19.8: Nuclear Stability
- Page ID
- 49600
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
Why is it that certain combinations of nucleons are stable in a nucleus while others are not? A complete answer to this question cannot yet be given, largely because the exact nature of the forces holding the nucleons together is still only partially understood. We can, however, point to several factors which affect nuclear stability. The most obvious is the neutron/proton ratio. As we discuss in "Further Modes of Decay", if this is too high or too low, it makes for an unstable nucleus.
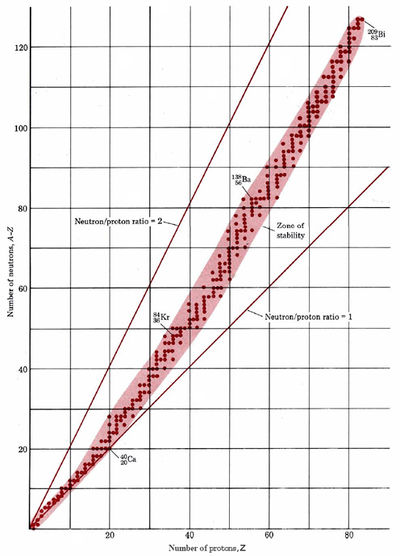
If we plot the number of neutrons against the number of protons for all known stable (i.e., nonradioactive) nuclei, we obtain the result shown in Figure \(\PageIndex{1}\). All the stable nuclei lie within a definite area called the zone of stability. For low atomic numbers most stable nuclei have a neutron/proton ratio which is very close to 1. As the atomic number increases, the zone of stability corresponds to a gradually increasing neutron/proton ratio. In the case of the heaviest stable isotope, \(\ce{_{83}^{209}Bi}\) for instance, the neutron/proton ratio is 1.518. If an unstable isotope lies to the left of the zone of stability in Figure \(\PageIndex{1}\), it is neutron rich and decays by β emission. If it lies to the right of the zone, it is proton rich and decays by positron emission or electron capture.
Another factor affecting the stability of a nucleus is whether the number of protons and neutrons is even or odd. Among the 354 known stable isotopes, 157 (almost half) have an even number of protons and an even number of neutrons. Only five have an odd number of both kinds of nucleons. In the universe as a whole (with the exception of hydrogen) we find that the even-numbered elements are almost always much more abundant than the odd-numbered elements close to them in the periodic table.
Finally there is a particular stability associated with nuclei in which either the number of protons or the number of neutrons is equal to one of the so-called "magic" numbers 2, 8, 20, 28, 50, 82, and 126. These numbers correspond to the filling of shells in the structure of the nucleus. These shells are similar in principle but different in detail to those found in electronic structure. Of particular stability, and also of high abundance in the universe, are nuclei in which both the-number of protons and the number of neutrons correspond to magic numbers. Examples are 42He, 168O, 4020Ca, and 20882Pb.
Find which element has the largest number of isotopes, using Figure \(\PageIndex{1}\). Likewise find which is the number of neutrons which occurs most frequently. What do you notice about the numbers of protons and neutrons in each case?
Solution:
Tin has 10 isotopes, and its atomic number 50 is a magic number. A total of 7 stable isotopes have 82 neutrons in the nucleus, more than for any other number of neutrons. Again the number is a magic number.


