contains more than one atom of both elements, then prefixes are used for both. Thus N2O3 is dinitrogen trioxide, as shown in Figure 2.13.
- In some names, the final a or o of the prefix is dropped to avoid awkward pronunciation. Thus OsO4 is osmium tetroxide rather than osmium tetraoxide.
- Prefixes derived from Greek stems are used to indicate the number of each type of atom in the formula unit (Table \(\PageIndex{2}\)). The prefix mono- (“one”) is used only when absolutely necessary to avoid confusion, just as the subscript 1 is omitted when writing molecular formulas.
- Write the name of the compound.
- Binary compounds of the elements with oxygen are generally named as “element oxide,” with prefixes that indicate the number of atoms of each element per formula unit. For example, CO is carbon monoxide. The only exception is binary compounds of oxygen with fluorine, which are named as oxygen fluorides.
- Certain compounds are always called by the common names that were assigned before formulas were used. For example, H2O is water (not dihydrogen oxide); NH3 is ammonia; PH3 is phosphine; SiH4 is silane; and B2H6, a dimer of BH3, is diborane. For many compounds, the systematic name and the common name are both used frequently, requiring familiarity with both. For example, the systematic name for NO is nitrogen monoxide, but it is much more commonly called nitric oxide. Similarly, N2O is usually called nitrous oxide rather than dinitrogen monoxide. Notice that the suffixes -ic and -ous are the same ones used for ionic compounds.
Start with the element at the far left in the periodic table and work to the right. If two or more elements are in the same group, start with the bottom element and work up.
Write the name of each binary covalent compound.
- SF6
- N2O4
- ClO2
Given: molecular formula
Asked for: name of compound
Strategy:
- List the elements in order according to their positions in the periodic table. Identify the number of each type of atom in the chemical formula and then use Table \(\PageIndex{2}\) to determine the prefixes needed.
- If the compound contains oxygen, follow step 3a. If not, decide whether to use the common name or the systematic name.
Solution:
- A Because sulfur is to the left of fluorine in the periodic table, sulfur is named first. Because there is only one sulfur atom in the formula, no prefix is needed. B There are, however, six fluorine atoms, so we use the prefix for six: hexa- (Table \(\PageIndex{2}\)). The compound is sulfur hexafluoride.
- A Because nitrogen is to the left of oxygen in the periodic table, nitrogen is named first. Because more than one atom of each element is present, prefixes are needed to indicate the number of atoms of each. According to Table \(\PageIndex{2}\) "Prefixes for Indicating the Number of Atoms in Chemical Names", the prefix for two is di-, and the prefix for four is tetra-. B The compound is dinitrogen tetroxide (omitting the a in tetra- according to step 2c) and is used as a component of some rocket fuels.
- A Although oxygen lies to the left of chlorine in the periodic table, it is not named first because ClO2 is an oxide of an element other than fluorine (step 3a). Consequently, chlorine is named first, but a prefix is not necessary because each molecule has only one atom of chlorine. B Because there are two oxygen atoms, the compound is a dioxide. Thus the compound is chlorine dioxide. It is widely used as a substitute for chlorine in municipal water treatment plants because, unlike chlorine, it does not react with organic compounds in water to produce potentially toxic chlorinated compounds.
Write the name of each binary covalent compound.
- IF7
- N2O5
- OF2
Answer
- iodine heptafluoride
- dinitrogen pentoxide
- oxygen difluoride
Write the formula for each binary covalent compound.
- sulfur trioxide
- diiodine pentoxide
Given: name of compound
Asked for: formula
Strategy:
List the elements in the same order as in the formula, use Table \(\PageIndex{2}\) to identify the number of each type of atom present, and then indicate this quantity as a subscript to the right of that element when writing the formula.
Solution:
- Sulfur has no prefix, which means that each molecule has only one sulfur atom. The prefix tri- indicates that there are three oxygen atoms. The formula is therefore SO3. Sulfur trioxide is produced industrially in huge amounts as an intermediate in the synthesis of sulfuric acid.
