12.7: Group VIA- Chalcogens
- Page ID
- 49509
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)As we approach the right-hand side of the periodic table, similarities among the elements within a group become greater again. This is true of group VIA. Except polonium, which is radioactive and usually omitted from discussion, all members of the group form X2– ions when combined with highly electropositive metals. The tendency to be reduced to the –2 oxidation state decreases significantly from top to bottom of the group, however, and tellurium shows some metallic properties. The group VIA elements are called chalcogens because most ores of copper (Greek chalkos) are oxides or sulfides, and such ores contain traces of selenium and tellurium. Atomic properties of the chalcogens are summarized in the table.
| Element | Symbol | Electron Configuration | Usual Oxidation State | Radius/pm | |
|---|---|---|---|---|---|
| Covalent | Ionic (X2-) | ||||
| Oxygen | O | [He]2s22p4 | -2 | 66 | 140 |
| Sulfur | S | [Ne]3s23p4 | +6, +4, -2 | 104 | 184 |
| Selenium | Se | [Ar]4s23d104p4 | +6, +4, -2 | 117 | 198 |
| Tellurium | Te | [Kr]5s24d105p4 | +6, +4, -2 | 135 | 221 |
| Symbol | Ionization Energy/MJ mol–1 | Density/ g cm–3 | Electro- negativity | Melting Point (in °C) | ||
|---|---|---|---|---|---|---|
| First | Second | Third | ||||
| O | 1.320 | 3.395 | 5.307 | 1.43×10-3 | 3.5 | -218 |
| S | 1.006 | 2.257 | 3.367 | 2.06 | 2.5 | 119 |
| Se | 0.947 | 2.051 | 2.980 | 4.82 | 2.4 | 217 |
| Te | 0.876 | 1.800 | 2.704 | 6.25 | 2.1 | 450 |
At ordinary temperatures and pressures, oxygen is a gas. It exists in either of two allotropic forms: O2, which makes up 21 percent of the earth’s atmosphere, or O3 (ozone), which slowly decomposes to O2. O3 can be prepared by passing an electrical discharge through O2 or air:
\[\text{3O}_2(g) \xrightarrow{\text{electrical discharge}} \text{2O}_3(g) \nonumber \]
This reaction occurs naturally as a result of lightning bolts. O3 is also produced by any device which produces electrical sparks. You may have noticed its distinctive odor in the vicinity of an electric motor, for example.
Ozone is formed in the earth’s stratosphere (between altitudes of 10 and. 50 km) by ultraviolet rays whose wavelengths are shorter than 250 nm:
\[\text{O}_2 \xrightarrow{\text{ultraviolet light}} \text{2O} \nonumber \]
\[\text{O} + \text{O}_2 \rightarrow \text{O}_3\label{3} \]
The ozone itself absorbs longer-wavelength ultraviolet radiation (up to 340 nm), preventing these harmful rays fom reaching the earth’s surface. Otherwise these rays would increase the incidence of human skin cancer and cause other environmental problems. In recent years convincing evidence has been obtained to show that nitrogen oxide emissions from supersonic transport (SST) airplanes (which fly in the stratosphere) can reduce the concentration of ozone. Similar conclusions have been drawn regarding chlorofluorocarbons(sometimes referred to as CFCs) used as propellants in aerosol hair sprays and deodorants. Once in the atmosphere, a photochemical reaction causes atomic chlorine to be broken off from CFCs. This atomic chlorine can then participate in a catalytic ozone depleting reaction:
\[\text{Cl} + \text{O}_3 \rightarrow \text{ClO} + \text{O}_2 \nonumber \]
\[\text{ClO} + \text{O}_3 \rightarrow \text{Cl} + \text{2O}_2 \nonumber \]
Atomic chlorine is regenerated, meaning that each CFC molecule has the potential to deplete large amounts of ozone. In the 1980s, it was determined that use of chemicals such as CFCs were thinning stratospheric ozone. This is also when the "ozone hole" over Antarctica was discovered. In response to the depletion of ozone, and the danger presented by it, the Montreal Protocol on Substances that Deplete the Ozone Layer was signed by leaders of multiple countries, with the goal to phase out production and use of CFCs and other chemicals harmful to the ozone layer. Today, 191 countries have signed the protocol, and while it is projected to take until 2075 for ozone levels to return to normal, the Montreal Protocol has so far proven a success[1].
