Determination of Chloride using Potentiometry
- Page ID
- 78035
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)1. Purpose
This procedure will determine the concentration of chloride ion with a chloride specific ion electrode using potentiometry.
2. Background
Potentiometry is an electrochemical method in which the potential of an electrochemical cell is measured while little to no current is passed through the sample. In titrimetric methods, this measurement can be used to indicate the end point. When an ion selective electrode is used, the measured potential is related to the ion concentration in solution and a quantitative determination can directly be made. The instrumentation used to perform potentiometry is straightforward and relatively inexpensive, consisting of an indicator electrode, a reference electrode, and a potential measuring device. One of the most common applications of potentiometry is in the measurement of pH.
As indicated by the name, ion-selective electrodes (ISE’s) possess a high degree of selectivity. These electrodes are routinely used in clinical laboratories to determine various ion concentrations (such as calcium ion) in blood samples. In this laboratory, the electrode used is specific for chloride ion. Because specific ion electrodes measure activity and not concentration, a large amount of an inert strong electrolyte (e.g. nitrate ion) can be added to fix the ionic strength to a constant value. When the ionic strength is constant, the activity is constant and concentration can be accurately measured. In this laboratory you will fix the ionic strength with an ionic strength adjustment buffer (ISAB). You will be asked to determine the concentration of chloride in an unknown sample at the ppm level.
The chloride ion selective electrode you will use is a crystalline solid-state electrode that contains a membrane, as shown in the diagram below (Figure 1). The membrane consists of a solid salt of silver sulfide / silver chloride (Ag2S / AgCl). The membrane must be insoluble in the analyte solution and contain the analyte ion of interest. The membrane is placed at the end of a solid plastic tube. This membrane is in contact with the analyte solution during the measurement. Inside of the tube is a reference solution, which contains a known and fixed concentration of analyte (Cl-) solution. The concentration difference between this inner solution and analyte solution causes the migration of charged species across the membrane. This ion exchange process at the surface of the membrane causes a potential to develop. Since the potential of both the reference electrode and the inner reference (immersed in the standard solution) are constant, any change in measured potential is caused only by a change in potential across the membrane and is a function of the analyte chloride ion activity (or concentration).
It is also important to note that Figure 1 depicts a reference electrode external to the ISE (indicator electrode). However, a reference electrode can also be placed inside of the tube containing the ISE. This is called a “combination electrode” and is the type that you will be using in this laboratory experiment.
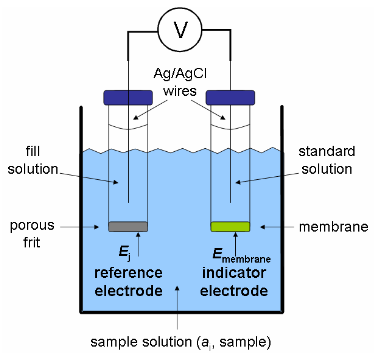
Figure 1. Schematic of a potentiometric measurement.
3. Materials and Equipment
- Sodium chloride
- Ionic strength adjustment buffer (ISAB)
- 1 L volumetric flask
- 250 mL beaker
- 250 mL volumetric flask
- 50 mL pipette
- 1 L plastic bottle
- Potentiometer
- Eppendorf micro pipette
- Magnetic stir bar
4. Safety / Special Handling Procedures
Protective eyewear must be worn at all times.
5. Experimental Method
5.1. Preparation of the standard solution
Accurately weigh pure sodium chloride to give about 2 grams of chloride ion (show calculations). Record this mass to 4 decimal places in your laboratory notebook and quantitatively transfer the NaCl with a wash bottle to a one liter volumetric flask. Dissolve, dilute to the mark with nanopure water, and mix. Calculate the concentration of chloride ion in parts per million. Transfer to a plastic bottle and label it "chloride working standard".
5.2. Preparation of the unknown
5.2.1. Obtain an unknown chloride sample and quantitatively transfer it into a 250 mL volumetric flask. Dilute to the mark with nanopure water and mix. Transfer to a plastic bottle and label appropriately.
5.2.2. Obtain the large bottle marked "ISAB" (ionic strength adjusting buffer). It contains approximately 0.2 M sodium nitrate solution. If the solution level is very low or the bottle is empty, prepare additional solution. If there is sufficient solution, go to step 5.2.3.
5.2.2.1.Weigh out pure sodium nitrate on the top loader scale. Add this to the large 2 liter bottle. Add water to mark on top shoulder of the bottle and mix. Note: This solution is not considered a standard solution.
5.2.3. You will prepare three samples of the unknown using three separate 250-mL beakers. To do so, pipette a 50.00 mL aliquot of ISAB into three different clean and dry 250 mL beakers. Then pipette 50.00 mL aliquots of the unknown solution into each beaker. These three diluted samples will be individually analyzed by the instrument.
5.3. Use of the Accumet Model 15 Specific Ion Potentiometer:

Figure 2. Accumet Model 15 Potentiometer
5.3.1. Bring the following to the instrument room:
- Beaker with the sample
- Bottle with the chloride standard
- Eppendorf micro pipette (blue top. set to deliver 1000 μL with a new plastic tip
- Wash bottle
- Kim Wipes (if none are already there)
- Empty beaker for rinsing purposes
- Magnetic stir bar
5.3.2. Remove the combination chloride/reference electrode from the storage vial and set it in the holder. Refer to Figure 2 for a picture of the potentiometer and Figure 3 for an image of the electrode.
5.3.3. The bottom of the electrode is a flat crystal surface and must not be touched with fingers (only Kim wipes). Open the fill hole at the top of the electrode. See Figure 3 below.

Figure 3. Chloride electrode (left) with the fill hole closed (middle) and open (right).
5.3.4. Rinse the electrode with nanopure water. Collect the rinses in the empty beaker and blot the electrode dry with a Kim wipe.
5.3.5. Place the sample beaker on the magnetic stirrer and carefully drop in the magnetic bar without splashing.
5.3.6. Insert the electrode so that not only the electrode but also the salt bridge is immersed in the liquid. See Figure 4 below.

Figure 4. Proper positioning of electrode in beaker.
5.3.7. Adjust the stirrer to a slow speed (note: clockwise increases the speed). Do not allow the stirrer to touch the electrode.
5.3.8. Make sure that the potentiometer is plugged in and turned on. Press the "meas/monitor" button so that the instrument is actively reading the potential of the solution (if you see a symbol in the shape of a key on the screen, it is in the locked mode and so press the meas/monitor button again to unlock the instrument). Verify that the meter reads "mV" at the bottom of the screen. The accuracy of the instrument is to the nearest tenth of a millivolt (0.1 mV). When a steady reading is produced, record the millivolt reading in your notebook.
5.3.9. Spike the sample with 1000 μL of the standard chloride solution. Again, when the instrument produces a steady reading, record the millivolt value in your notebook.
Note: For a review of proper micropipette usage see Appendix A of this procedure.
5.3.10. Repeat steps 5.3.1 through 5.3.9 for a total of three samples prepared. Rinse the electrode and dry between each sample reading (post spike addition).
5.3.11. Rinse the electrode with nanopure water and place in the storage flask. Make sure that the fill hole is closed. Refer to Figure 4 for placement.
6. Data Analysis / Calculations
Note: These calculations are quite long. It is a good idea to write them out on scrap paper, double check them, and then record the calculations neatly in your notebook.
The calculation for the standard addition method in potentiometry uses the Nernst equation, where the potential is measured in volts (what were the units you recorded??). Note that while the Nernst Equation typically involves the molarities (or, more accurately, the activities) of ions, it is perfectly acceptable to use ppm values in the following calculations:
(before the spike): \(E' = k + \dfrac{0.05916}{z} \log[\ce{Cl-}]_{unk}\)
(after the spike): \(E'' = k + \dfrac{0.05916}{z} \log ([\ce{Cl-}]'_{unk} + [\ce{Cl-}]'_{std})\) where \(\mathrm{[Cl^-]’ = DF \times [Cl^-]}\),
Note that in the second equation, the addition of the standard spike changes the volume of solution in the beaker and so both the standard and unknown concentrations are diluted from their original values. You must account for this by determining the different dilution factors (i.e., [Cl-]’ = DF x [Cl-], where DF is the dilution factor) for the standard and unknown in the second equation.
The other variables in the equations are potential (E, in volts) and charge (z) of the chloride ion. The “k” is a constant, therefore, identical in both equations. This information should allow you to manipulate the equations and solve for [Cl-]unk. Once you have determined [Cl-]unk in your unknowns diluted by the ISAB, you will need to use an aliquot factor to determine the original concentration of chloride in your unknown Perform separate calculation for each individual trial then calculate the average chloride concentration in ppm. DO NOT average the mV readings and perform the calculation on the average readings.
7. Reporting Requirements
In your conclusion, report the individual results for each trial and the average concentration (in ppm) chloride in the original unknown (the 250 mL solution) to one decimal place. Also calculate the percent relative deviation of your trials.


