17.11: Storage Batteries
- Page ID
- 49566
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)A great disadvantage of the two cells we describe in Commercial Galvanic Cells is that they must be discarded once the cell reaction has gone to completion. Since they contain valuable metals like mercury and manganese whose supplies will soon become more limited, we can expect that batteries like this will become progressively more expensive and the impetus will grow to recycle the materials they contain, perhaps through a system of returnable deposits. A much more satisfactory solution to this problem, though, is the increased use of batteries which are rechargeable. Such batteries are called storage batteries, and they have the property that once the cell reaction has gone to completion, it can easily be reversed by electrolysis.
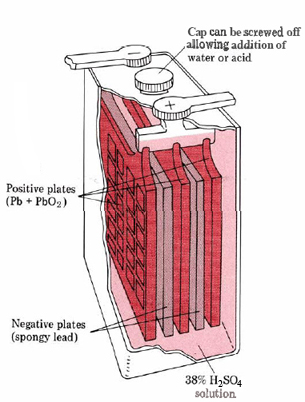 Figure \(\PageIndex{1}\) The lead storage battery.
Figure \(\PageIndex{1}\) The lead storage battery.
\[\text{Pb}, \text{PbSO}_4(s)\mid \text{H}_2\text{SO}_4(5 M)\mid \text{PbSO}_4(s), \text{PbO}_2(s), \text{Pb} \nonumber \] and the electrode reactions are and \[\ce{Pb(s) + SO4^{2–}(aq) -> PbSO4(s) + 2}e^– \nonumber \]
and
\[\ce{2}e^– \ce{+ 4H^{+} + SO_{4}^{2–} + PbO_{2} \rightarrow PbSO_{4}(s) + 2H_{2}O} \nonumber \]
giving an overall cell reaction \[\ce{Pb(s) + PbO_{2}(s) + 4H^{+} + 2SO_{4}^{2–} \rightarrow 2PbSO_{4} + 2H_{2}O} \nonumber \] Both electrodes are similarly constructed of a flat grid of lead. For the electrode, the interstices are filled with finely divided spongy Pb of high surface area, while for the right-hand electrode chocolate-brown lead dioxide powder, PbO2, is used.
As the cell discharges, both electrodes become plated with white PbSO4. Some storage batteries are made with transparent polystyrene cases. When this is the case, it is very apparent when the battery has been discharged since both electrodes begin to acquire a similar white appearance. The discharge of the battery also results in the consumption of H2SO4 [see Eq. (1)]. As the H2SO4 becomes more dilute, its density decreases, and so by measuring the density of the acid, we can decide whether the battery needs recharging or not.
Once a lead storage battery has been discharged, it can be recharged by electrolyzing it with a source of direct current. This results in the cell reaction reversing itself. The PbSO4 disappears from both electrodes, and the concentration of H2SO4 increases. Even when the cell becomes fully charged, little damage is done by continued electrolysis. H2 gas is evolved at one electrode and O2 at the other. Lead storage cells do not last indefinitely, however. Some of the PbSO4 formed at the electrodes becomes dislodged and falls to the bottom of the container where it is no longer available to take part in the recharging electrolysis. If lead storage batteries are allowed to discharge completely, this loss of PbSO4 particularly liable to occur. Batteries which are not mistreated in this way inevitably last longer. In a car battery three or six lead cells are connected in series. Since each produces 2.0 V when fully charged, the resultant potential difference is 6 or 12 V.
A second everyday example of a storage battery is the nickel-cadmium battery now commonly used in electronic calculators. These cells have the following construction:
\[\ce{Cd(s), CdO(s)} \mid \ce{KOH(1 M)} \mid \ce{Ni(OH)_{3}(s), Ni(OH)_{2}(s), Ni} \nonumber \] with the following electrode half-equations: \[\ce{Cd(s) + 2OH^{–} \rightarrow CdO + H_{2}O + 2}e^– \nonumber \]
\[\ce{2}e^– \ce{+ 2Ni(OH)_{3} \rightarrow 2Ni(OH)_{2} + 2OH^{–}} \nonumber \]
This cell produces a potential difference of 1.25 V and can be used in place of the dry cell for many purposes. A simple contribution anyone can make toward conserving the world’s supply of zinc is to invest in a simple battery charger and use nickel-cadmium storage batteries to replace dry cells.


