6.14: Solutions for Selected Problems.
- Page ID
- 200849
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
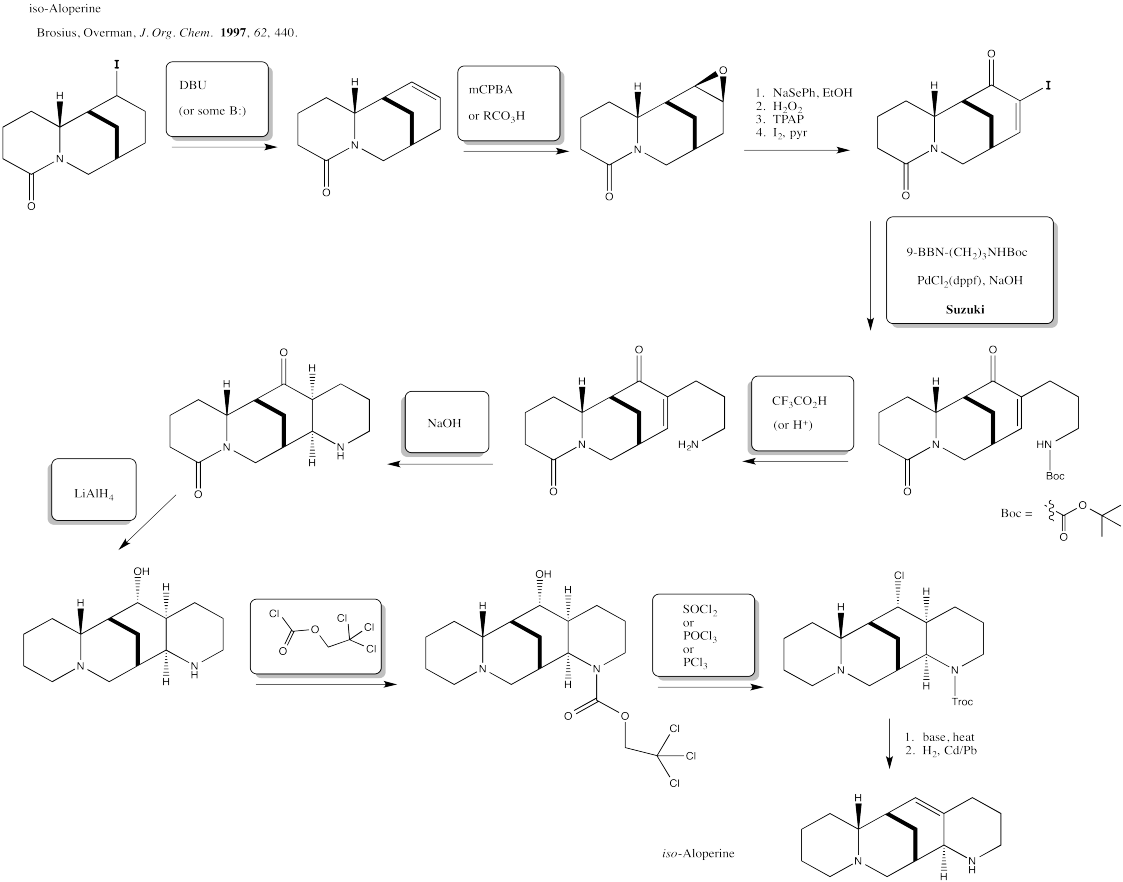
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Exercise 6.1.1:
El = electrophile; Nu = nucleophile
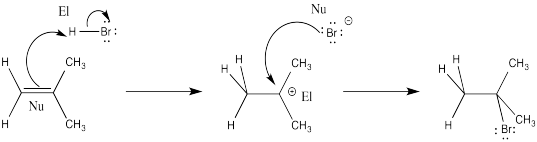
Exercise 6.1.2:
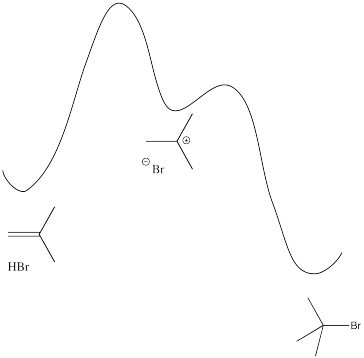
Exercise 6.1.3:
\[Rate = \frac{d[alkyl \: bromide]}{dt} = k[alkene][HBr] \nonumber\]
Exercise 6.1.4:
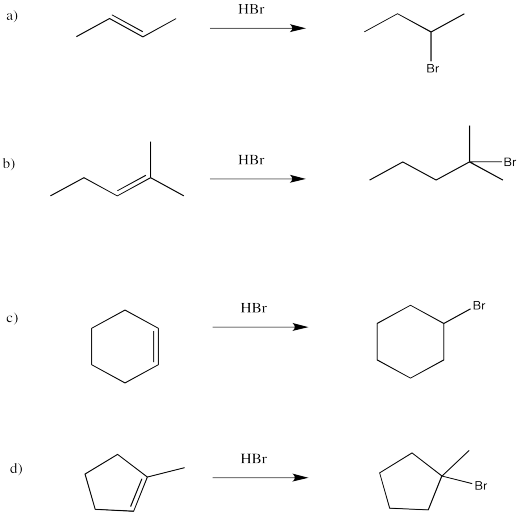
Exercise 6.2.1:
a, e, f, g are prochiral.
Exercise 6.2.2:
a) re b) si c) re d) si e) either; if the Br adds on one end of the double dond it is re, but at the other it is si f) re
Exercise 6.2.3:
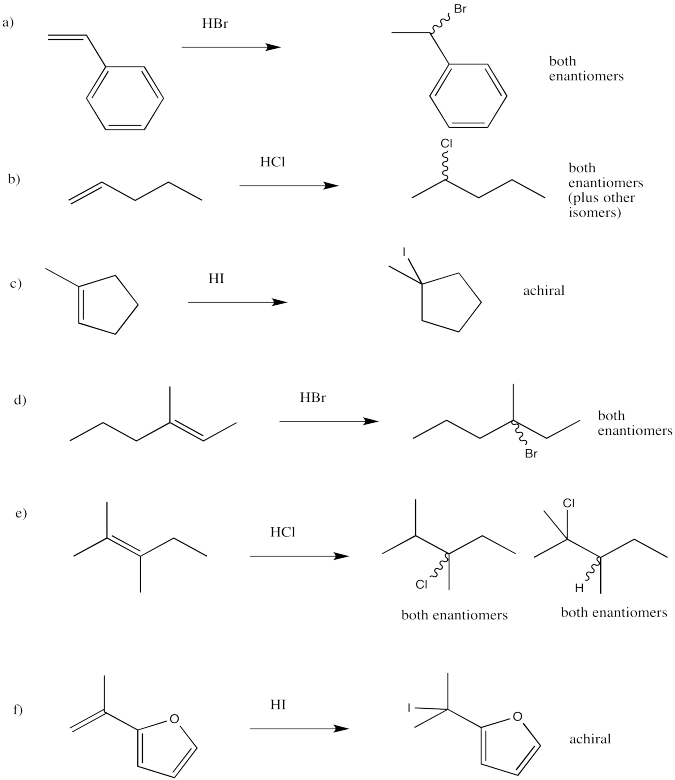
Exercise 6.2.4:
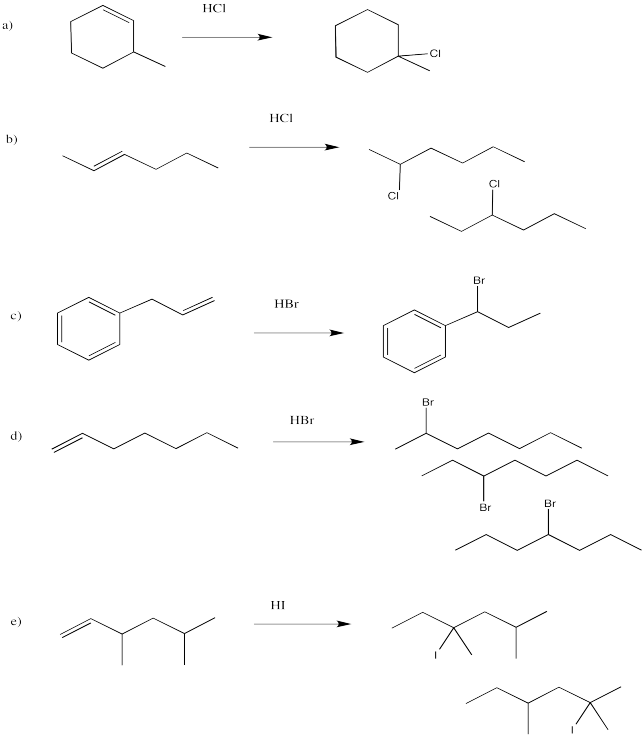
Exercise 6.3.1:
If the acid is regenerated at the end of the reaction, it isn't a reagent. It is a catalyst. It makes addition of water to the double bond occur much more quickly than if water acted alone, since water would never manage to protonate the alkene.