- The prefix di- tells you that each molecule has two iodine atoms, and the prefix penta- indicates that there are five oxygen atoms. The formula is thus I2O5, a compound used to remove carbon monoxide from air in respirators.
Write the formula for each binary covalent compound.
- silicon tetrachloride
- disulfur decafluoride
Answer
- SiCl4
- S2F10
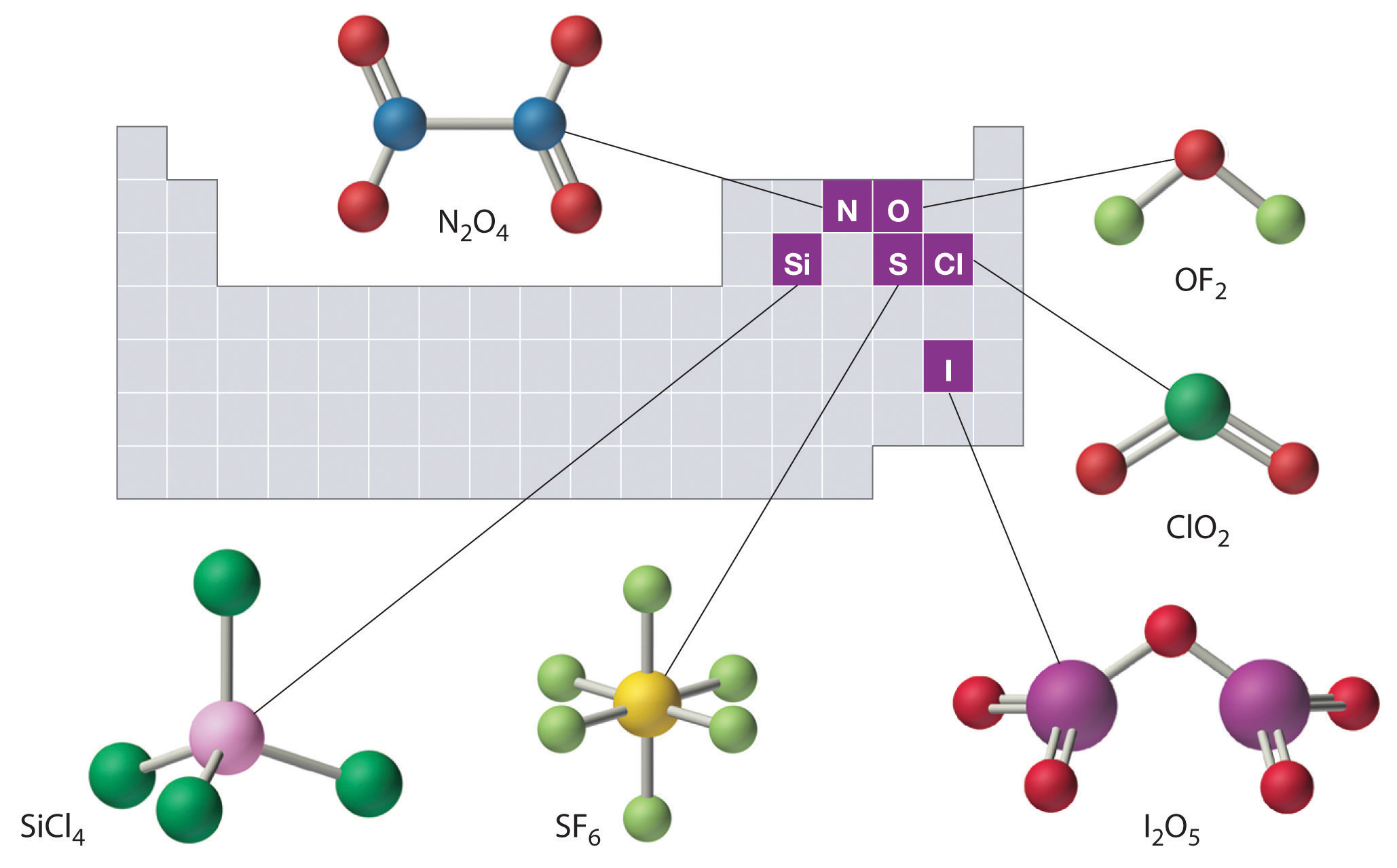
The structures of some of the compounds in Examples 3.6.3 and 3.6.4 are shown in Figure \(\PageIndex{2}\) along with the location of the “central atom” of each compound in the periodic table. It may seem that the compositions and structures of such compounds are entirely random, but this is not true. After mastering the material discussed later on this course, one is able to predict the compositions and structures of compounds of this type with a high degree of accuracy.