O3 is also an important component of photochemical smog. It is produced when O atoms (formed by breaking N—O bonds in NO2) react with molecules according to Eq. \(\ref{3}\). O3 is a stronger oxidizing agent than O2. It reacts with unsaturated hydrocarbons (alkenes) in evaporated gasoline to produce aldehydes and ketones which are eye irritants. Rubber is a polymeric material which contains 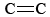 bonds, and so it too reacts with O3. Further, ground level ozone and the accompanying smog has proven a significant health concern, irritating and damaging the respiratory system and also having links to asthma[2]. So ozone is beneficial when in the upper atmosphere, but has adverse effects when at ground level.
bonds, and so it too reacts with O3. Further, ground level ozone and the accompanying smog has proven a significant health concern, irritating and damaging the respiratory system and also having links to asthma[2]. So ozone is beneficial when in the upper atmosphere, but has adverse effects when at ground level.
Sulfur occurs in a variety of allotropic forms. At room temperature the most stable form is rhombic sulfur. This yellow solid consists of S8 molecules (seen in the Jmol below) packed in a crystal lattice which belongs to the orthorhombic system (listed on the page discussing crystal systems).
Figure \(\PageIndex{1}\) S8 molecule: The initial ball and stick model can be manipulated in three dimensional space. Click on VdW radii to see a space filling model of the same molecule.
When heated to 96°C, solid rhombic sulfur changes very slowly into monoclinic sulfur, in which one-third of the S8 molecules are randomly oriented in the crystal lattice. When either form of sulfur melts, the liquid is at first pale yellow and flows readily, but above 160°C it becomes increasingly viscous. Only near the boiling point of 444.6°C does it thin out again. This unusual change in viscosity with temperature is attributed to opening of the eight-membered ring of S8 and formation of long chains of sulfur atoms. These intertwine and prevent the liquid from flowing. This explanation is supported by the fact that if the viscous liquid is cooled rapidly by pouring it into water, the amorphous sulfur produced can be shown experimentally to consist of long chains of sulfur atoms.
Both selenium and tellurium have solid structures in which the atoms are bonded in long spiral chains. Both are semiconductors, and the electrical conductivity of selenium depends on the intensity of light falling on the element. This property is utilized in selenium photocells, which are often used in photographic exposure meters.
Selenium is also used in rectifiers to convert alternating electrical current to direct current. Compounds of selenium and tellurium are of little commercial importance, and they often are toxic. Moreover, many of them have foul odors, are taken up by the body, and are given off in perspiration and on the breath. These properties have inhibited study of tellurium and selenium compounds.
Chemical Reactions and Compounds
Oxygen Since oxygen has the second largest electronegativity among all the elements, it is found in the –2 oxidation state in most compounds. Important oxides have already been discussed in sections dealing with the elements from which they form, and so we will deal only with unusual oxidation states of oxygen here. One of these is the +2 state found in OF2, the most common compound in which oxygen is combined with the more electronegative fluorine. We have already mentioned the –½ and –1 states observed in alkali-metal superoxides and peroxides, but one important peroxide, hydrogen peroxide (H2O2), has not yet been discussed.