Exercise 6.3.2:
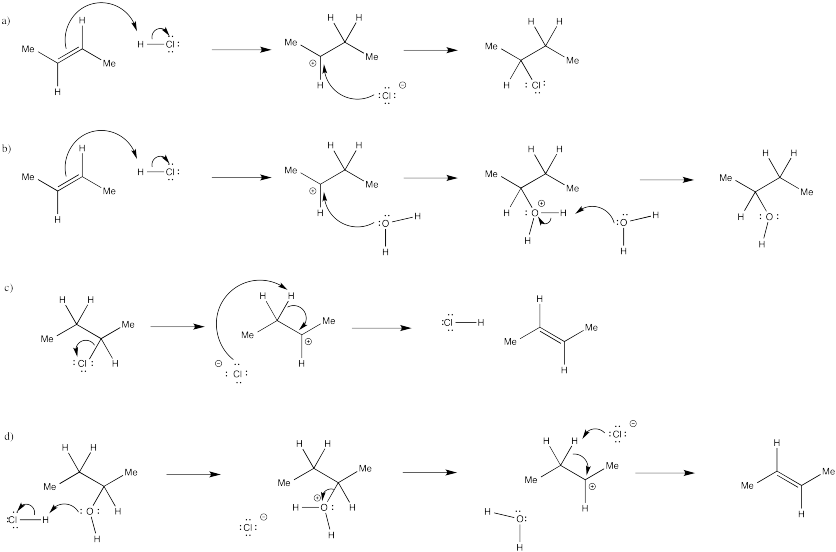
Exercise 6.3.3:
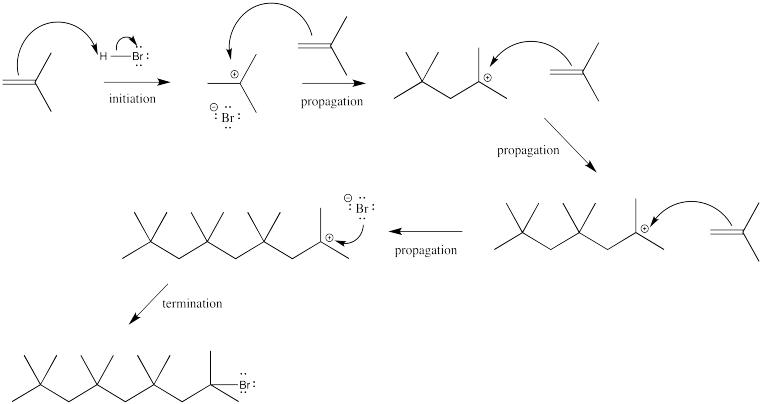
Exercise 6.4.1:
Two products are formed and they are enantiomers.
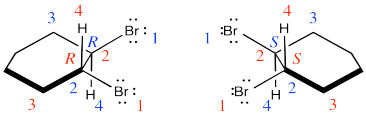
Exercise 6.4.2:
They are diastereomers. One chiral center has the same configuration in both compounds but the others are opposite.
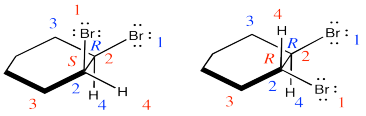
Exercise 6.4.3:
The second bromine could occupy any of the secondary positions if there were a true carbocation. That doesn't happen; the second bromine occupies only the position next to the other bromine.
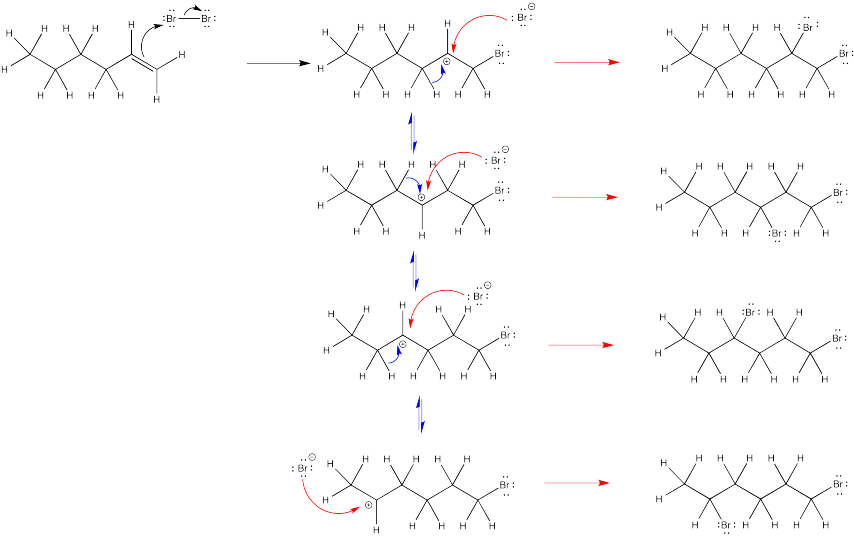
Exercise 6.4.5:
The nucleophile in the second step changes under different conditions.
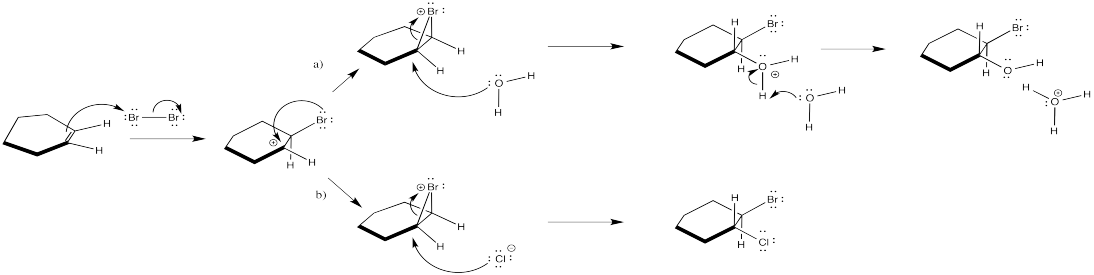
Exercise 6.4.6:
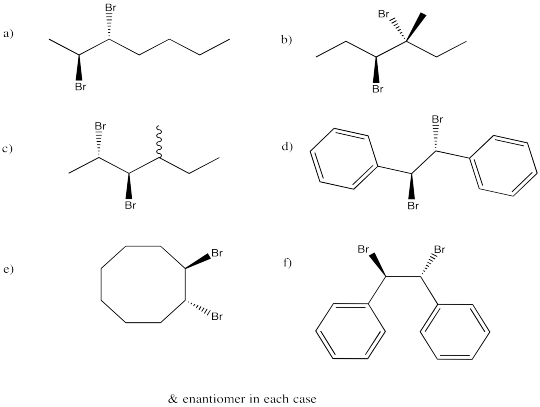
Exercise 6.5.2:
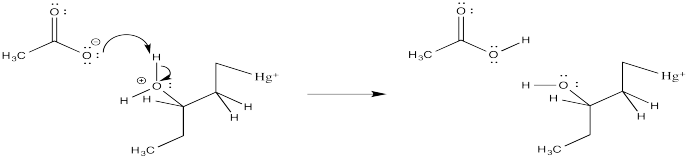
Exercise 6.5.3:
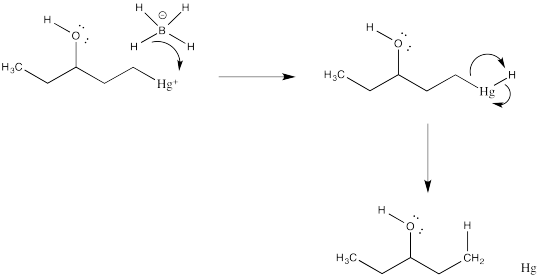