Nomenclature of Nonmetals: https://youtu.be/VgHCrtpDWJk
The Learning Objective of this Module is to identify and name some common acids and bases.
For our purposes at this point in the text, we can define an acid as a substance with at least one hydrogen atom that can dissociate to form an anion and an H+ ion (a proton) in aqueous solution, thereby forming an acidic solution. We can define bases as compounds that produce hydroxide ions (OH−) and a cation when dissolved in water, thus forming a basic solution. Solutions that are neither basic nor acidic are neutral. We will discuss the chemistry of acids and bases in more detail later, but in this section we describe the nomenclature of common acids and identify some important bases so that you can recognize them in future discussions. Pure acids and bases and their concentrated aqueous solutions are commonly encountered in the laboratory. They are usually highly corrosive, so they must be handled with care.
Acids
The names of acids differentiate between (1) acids in which the H+ ion is attached to an oxygen atom of a polyatomic anion (these are called oxoacids, or occasionally oxyacids) and (2) acids in which the H+ ion is attached to some other element. In the latter case, the name of the acid begins with hydro- and ends in -ic, with the root of the name of the other element or ion in between. Recall that the name of the anion derived from this kind of acid always ends in -ide. Thus hydrogen chloride (HCl) gas dissolves in water to form hydrochloric acid (which contains H+ and Cl− ions), hydrogen cyanide (HCN) gas forms hydrocyanic acid (which contains H+ and CN− ions), and so forth (Table \(\PageIndex{3}\)). Examples of this kind of acid are commonly encountered and very important. For instance, your stomach contains a dilute solution of hydrochloric acid to help digest food. When the mechanisms that prevent the stomach from digesting itself malfunction, the acid destroys the lining of the stomach and an ulcer forms.
Acids are distinguished by whether the H+ ion is attached to an oxygen atom of a polyatomic anion or some other element.
| Formula | Name in Aqueous Solution | Name of Gaseous Species |
|---|---|---|
| HF | hydrofluoric acid | hydrogen fluoride |
| HCl | hydrochloric acid | hydrogen chloride |
| HBr | hydrobromic acid | hydrogen bromide |
| HI | hydroiodic acid | hydrogen iodide |
| HCN | hydrocyanic acid | hydrogen cyanide |
| H2S | hydrosulfuric acid | hydrogen sulfide |
If an acid contains one or more H+ ions attached to oxygen, it is a derivative of one of the common oxoanions, such as sulfate (SO42−) or nitrate (NO3−). These acids contain as many H+ ions as are necessary to balance the negative charge on the anion, resulting in a neutral species such as H2SO4 and HNO3.
The names of acids are derived from the names of anions according to the following rules:
- If the name of the anion ends in -ate, then the name of the acid ends in -ic. For example, because NO3− is the nitrate ion, HNO3 is nitric acid. Similarly, ClO4− is the perchlorate ion, so HClO4 is perchloric acid. Two important acids are sulfuric acid (H2SO4) from the sulfate ion (SO42−) and phosphoric acid (H3PO4) from the phosphate ion (PO43−). These two names use a slight variant of the root of the anion name: sulfate becomes sulfuric and phosphate becomes phosphoric.
