Solutions, Solvation, and Dissociation
- Page ID
- 53091
Skills to Develop
- Describe factors affecting dissolution
Dissolution means the process of dissolving or forming a solution. When dissolution happens, the solute separates into ions or molecules, and each ion or molecule is surrounded by molecules of solvent. The interactions between the solute particles and the solvent molecules is called solvation. A solvated ion or molecule is surrounded by solvent.
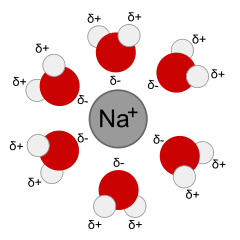
- A sodium ion solvated by water, from Wikimedia Commons.
Technically a solvent can mean anything that is the more abundant component of a homogeneous mixture, but usually it means a volatile liquid that things can easily dissolve in. (Volatile means that it can easily evaporate, like water or alcohol.) The most common solvent is water. When you scuba dive in the ocean, you will need to rinse your gear with water afterwards to remove the salt. The salt dissolves in the water, gets washed away, and then the water evaporates, leaving the gear clean. This is the typical action of a solvent.
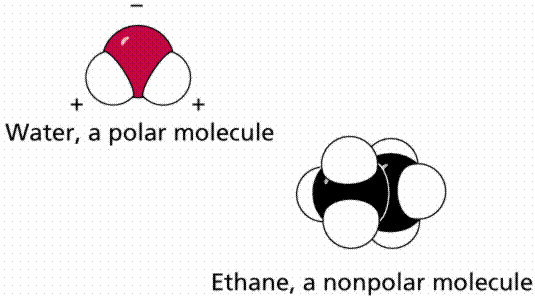
Solvents are either polar or non-polar. A polar solvent has partial negative and positive charges. For instance, water has a partial negative charge on O and a partial positive charge on H. The symbol δ means a partial charge, less than the charge on one proton or electron, such as δ+ or δ–. This helps the solvent interact with (solvate) ions and polar molecules through Coulomb interactions. A non-polar solvent is one that is electrically neutral all over, or almost so. Oil, or the gas in your car, are examples of non-polar liquids that could be used as solvents. Non-polar solvents are only good for dissolving non-polar solutes, which is why water, salt and sugar don't mix into oil.
Contributors and Attributions
Emily V Eames (City College of San Francisco)