Exercise 6.5.4:
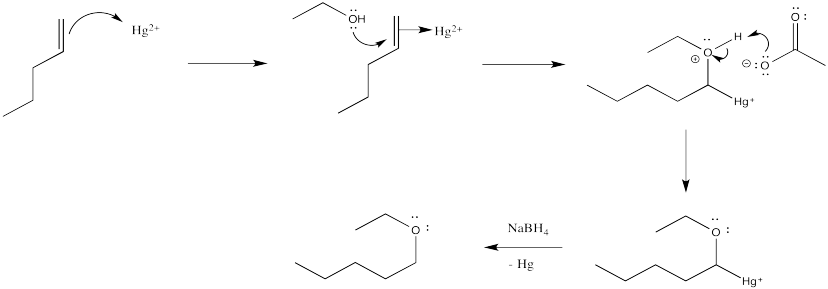
Exercise 6.5.5:
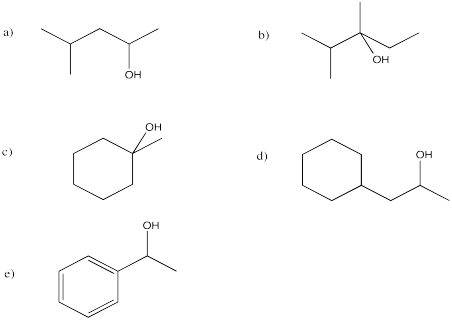
Exercise 6.5.6:
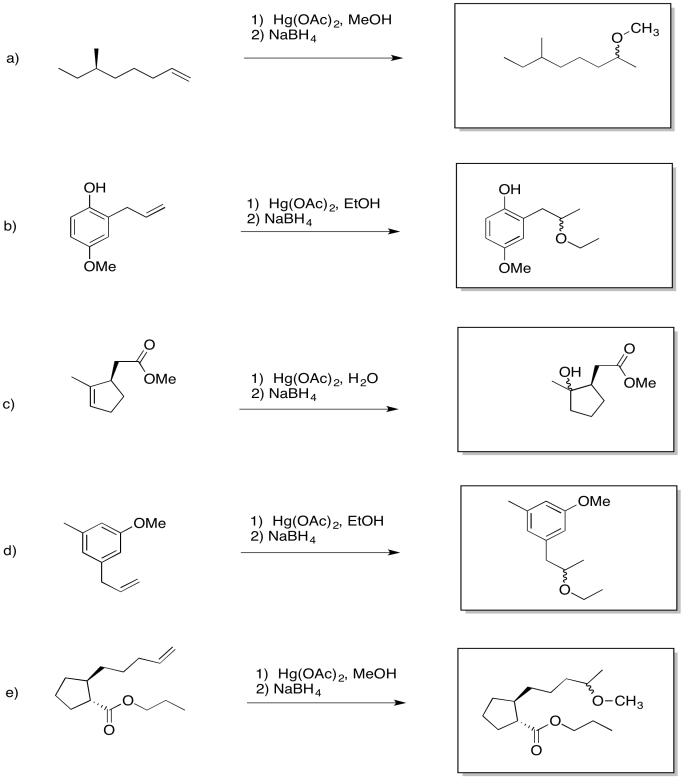
Exercise 6.6.1:
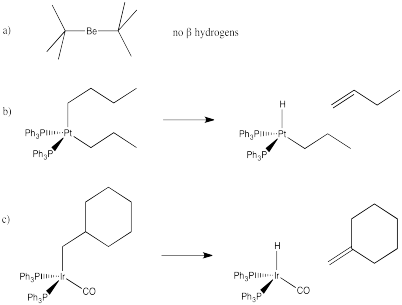
Exercise 6.6.2:
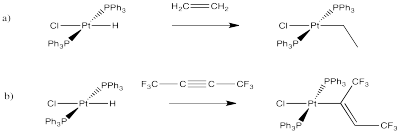
Exercise 6.6.3:
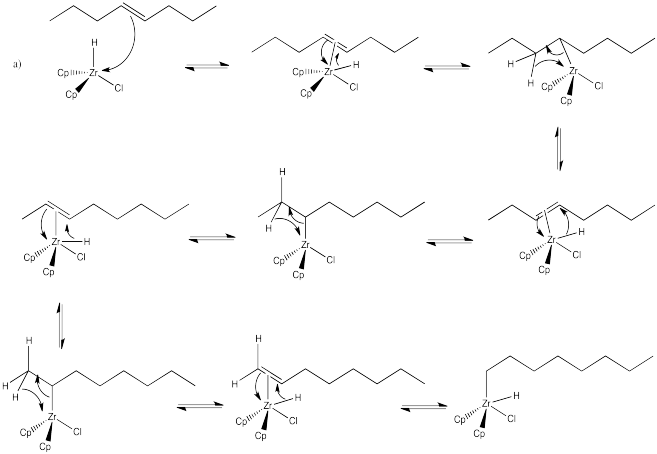
Exercise 6.6.4:
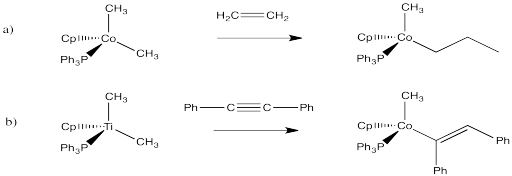
Exercise 6.6.5:
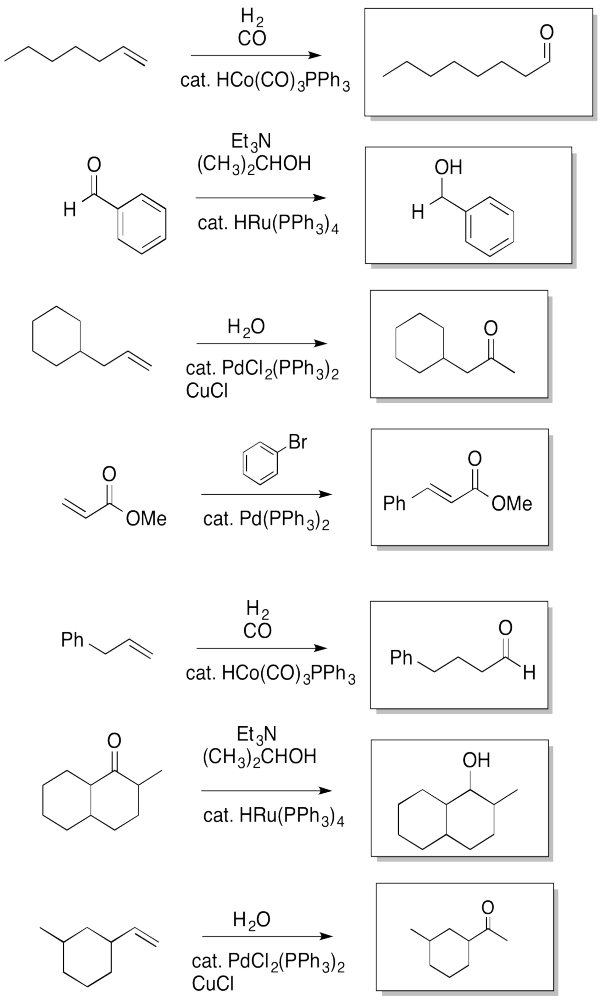
Exercise 6.6.6:
