1.6: Reduction of Ores
- Page ID
- 190008
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Metal ores are typically salts, such as oxides, carbonates or sulfides Conversion of these ores into metals requires oxidation/reduction reactions.
That's not always the case. Some early forays into metallurgy involved native gold (native meaning the metal is found in its elemental state in nature). Gold is relatively soft. It could be easily worked and shaped by heating it. Occasionally, native silver and copper can also be found.
Explain, with the help of a table of standard reduction potentials, why silver and gold can sometimes be found as elements rather than salts.
- Answer
-
Both reduction potentials are very positive.
Ag+ + e- → Ag (s) E0 = + 0.796 V
Au+ + e- → Au (s) E0 = + 1.83 V
That means both metals are likely to be found in the reduced state.
However, a major leap forward came when people learned to make alloys, mixing in small amounts of other metals to make harder, sturdier materials. For example, the addition of tin to copper ushered in "the bronze age". Tin itself had to be made from its ore via smelting; the earliest evidence for this process comes from what is now Turkey, where it was performed over eight thousand years ago. However, alloys were apparently not discovered until several thousand years later.
In smelting, ore is heated to a high temperature in the presence of carbon sources, such as charcoal or coke. The partial combustion of the carbon source produces carbon monoxide which acts as a reducing agent.
Show the half-reactions involved in the reduction of tin oxide with carbon monoxide. Use them to come up with a balanced reaction for the process, and calculate the standard potential for the reaction.
- Answer
-
SnO (s) + 2H+ + 2 e- → Sn (s) + H2O E0 = -0.10 V
CO(g) + H2O → CO2(g) + 2H+ + 2 e- E0 = - (-0.11 V)
SnO (s) + CO (g) → Sn (s) + CO2 (g) ΔE0 = + 0.01 V
Another major advance in metallurgy involved the conversion of iron ores into iron and steel. There is evidence that iron smelting in sub-Saharan Africa and Sri Lanka about three thousand years ago. Archaeological evidence in Sri Lanka shows that smelters were located on mountainsides facing the ocean, where constant winds provided ample oxygen to produce fires hot enough for smelting.
In the United States, the discovery of iron ores in the states along the Great Lakes, the use of the Great Lakes as a transportation network, and the availability of anthracite coal in Pennsylvania fueled the development of an American steel industry and the rise of a major industrial power. The fact that the great lakes states are still referred to as the "rust belt" is a testament to the manufacturing prowess of the region throughout the twentieth century, which proceeded from having all the necessary features for an iron-based economy in close geographic proximity.
Show the half-reactions involved in the reduction of iron oxide with carbon monoxide. Assume the iron oxide is prsent as magnetite, Fe3O4. Use them to come up with a balanced reaction for the process, and calculate the standard potential for the reaction.
- Answer
-
Fe3O4(s) + 8 H+ + 8 e- → 3Fe(s) + 4 H2O E0 = +0.085 V
CO(g) + H2O → CO2(g) + 2H+ + 2 e- E0 = - (-0.11 V)
Fe3O4(s) + 4CO (g) → 3Fe(s) + 4CO2 (g) ΔE0 = + 0.195 V
Show the half-reactions involved in the reduction of aluminum oxide with carbon monoxide. Assume the aluminum is present as an ion in Al2O3. Use the half-reactions to come up with a balanced reaction for the process, and calculate the standard potential for the reaction.
- Answer
-
Al3+(aq) + 3e- → Al (s) E0 = -1.662 V
CO(g) + H2O → CO2(g) + 2H+ + 2 e- E0 = - (-0.11 V)
2 Al3+(aq) + 3CO (g) + 3H2O → 2Al(s) + 3CO2 (g) ΔE0 = - 1.772 V
Aluminum is a very important material in our economy. It is lightweight, strong, and forms a very hard oxide coating when exposed to the elements, rather than the rust that results from weathering steel. In contrast to the steel industry, the aluminum industry is a far-flung operation in which ore mined on one continent may be shipped to another for processing. However, aluminum metal isn't accessible via smelting. So how is it done?
Just as a thermodynamically favored redox reaction can produce a voltage in a circuit, if we already have a voltage produced by another source, we can drive an unfavorable redox reaction to completion. We can drive the reaction backwards.
Quebec is a major producer of aluminum, despite being endowed with virtually no aluminum ore. Bauxite, the major aluminum-containing ore, is a mixture of minerals of formulae Al(OH)3 or AlO(OH) found amalgamated with other clays and minerals. It is found near the earth's surface in tropical and sub-tropical areas, left behind after of millenia of erosion and drainage of more soluble materials from underlying bedrock. The major producers of bauxite are Vietnam, Australia and Guinea, as well as a number of countries in South America.
Why ship bauxite all the way to the taiga to make aluminum? Aluminum production requires a lot of electrons, and those electrons can't be provided by coal or coke. Instead, they usually come from massive hydroelectric generating stations, such as the 16,000 megawatt James Bay Project in northern Quebec. To make aluminum, you go where electricity is cheap and plentiful.
The bauxite is first processed to help remove all those other materials that come mixed with the aluminum ore. It is dissolved in base, filtered and re-precipitated with acid. The residue is heated to drive off water, leaving pure alumina (Al2O3).
Instead of performing this redox reaction in aqueous solution, it is done in the molten state. Alumina has a melting point around 2,000 oC, but that temperature drops to a much more manageable 1,000 oC if a "flux" is added. To avoid contaminating the aluminum ions, cryolite has often been used as the flux, because it is also an aluminum salt (Na3AlF6).
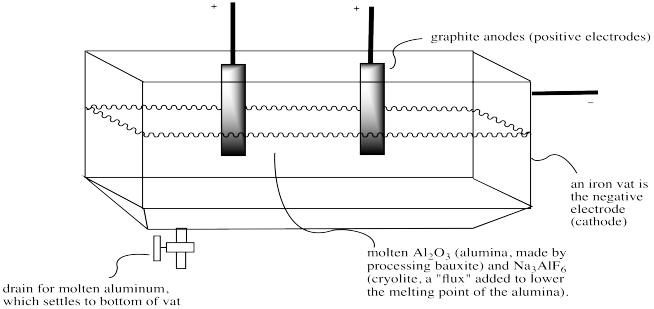
The alumina is melted in an iron vat, which conveniently functions as one of the electrodes in the redox reaction. It is the cathode, supplying electrons. Graphite anodes draw electrons out of the bath to complete a circuit. Two reactions occur: aluminum ions are reduced to aluminum at the cathode, which drops to the bottom of the vat and is drained away periodically. Oxide ions are oxidized to molecular oxygen at the anodes. However, at these temperatures, the oxygen quickly reacts with the carbon anodes to produce carbon dioxide -- that is, the anodes actually disappear as the reaction proceeds.
Take a look at the redox reaction happening in the vat.
- Provide a half reaction for reduction of aluminum ion.
- Provide a half reaction for oxidation of the oxide anion.
- Provide an overall, balanced reaction.
- Calculate the standard potential for this reaction. (Don't worry about the lack of an aqueous solution; we'll just get an estimate of the real potential. Also, you can use the value of the reduction potential of oxygen to yield hydroxide as an approximation)
- Answer
-
Al3+ + 3e- → Al (s) E0 = -1.662 V
2 O2- → O2(g) + 4e- E0 = - (.40 V, estimated)
4 Al3+(aq) + 6 O2- (aq) → 4 Al (s) + 3O2 (g) ΔE0 = - 2.062 V
Cryolite (Na3AlF6) is added to get alumina to melt at a lower temperature. Unlike bauxite, it's a somewhat rare mineral found in Greenland and Quebec. Presumably, the aluminum ions in the cryolite also get reduced. Wouldn't the rare cryolite quickly get used up? Explain why this isn't a problem.
- Answer
-
Even if the aluminum ions from the cryolite are reduced to Al0, they will be replenished by new aluminum ions from the bauxite ore.