- If the name of the anion ends in -ite, then the name of the acid ends in -ous. For example, OCl− is the hypochlorite ion, and HOCl is hypochlorous acid; NO2− is the nitrite ion, and HNO2 is nitrous acid; and SO32− is the sulfite ion, and H2SO3 is sulfurous acid. The same roots are used whether the acid name ends in -ic or -ous; thus, sulfite becomes sulfurous.
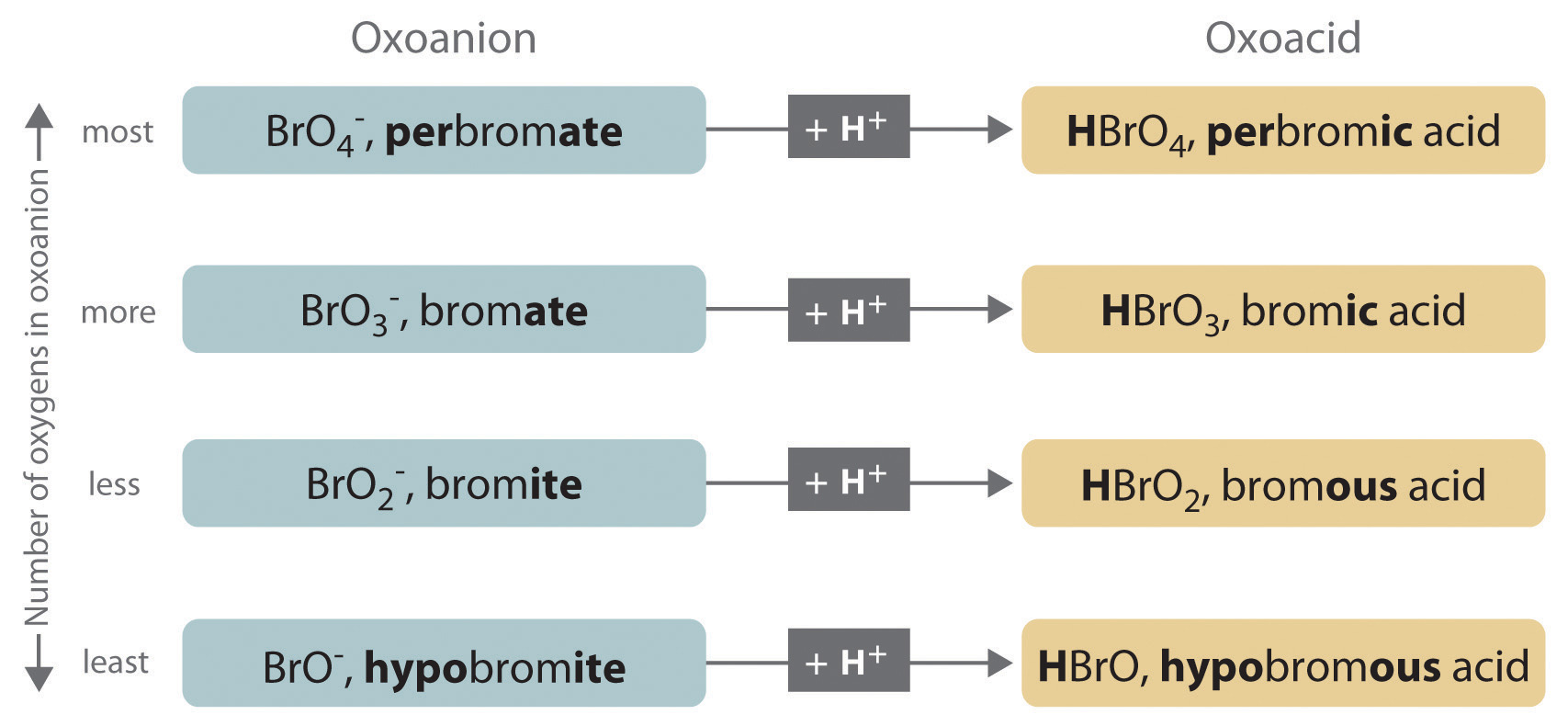
The relationship between the names of the oxoacids and the parent oxoanions is illustrated in Figure \(\PageIndex{3}\), and some common oxoacids are in Table \(\PageIndex{4}\).