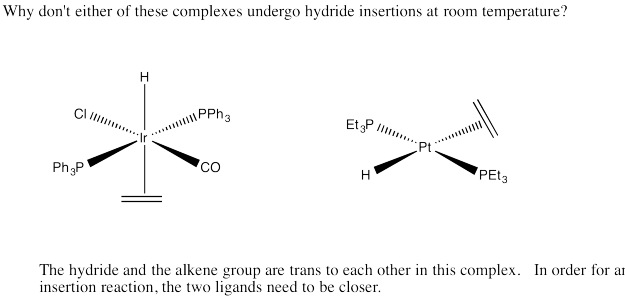
Exercise 6.6.7:
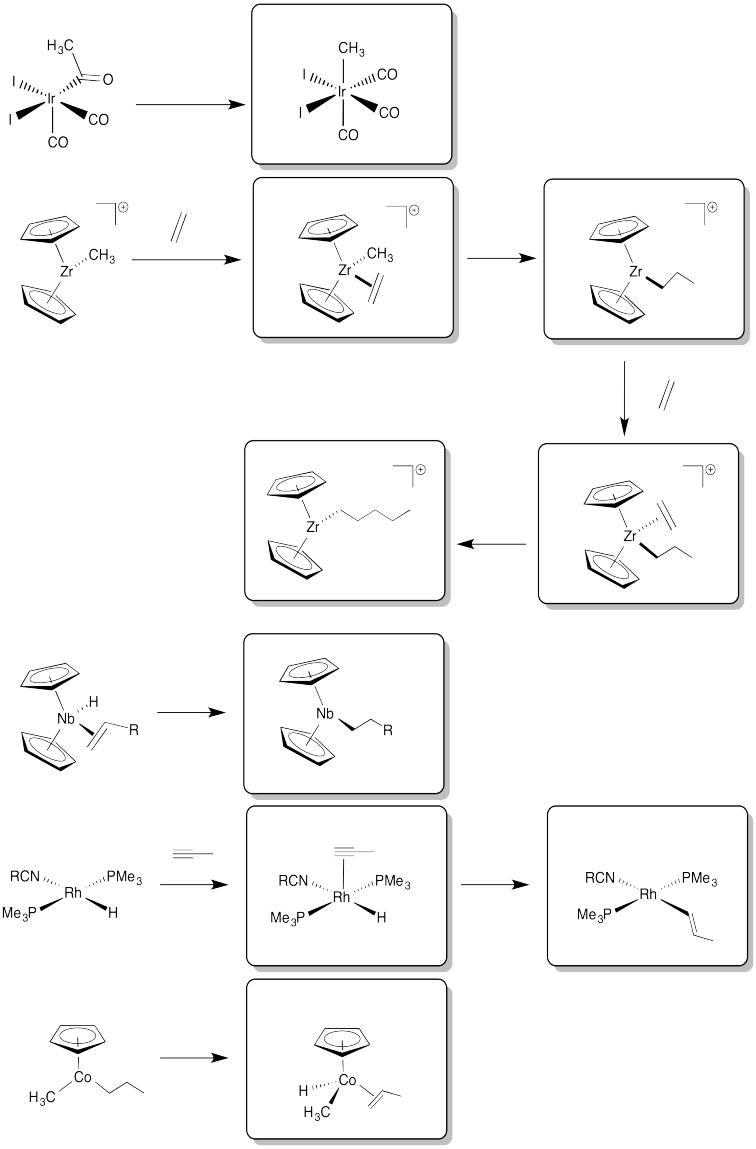
Exercise 6.6.8:
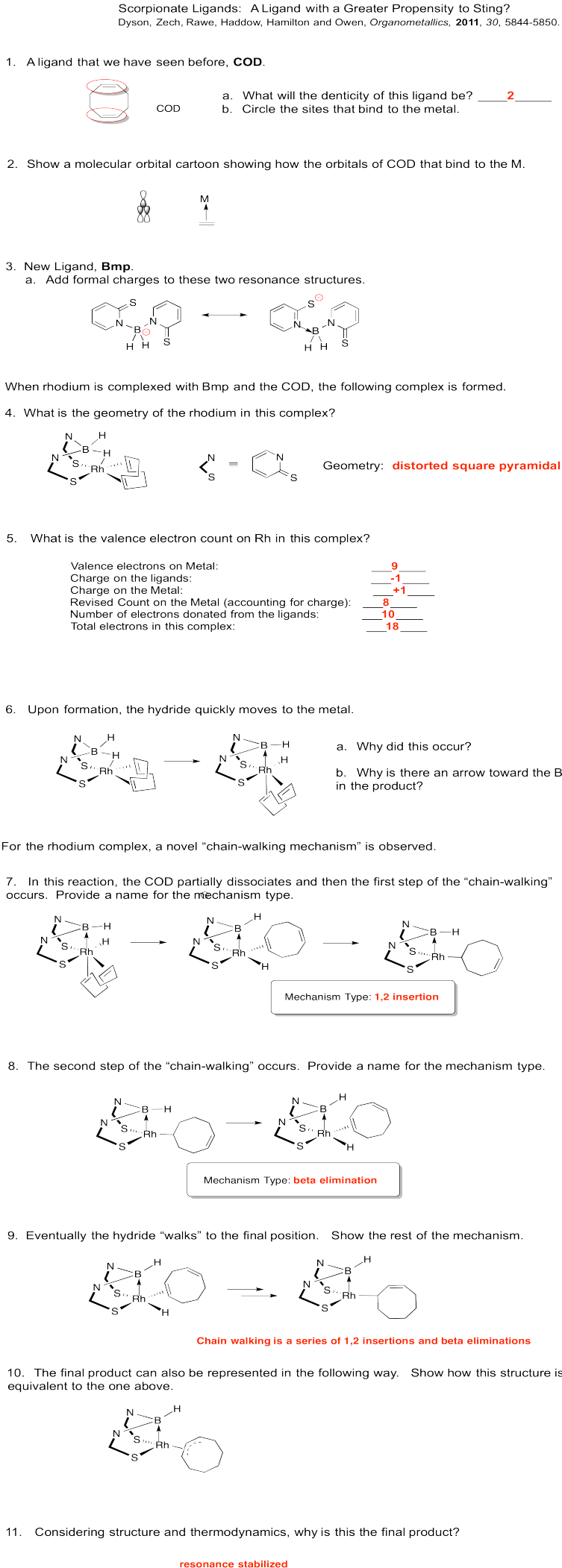
Exercise 6.6.9:
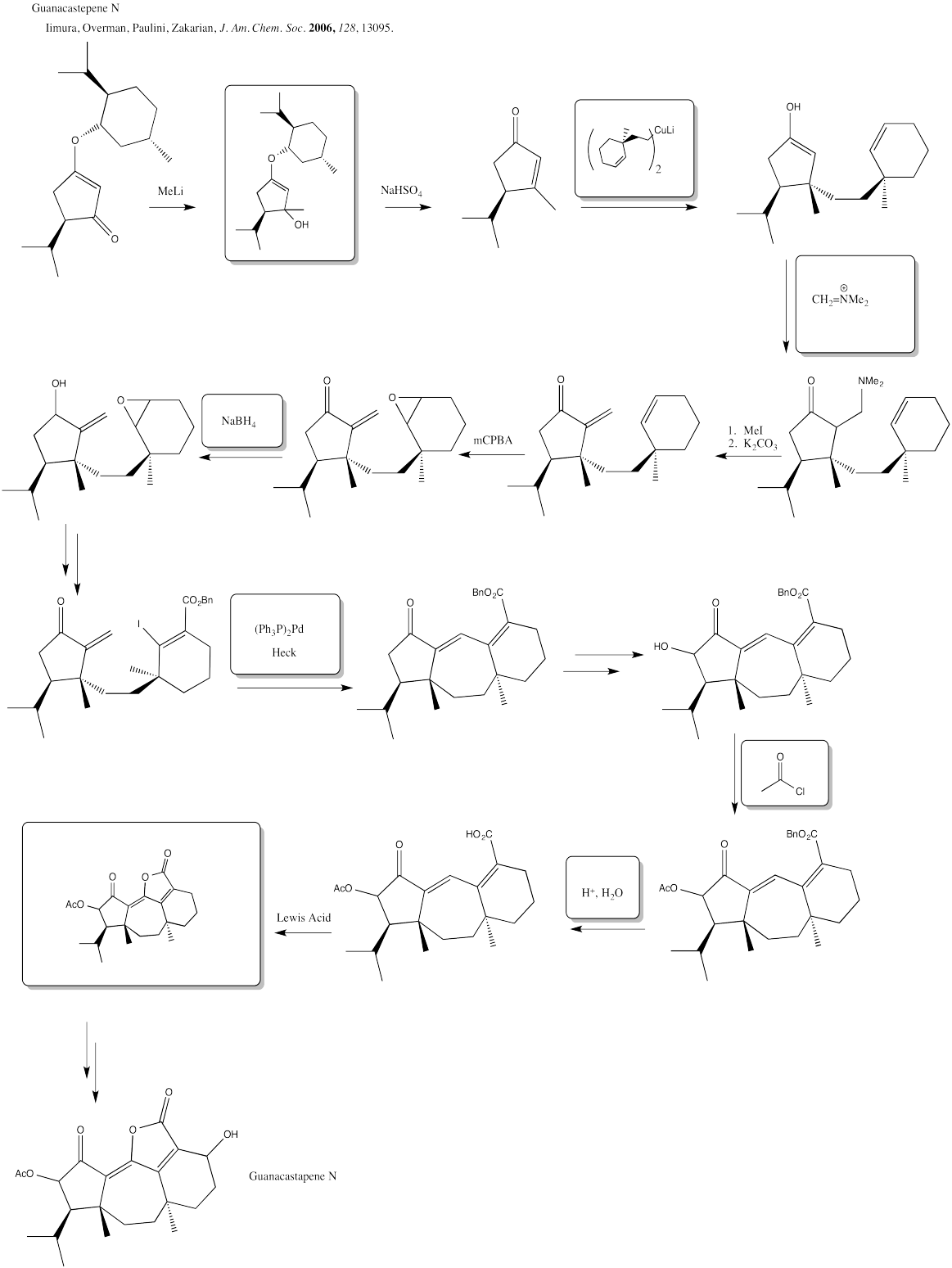
Exercise 6.6.10:
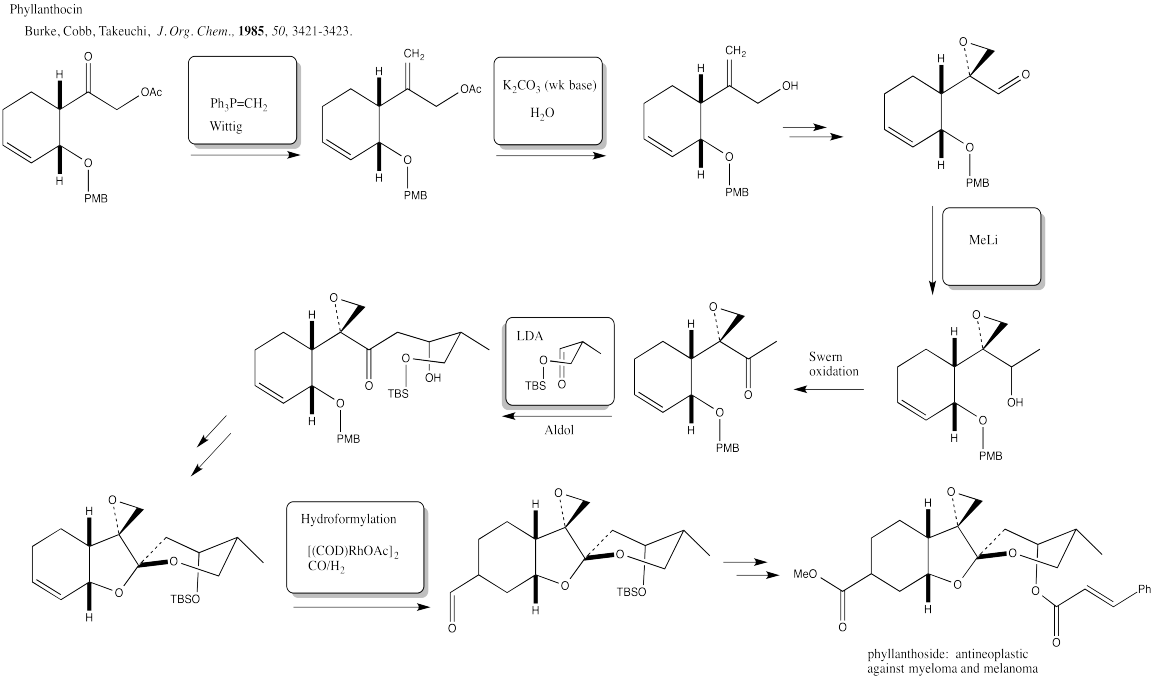
Exercise 6.7.2:
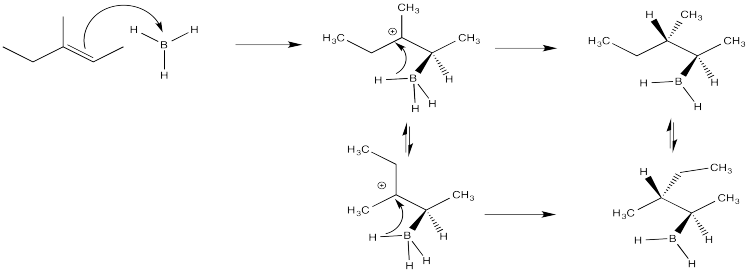
Exercise 6.7.3:
Crowding is more severe in the structure on the left than in the structure on the right. The structure on the right, representing an