Kinetics II (Worksheet)
- Page ID
- 16593
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Name: ______________________________
Section: _____________________________
Student ID#:__________________________
Work in groups on these problems. You should try to answer the questions without referring to your textbook. If you get stuck, try asking another group for help.
Q1.
It is known that compounds called chlorofluorocarbons (CFC.s) (eg. \(CFCl_3\)) will break up in the presence of ultraviolet radiation, such as found in the upper atmosphere, forming single chlorine atoms (radicals):
\[CFCl_3 \rightarrow CFCl_2 + Cl \nonumber \]
The \(Cl\) atoms then react with ozone (\(O_3\) ) as outlined in the following mechanism.
Step 1: \(Cl + O_3 \rightarrow ClO + O_2\)
Step 2: \(ClO + O \rightarrow Cl + O_2\) (single "O" atoms occur naturally in the atmosphere.)
- Write the equation for the overall reaction. (Using steps 1 and 2)
- What is the catalyst in this reaction?
- Identify an intermediate in this reaction.
- Explain how a small amount of chlorofluorocarbons can destroy a large amount of ozone.
- What breaks the bond in the \(CFCl_3\) and releases the free Cl atom?
Q2.
Given the following mechanism, answer the questions below:
Step 1: \(O_3 + NO \rightarrow NO_2 + O_2\) (slow)
Step 2: \(NO_2 + O \rightarrow NO + O_2\) (fast)
- Give the equation for the overall reaction . __________________________________
- What could the catalyst be in this mechanism? ________________________
- What is an intermediate in this mechanism? ______________________________
- Given that the uncatalyzed overall reaction is a slow exothermic reaction, draw a potential energy graph which shows the possible shape of the curve for the uncatalyzed reaction. On the same graph, show a possible curve for the catalyzed reaction.
Q3
Consider the following mechanism:
Step 1: \(H_2O_2 + I^- \rightarrow H_2O + IO^-\) (slow)
Step 2: \(H_2O_2 + IO^- \rightarrow H_2O + O_2 + I^-\) (fast)
- Give the equation for the overall reaction. ___________________________________
- What acts as a catalyst in this mechanism? _________________________________
- What acts as an intermediate in this mechanism? ____________________________
Q4.
What is meant by the rate determining step in a reaction mechanism?
Q5.
What is meant by a reaction mechanism ?
Q6.
How are reaction mechanisms determined?
Q7.
Given the following Potential Energy Diagram for a 3 step reaction, answer the questions below it :
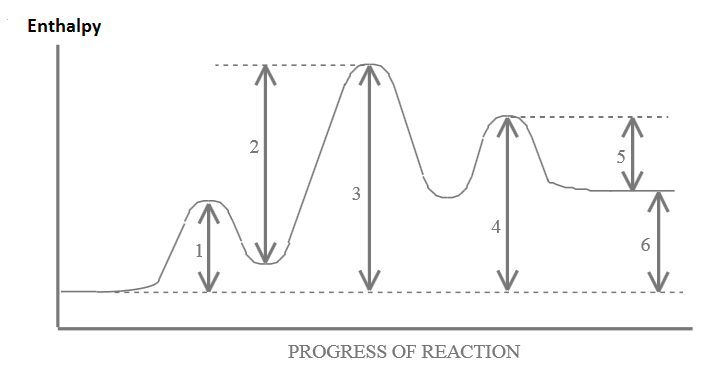
- Which arrow indicates the activation energy for the first step of the reverse reaction? ________
- Which arrow indicates the activation energy for the first step of the forward reaction? ________
- Which arrow indicates the activation energy for the second step of the forward reaction? ________
- Which arrow indicates the enthalpy change ( \(\Delta{H}\)) for the overall forward reaction? ________
- Which arrow indicates the enthalpy change ( \(\Delta{H}\)) for the overall reverse reaction? ________
- Which arrow indicates the activation energy for the overall forward reaction? ________
- Which step would be the rate determining step in the forward reaction? ________
- In a dashed line or another color sketch a possible curve that would represent the route for the uncatalyzed overall reaction. Label this on the graph.
Q8
Given the reaction:
\[4HBr + O_2 \rightarrow 2H_2O + 2Br_2 \nonumber \]
- Would you expect this reaction to take place in a single step? Why or why not?
- This reaction is thought to take place by means of the following mechanism:
Step 1: \(HBr + O_2 \rightarrow HOOBr \) (slow)
Step 2:\(HBr + HOOBr \rightarrow 2HOBr\) (fast)
Step 3: \(2HBr + 2HOBr \rightarrow 2H_2O + 2Br_2\) (fast)
- Identify the two intermediates.
- A catalyst is discovered which increases the rate of Step 3. How will this affect the rate of the overall reaction? Explain your answer.
- A catalyst is discovered which increases the rate of Step 1. How will this affect the rate of the overall reaction? Explain your answer.
- Which step has the greatest activation energy? __________________________
- How many "peaks" will the potential energy diagram for the reaction mechanism have?
- Which step is called the rate determining step in this mechanism?
- In order to have successful collisions, the colliding particles must have both the proper amount of energy and the proper _______________ (fill in blank)?
- On the set of axes below, draw the shape of the curve you might expect for the reaction in this question. The overall reaction is exothermic! Make sure you get the "bumps" the correct relative sizes.

Q9
The equation for an overall reaction is: \[I^- + OCl^- \rightarrow IO^- + Cl^- \nonumber \]
- The following is a proposed mechanism for this reaction. One of the species has been left out. Determine what that species is (in lieu of the question mark). Make sure the charge is correct if it has one!
Step 1: \(OCl^- + H_2O \rightarrow HOCl + OH^-\) (fast )
Step 2: \(I^- + HOCl \rightarrow IOH + Cl^- \) (slow )
Step 3: \(IOH + OH^- \rightarrow ? + H_2O \)(fast)
- Which species in the mechanism above acts as a catalyst ?
- Which three species in the mechanism above are intermediates? __________________
- Step ______________ is the rate determining step.
- On the set of axes below, draw the shape of the curve you might expect for the reaction in this question. The overall reaction is endothermic ! Make sure you get the "bumps" the correct relative sizes.

Q10
Given the following steps for a mechanism:
Step 1: \(Br2 \rightarrow 2Br\) (fast)
Step 2: \(Br + OCl_2 \rightarrow BrOCl + Cl\) (slow)
Step 3: \(Br + Cl \rightarrow BrCl\) (fast)
- Write the equation for the overall reaction. __________________________________________________________________
- A substance is added that decreases the activation energy for step 1. Will this speed up, slow down, or have no effect on the rate of the overall reaction? ________________Give a reason for your answer. ___________________________________________
- Is there a catalyst in this mechanism? __________. If so, what is it? ____________
- Is there an intermediate in this mechanism?________. If so, what is it? __________
- Which step is the rate determining step ? _________________________________