H2O2 can be prepared by electrolysis of solutions containing sulfate ions. H2O2 is a weak acid, and it can serve as an oxidizing agent (oxygen being reduced to the –2 state) or as a reducing agent (oxygen being oxidized to the 0 state). Like the peroxide ion, the H2O2 molecule contains an O—O single bond. This bond is rather weak compared with many other single bonds, and this contributes to the reactivity of H2O2. The compound decomposes easily, especially if exposed to light or contaminated with traces of transition metals. The decomposition
\[\text{2H}_2\text{O}_2(l) \rightarrow \text{2H}_2\text{O}(l) + \text{O}_2(g) \nonumber \] can occur explosively in the case of the pure liquid. Sulfur Although this element is only sixteenth in abundance at the surface of the earth, it is one of the few that has been known and used throughout history. Deposits of elemental sulfur are not uncommon, and, because they were stones that would burn, were originally called brimstone. Burning sulfur produces sulfur dioxide,
\[\text{S}_8(s) + \text{8O}_2(g) \rightarrow \text{8SO}_2(g) \nonumber \]
This colorless gas has a choking odor and is more poisonous than carbon monoxide. It is the anhydride of sulfurous acid, a weak diprotic acid:
\[\text{SO}_2(g) + \text{H}_2\text{O}(l) \rightarrow \text{H}_2\text{SO}_3(aq) \nonumber \]
SO2 is also produced when almost any sulfur-containing substance is burned in air. Coal, for example, usually contains from 1 to 4% sulfur, and so burning coal releases SO2 to the atmosphere. Many metal ores are sulfides, and when they are heated in air, SO2 is produced. Copper, for example, may be obtained as the element by heating copper(I) sulfide:
\[\text{Cu}_2\text{S}(s) + \text{O}_2(g) \xrightarrow{\Delta } \text{2Cu}(s) + \text{SO}_2(g) \nonumber \]
Since SO2 is so poisonous, its release to the atmosphere is a major pollution problem. Once in the air, SO2 is slowly oxidized to sulfur trioxide, SO3:
\[\text{2SO}_2(g) + \text{O}_2(g) \rightarrow \text{2SO}_3(g) \nonumber \]
This compound is the anhydride of sulfuric acid, H2SO4:
\[\text{SO}_3(g) + \text{H}_2\text{O}(l) \rightarrow \text{H}_2\text{SO}_4(aq) \nonumber \]
Thus if air is polluted with SO2 and SO3, a fine mist of dilute droplets of can form. All three substances are very irritating to the throat and lungs and are responsible for considerable damage to human health.
The natural mechanism for removal of sulfur oxides from the air is solution in raindrops, followed by precipitation. This makes the rainwater more acidic than it would otherwise be, and acid rain is now common in industrialized areas of the United States and Europe. Acid rain can slowly dissolve limestone and marble, both of which consist of CaCO3:
\[\text{CaCO}_3(s) + \text{H}_3\text{O}^{+}(aq) \rightarrow \text{Ca}^{2+}(aq) + \text{HCO}_3^{-}(aq) + \text{H}_2\text{O}(l) \nonumber \]
Thus statues and buildings made of these materials may be damaged.
Despite the fact that a tremendous amount of sulfur is released to the environment by coal combustion and ore smelting, this element is not usually recovered from such processes. Instead it is obtained commercially from large deposits along the U.S. Gulf Coast and from refining of sour petroleum. Sour petroleum contains numerous sulfur compounds, including H2S, which smells like rotten eggs. The deposits of elemental sulfur in Texas and Louisiana are mined by the Frasch process. Water at 170°C is pumped down a pipe to melt the sulfur, and the latter is forced to the surface by compressed air. Most of the H2S or S8 obtained from these sources is oxidized to SO2, passed over a vanadium catalyst to make SO3, and dissolved in water to make H2SO4. In 2005 an estimated 190 billion kg of H2SO4 was produced in the world, making H2SO4 one of the most important industrial chemicals. About half of it is used in phosphate fertilizer production[3].
Pure H2SO4 is a liquid at room temperature and has a great affinity for H2O. This is apparently due to the reaction:
\[\text{H}_2\text{SO}_4 + \text{H}_2\text{O} \rightarrow \text{H}_3\text{O}^{+} + \text{HSO}_4^{-} \nonumber \]
Formation of H3O+ releases energy, and the reaction is exothermic. Concentrated H2SO4 is 93% H2SO4 and 7% H2O by mass, corresponding to more than twice as many H2SO4 as H2O molecules. Since many H2SO4 molecules still have protons to donate, concentrated H2SO4 also has a great affinity for H2O. It is often used as a drying agent and can be employed in condensation reactions which give off H2O.
- ↑ "Montreal Protocol-One Page Factsheet." Environmental Protection Agency. 6 September 2007. http://www.epa.gov/ozone/downloads/MP20_FactSheet.pdf
- ↑ Kenneth Olden. "Statement on Health Effects of Air Pollution before the Senate Committee on Health, Education, Labor and Pensions, Subcommittee on Health. Department of Health and Human Services. 3 September 2003.www.hhs.gov/asl/testify/t020903.html
- ↑ Bala Suresh and Yoshio Inoguchi. "Sulfuric Acid". SRI Consulting. July 2006. http://www.sriconsulting.com/CEH/Pub...orts/781.5000/