approach to the transition state of the reaction, is more favorable than the other one.

Exercise 6.7.4:
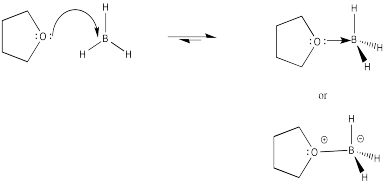
Exercise 6.7.5:
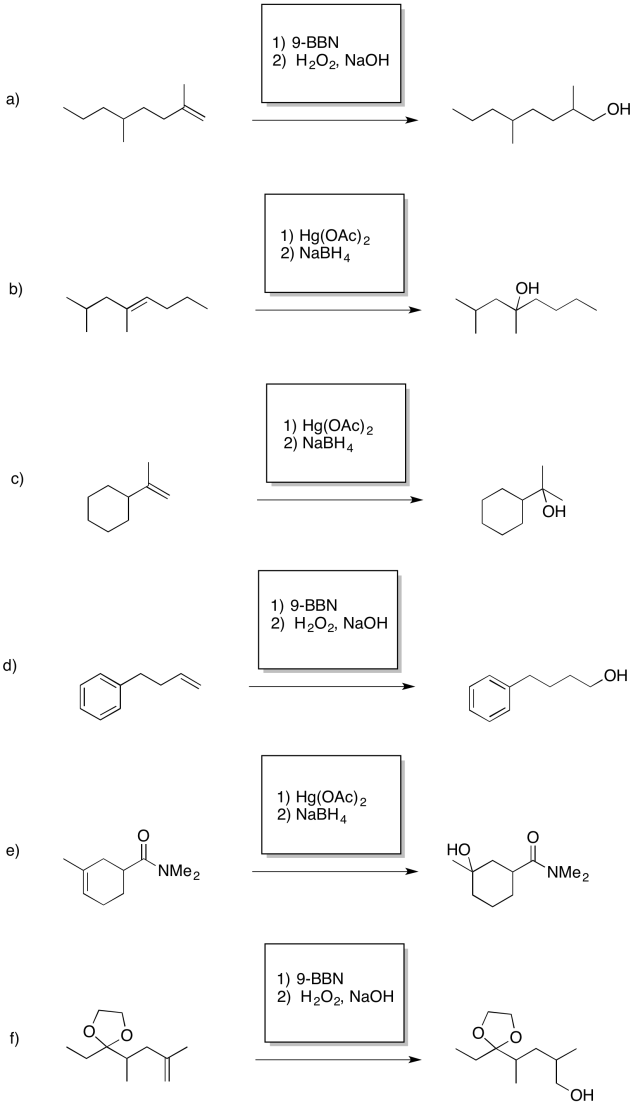
Exercise 6.7.6:
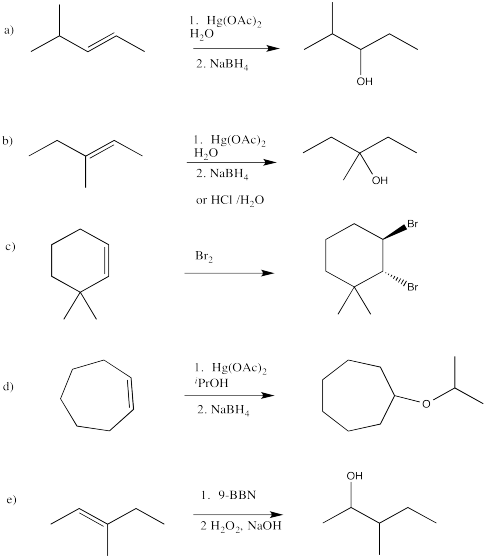
Exercise 6.7.7:
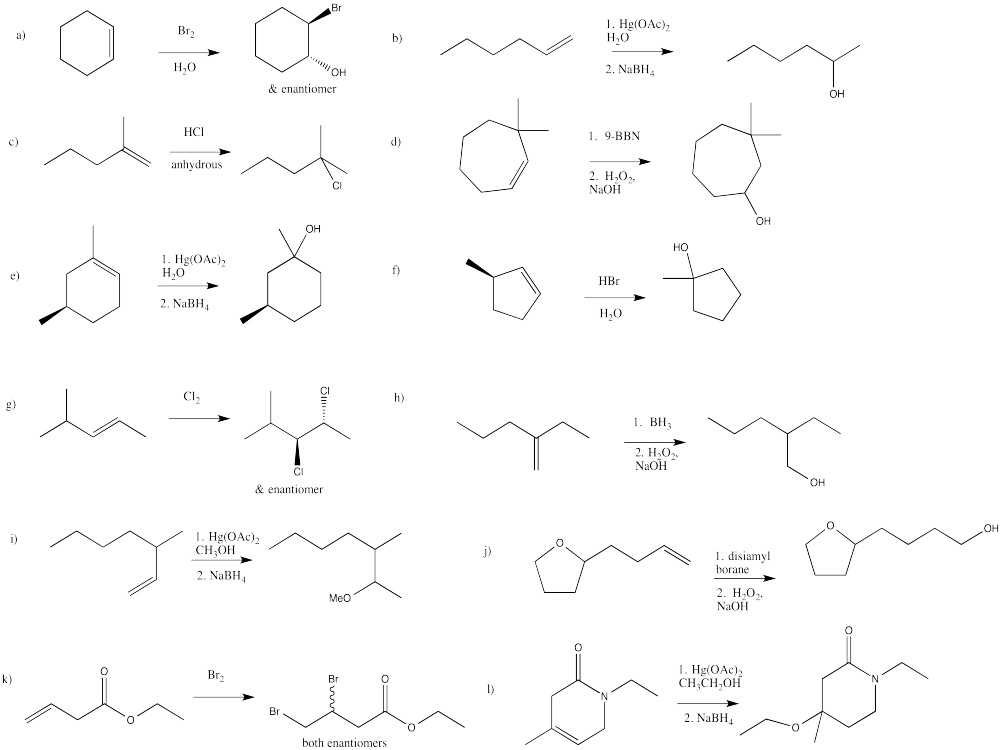
Exercise 6.8.1:

Exercise 6.8.2:
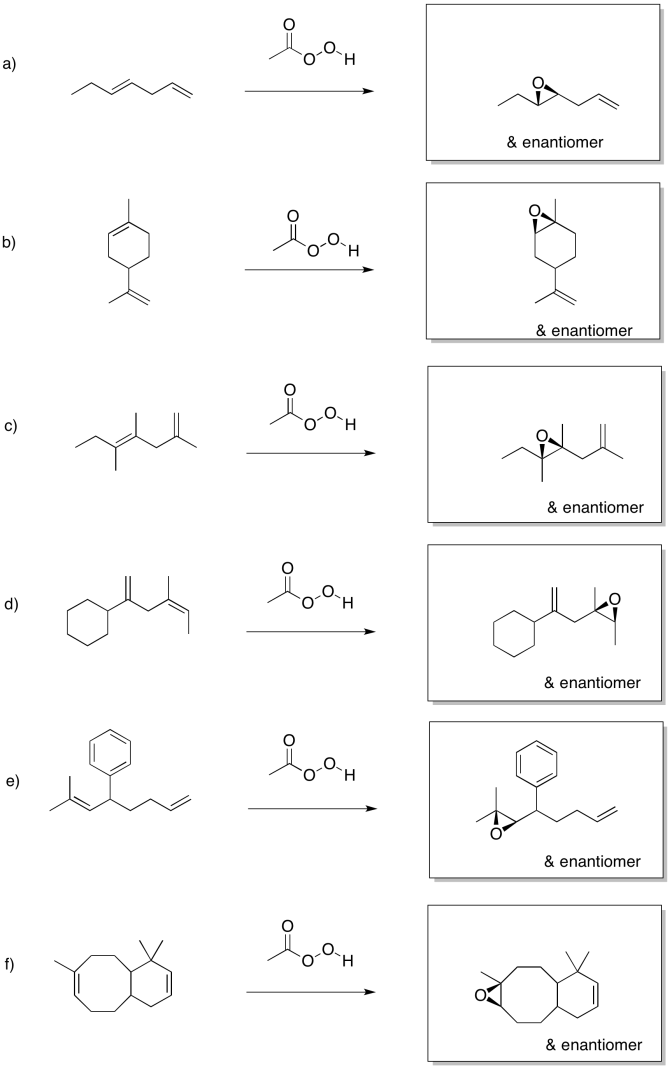
Exercise 6.8.3:

Exercise 6.8.4:
D-(-)-tartrate is the (2S,3S)-isomer. L-(+)-tartrate is the (2R,3R)-isomer. Each chiral center is configured opposite to the corresponding one in the other molecule, so the molecules are enantiomers.
Exercise 6.8.5:
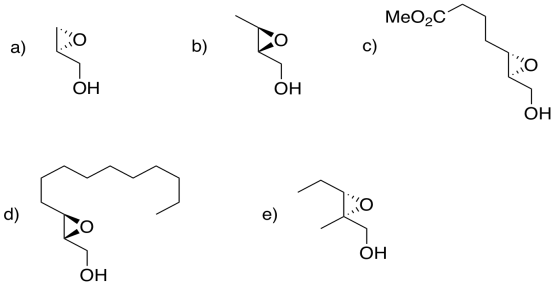
Exercise 6.8.6:
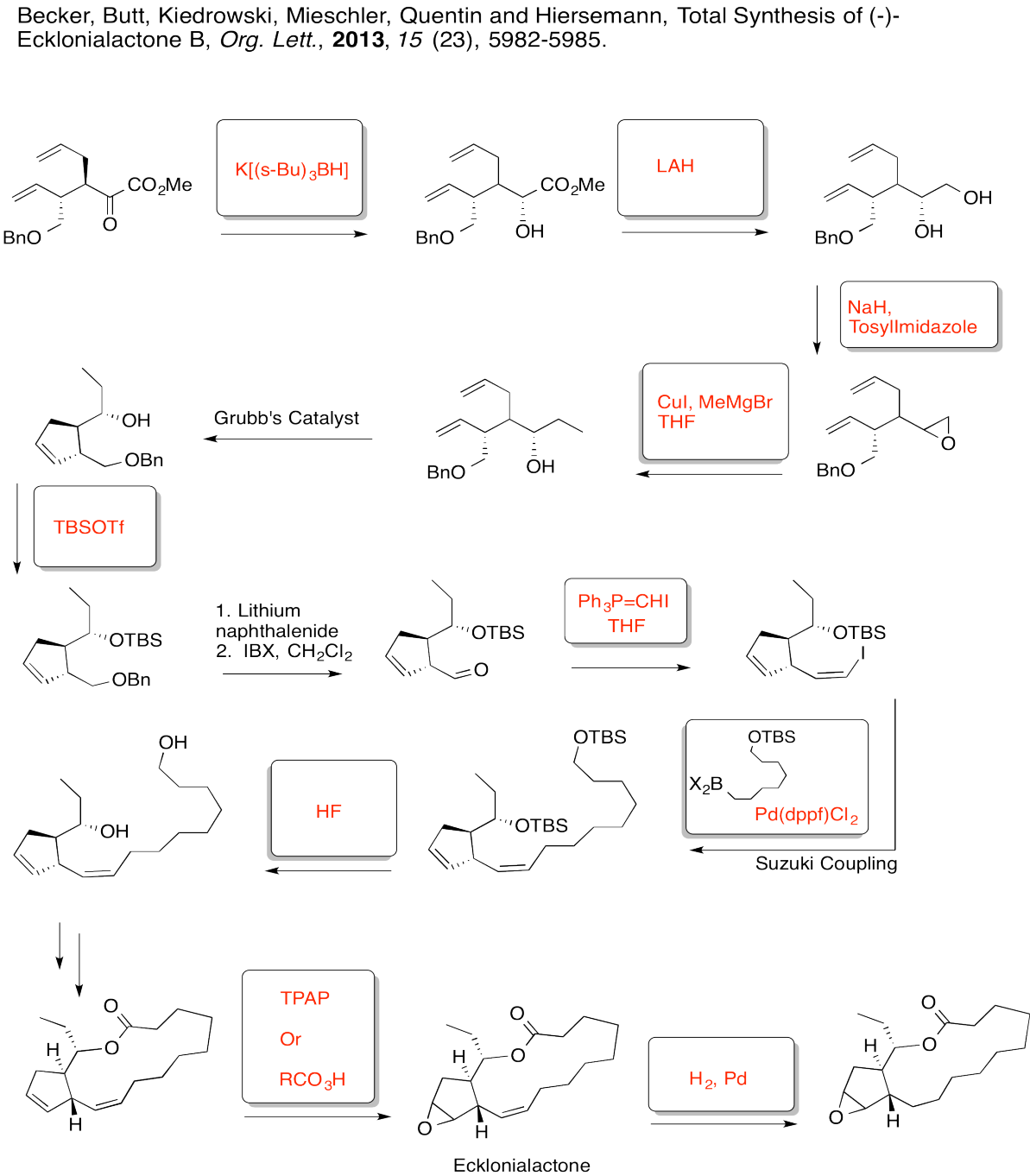
Exercise 6.8.7:
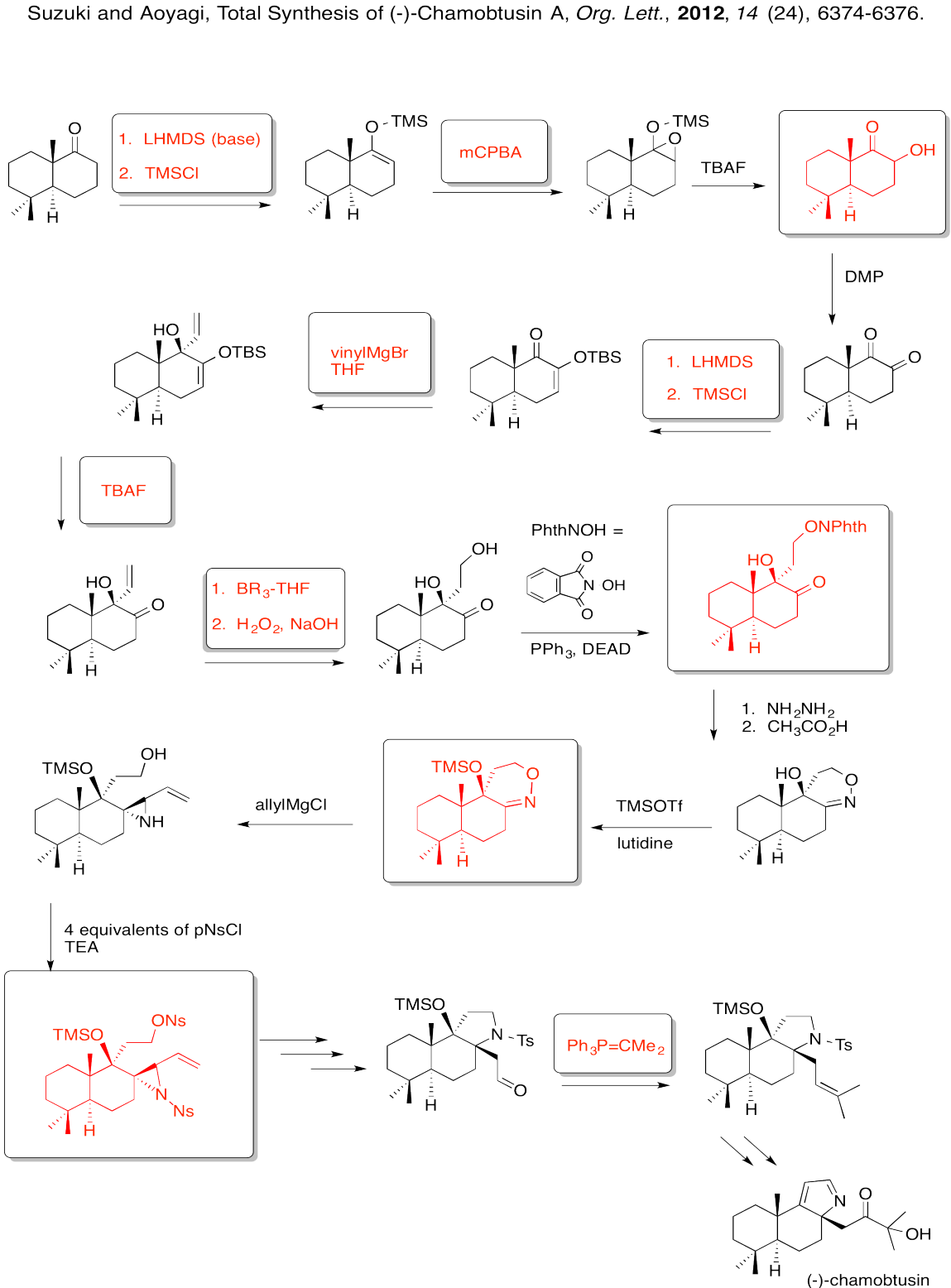
Exercise 6.8.8:
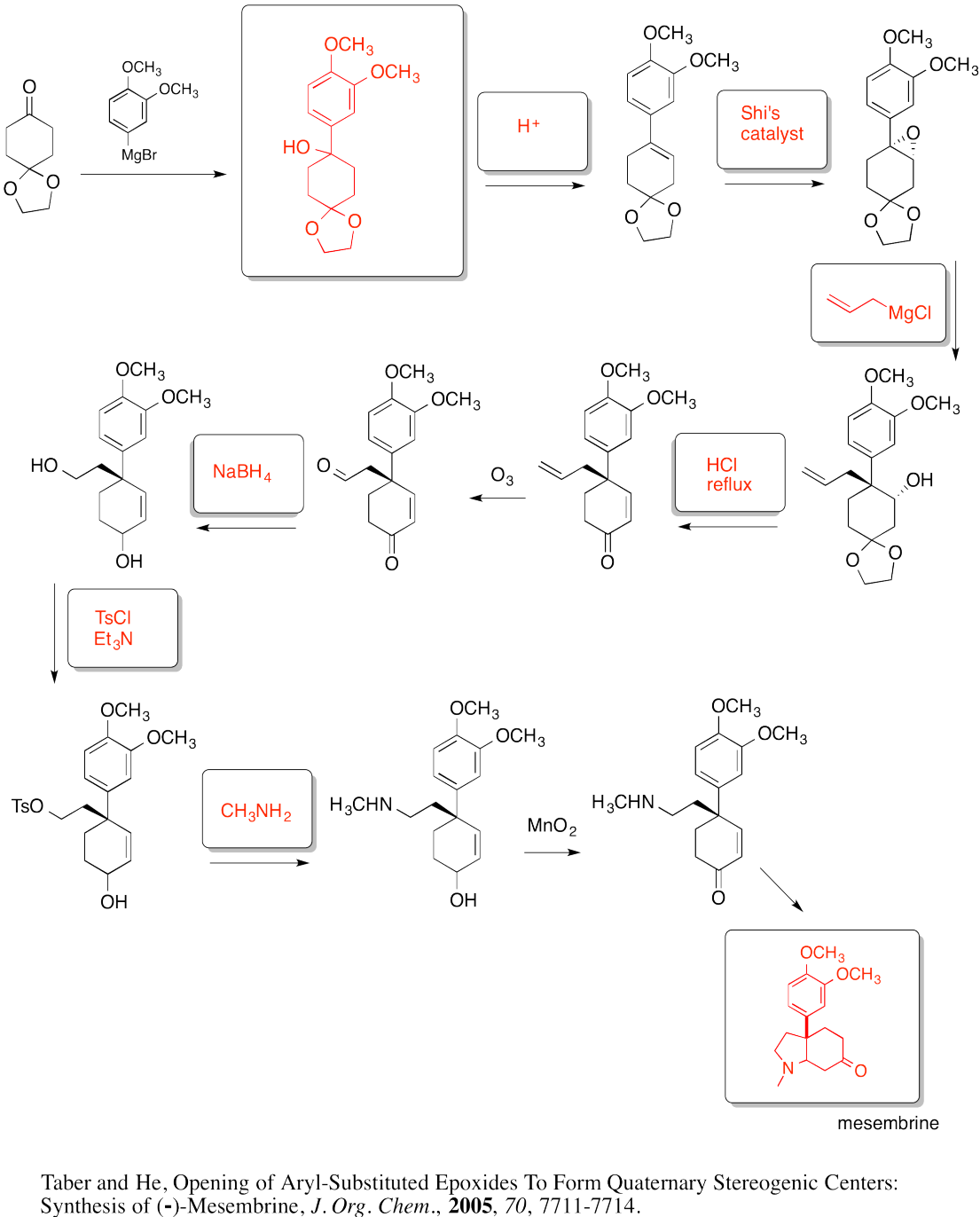
Exercise 6.9.1:
The chlorines can (weakly) share their electrons to fill the octet on carbon.