| Formula | Name |
|---|---|
| \(HNO_2\) | nitrous acid |
| \(HNO_3\) | nitric acid |
| \(H_2SO_3\) | sulfurous acid |
| \(H_2SO_4\) | sulfuric acid |
| \(H_3PO_4\) | phosphoric acid |
| \(H_2CO_3\) | carbonic acid |
| \(HClO\) | hypochlorous acid |
| \(HClO_2\) | chlorous acid |
| \(HClO_3\) | chloric acid |
| \(HClO_4\) | perchloric acid |
Name and give the formula for each acid.
- the acid formed by adding a proton to the hypobromite ion (OBr−)
- the acid formed by adding two protons to the selenate ion (SeO42−)
Given: anion
Asked for: parent acid
Strategy:
Refer to Table \(\PageIndex{3}\) and Table \(\PageIndex{4}\) to find the name of the acid. If the acid is not listed, use the guidelines given previously.
Solution:
Neither species is listed in Table \(\PageIndex{3}\) or Table \(\PageIndex{4}\), so we must use the information given previously to derive the name of the acid from the name of the polyatomic anion.
- The anion name, hypobromite, ends in -ite, so the name of the parent acid ends in -ous. The acid is therefore hypobromous acid (HOBr).
- Selenate ends in -ate, so the name of the parent acid ends in -ic. The acid is therefore selenic acid (H2SeO4).
Name and give the formula for each acid.
- the acid formed by adding a proton to the perbromate ion (BrO4−)
- the acid formed by adding three protons to the arsenite ion (AsO33−)
Answer:
- perbromic acid; HBrO4
- arsenous acid; H3AsO3
Nomenclature of Acids: https://youtu.be/in46UUzmSO4
Organic Acids
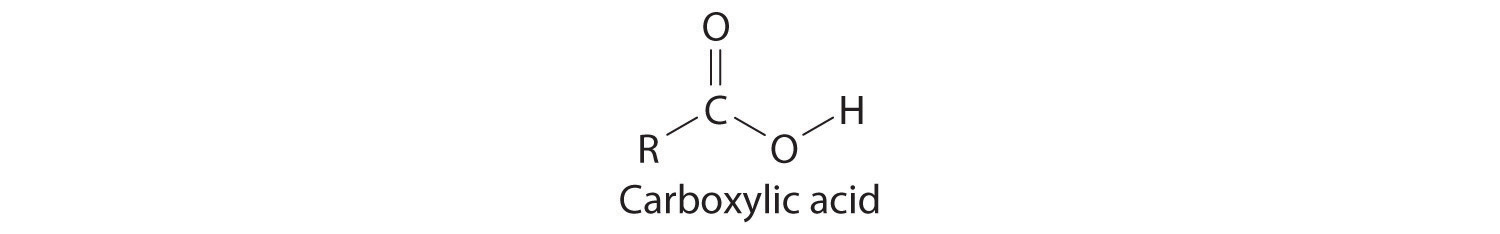
Many organic compounds contain the carbonyl group, in which there is a carbon–oxygen double bond. In carboxylic acids, an –OH group is covalently bonded to the carbon atom of the carbonyl group. Their general formula is RCO2H, sometimes written as RCOOH:
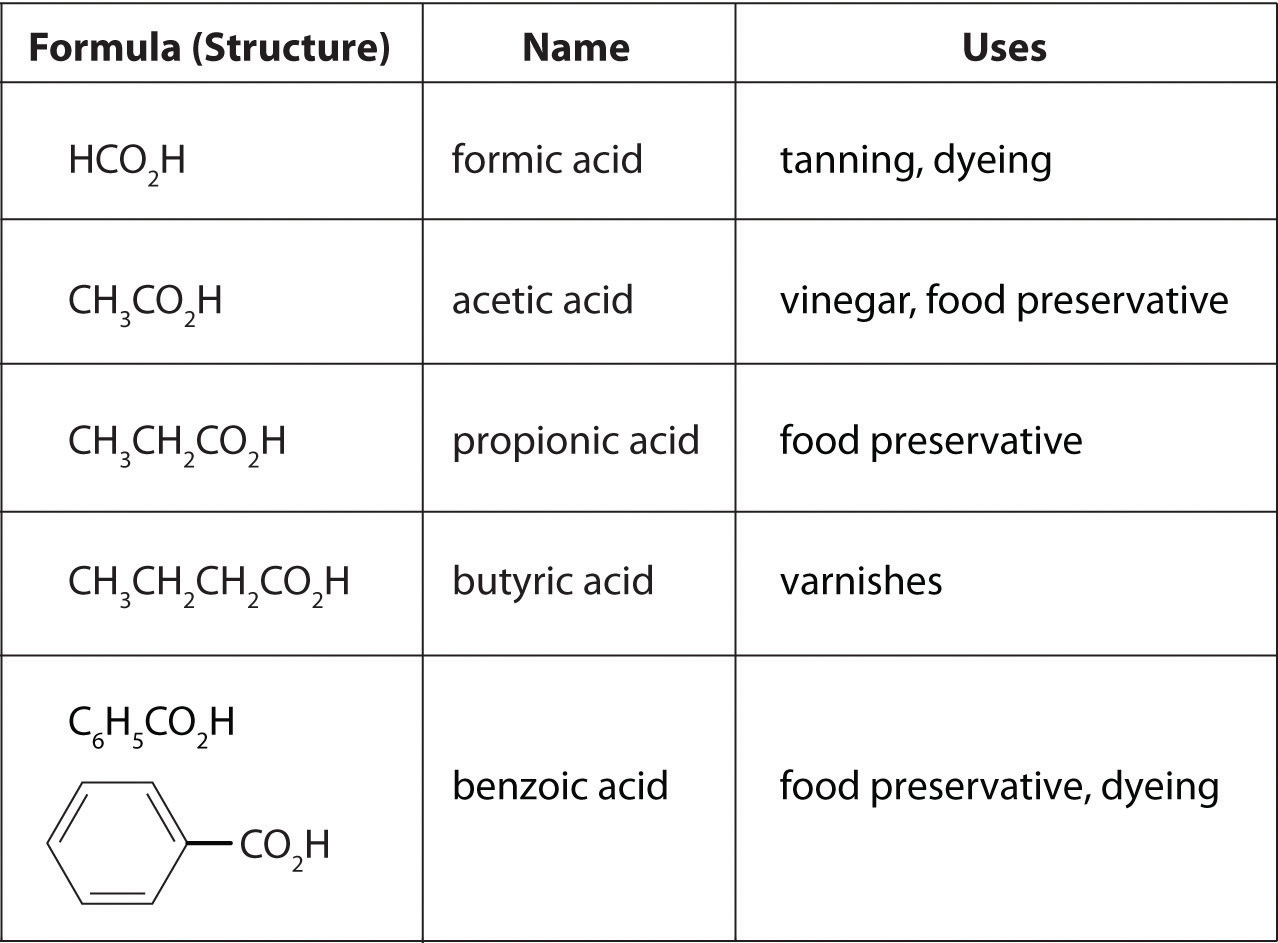
where R can be an alkyl group, an aryl group, or a hydrogen atom. The simplest example, HCO2H, is formic acid, so called because it is found in the secretions of stinging ants (from the Latin formica, meaning “ant”). Another example is acetic acid (CH3CO2H), which is found in vinegar. Like many acids, carboxylic acids tend to have sharp odors. For example, butyric acid (CH3CH2CH2CO2H), is responsible for the smell of rancid butter, and the characteristic odor of sour milk and vomit is due to lactic acid [CH3CH(OH)CO2H]. Some common carboxylic acids are shown in Table 3.6.5.
Table \(\PageIndex{5}\): Some Common Carboxylic Acids
Although carboxylic acids are covalent compounds, when they dissolve in water, they dissociate to produce H+ ions (just like any other acid) and RCO2− ions. Note that only the hydrogen attached to the oxygen atom of the CO2 group dissociates to form an H+ ion. In contrast, the hydrogen atom attached to the oxygen atom of an alcohol does not dissociate to form an H+ ion when an alcohol is dissolved in water. The reasons for the difference in behavior between carboxylic acids and alcohols will be discussed in Chapter 8.
Only the hydrogen attached to the oxygen atom of the CO2 group dissociates to form an H+ ion.
Bases
We will present more comprehensive definitions of bases in later chapters, but virtually every base you encounter in the meantime will be an ionic compound, such as sodium hydroxide (NaOH) and barium hydroxide [Ba(OH)2], that contain the hydroxide ion and a metal cation. These have the general formula M(OH)n. It is important to recognize that alcohols, with the general formula ROH, are covalent compounds, not ionic compounds; consequently, they do not dissociate in water to form a basic solution (containing OH− ions). When a base reacts with any of the acids we have discussed, it accepts a proton (H+). For example, the hydroxide ion (OH−) accepts a proton to form H2O. Thus bases are also referred to as proton acceptors.
Concentrated aqueous solutions of ammonia (NH3) contain significant amounts of the hydroxide ion, even though the dissolved substance is not primarily ammonium hydroxide (NH4OH) as is often stated on the label. Thus aqueous ammonia solution is also a common base. Replacing a hydrogen atom of NH3 with an alkyl group results in an amine (RNH2), which is also a base. Amines have pungent odors—for example, methylamine (CH3NH2) is one of the compounds responsible for the foul odor associated with spoiled fish. The physiological importance of amines is suggested in the word vitamin, which is derived from the phrase vital amines. The word was coined to describe dietary substances that were effective at preventing scurvy, rickets, and other diseases because these substances were assumed to be amines. Subsequently, some vitamins have indeed been confirmed to be amines.
Metal hydroxides (MOH) yield OH− ions and are bases, alcohols (ROH) do not yield OH− or H+ ions and are neutral, and carboxylic acids (RCO2H) yield H+ ions and are acids.
Summary
Common acids and the polyatomic anions derived from them have their own names and rules for nomenclature. The nomenclature of acids differentiates between oxoacids, in which the H+ ion is attached to an oxygen atom of a polyatomic ion, and acids in which the H+ ion is attached to another element. Carboxylic acids are an important class of organic acids. Ammonia is an important base, as are its organic derivatives, the amines.
Summary
- The composition of a compound is represented by an empirical or molecular formula, each consisting of at least one formula unit.
- Covalent inorganic compounds are named using a procedure similar to that used for ionic compounds, whereas hydrocarbons use a system based on the number of bonds between carbon atoms.
Covalent inorganic compounds are named by a procedure similar to that used for ionic compounds, using prefixes to indicate the numbers of atoms in the molecular formula. An empirical formula gives the relative numbers of atoms of the elements in a compound, reduced to the lowest whole numbers. The formula unit is the absolute grouping represented by the empirical formula of a compound, either ionic or covalent. Empirical formulas are particularly useful for describing the composition of ionic compounds, which do not contain readily identifiable molecules. Some ionic compounds occur as hydrates, which contain specific ratios of loosely bound water molecules called waters of hydration.