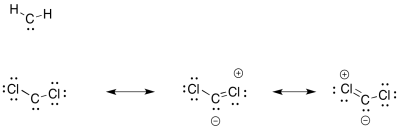
Exercise 6.9.2:
The oxygen can π-donate to help fill the octet on the carbon.
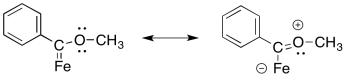
Exercise 6.9.3:
Not only can the nitrogens π-donate to help fill the octet on carbon, but this is an aromatic system. It is planar, cyclic, fully conjugated, with an odd number of electron pairs in the π-system.
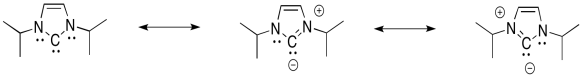
Exercise 6.9.4:
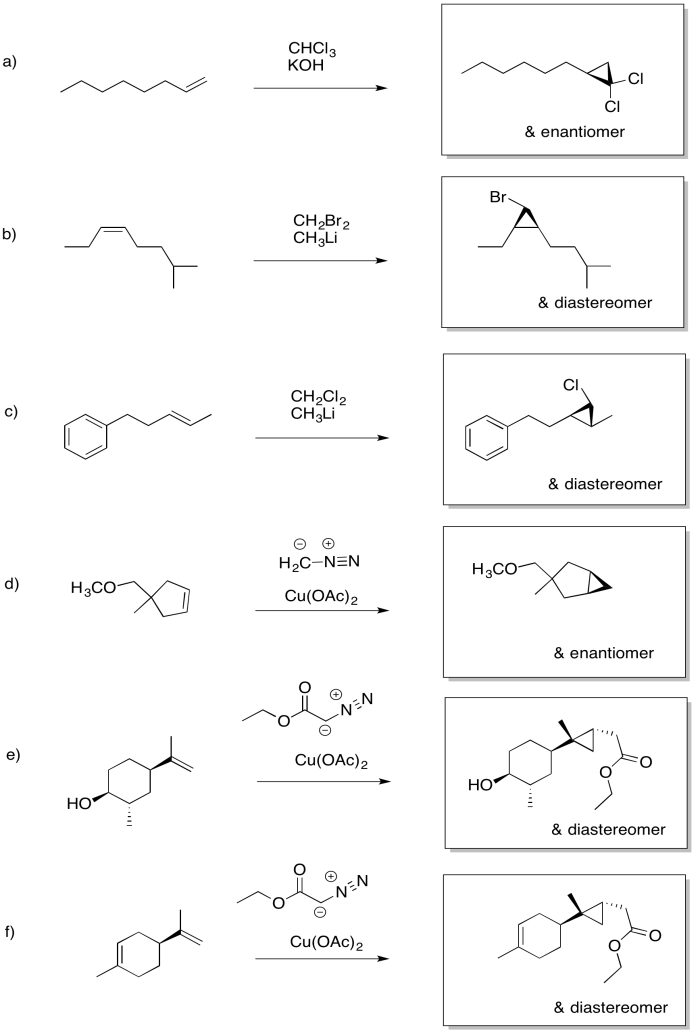
Exercise 6.10.1:
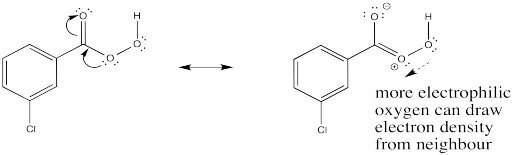
Exercise 6.10.2:
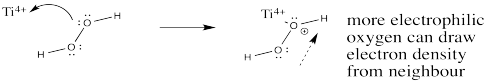
Exercise 6.10.3:
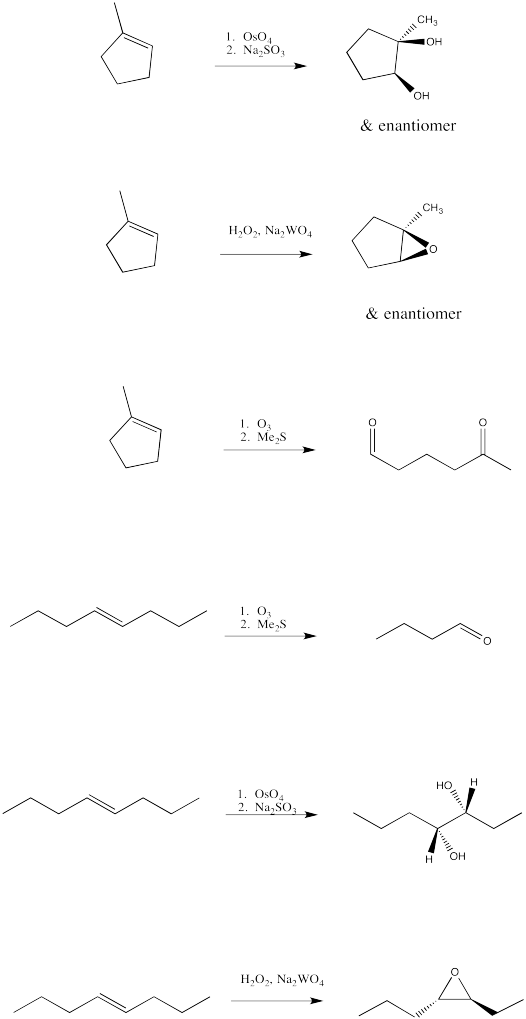
Exercise 6.10.4:
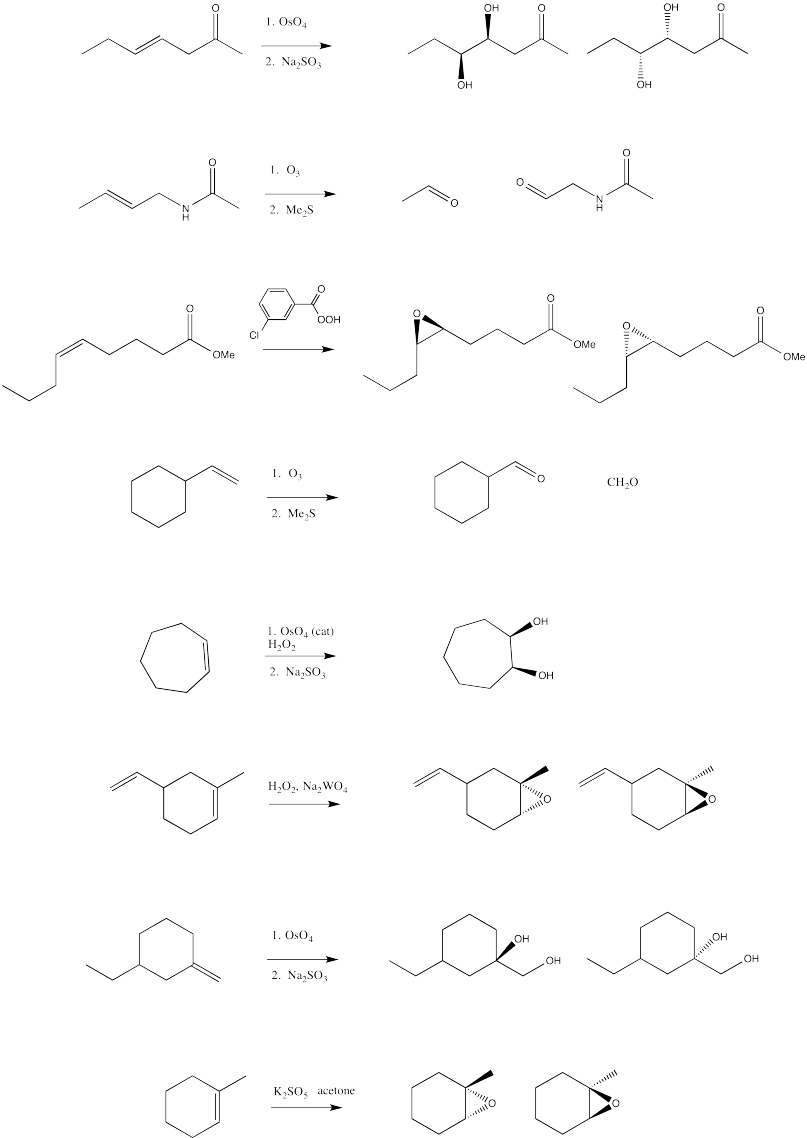
Exercise 6.10.5: