Background
- Page ID
- 120146
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Wisconsin is loaded with metal sulfide minerals. Native Americans mined the southwestern region of

Figure 1. Picture of emblem on Wisconsin state flag.
The most common sulfide mineral, pyrite (FeS2) is a very hard, heavy, shiny, yellow mineral, sometimes referred to as “fool’s gold”. Chemically, it is primarily composed of iron and pairs of sulfur atoms in the form of S22– dumbbells. The crystal structure can be viewed as a face-centered cubic lattice of Fe2+ ions with 8 sulfur atoms within the unit cell. Each sulfur atom has three Fe2+ ions and one sulfur atom as nearest neighbors. (See Figure 2).1 Other ions such as arsenic can substitute for sulfur in the crystal lattice forming a “solid solution” with up to ~6% arsenic concentration, so that “pyrite” also has other potentially hazardous metals locked into its crystal structure. At higher concentrations, a different mineral known as arsenopyrite, FeAsS, exists. A number of other sulfide minerals (based on lead, gold, silver, mercury, or copper, for instance) exist naturally in the environment. Examples include galena (PbS), argentite (Ag2S), cinnabar (HgS), chalcopyrite (CuFeS2), chalcocite (Cu2S), and covellite (CuS). These metal sulfide minerals are part of a large deposit that extends through northeastern
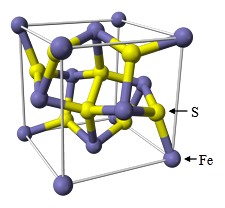
Figure 2. Structure of pyrite.1
Most of the sulfide minerals are located deep underground. However, several regions in central
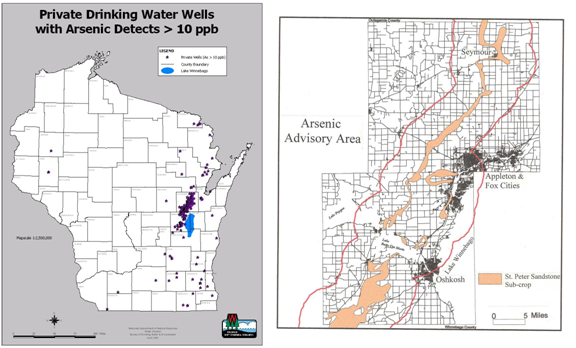
Figure 3. (a) Map showing private wells with arsenic levels greater than the 10 ppb. (b) Map showing the arsenic advisory area near
Recently in Wisconsin, public attention was drawn to the Crandon area, where there had been proposals to mine a sulfide ore deposit, primarily to obtain copper (found as copper sulfides) and zinc (found as zinc sulfide), along with trace amounts of silver and gold. The potential environmental problems associated with crushing and exposing large amounts of sulfide minerals, together with other environmental and economical concerns, led to a great deal of opposition to the mine and eventually resulted in the termination of the project. More information can be found on the Wisconsin Department of Natural Resources (
Sulfide minerals create similar environmental problems elsewhere. The most serious problem is in
The problems associated with metal sulfide minerals often go by the name of “acid mine drainage”, or “
Although the sulfide minerals are present naturally and slowly react under ambient conditions, mining activities accelerate their reactions and have the potential to create very significant, long-lasting environmental problems. Assessing the risk versus benefit of these activities is difficult in part because the underlying science is actually quite complex, making it difficult to quantitatively predict the environmental impact of these activities in the real environment. Additionally, it must be remembered that many of the raw materials must be obtained from somewhere, and mining operations not conducted in the
There exists a great deal of rhetoric regarding surrounding the environmental science of mining run-off. Opponents of mining and other activities with potential for damaging the environment often overstate the problems and point to worse-case scenarios, while mine operators/owners underestimate the likely environmental damage. What is nominally a scientific issue rapidly becomes politicized, and the public is left trying to figure out who (if anyone) to believe. As scientists, one of our roles is to provide unbiased scientific data and models that can be used to provide a rational, scientific basis for understanding and predicting the potential effects of human activities on the environment. A second, and often underutilized, responsibility is to communicate this information to the general public. Most people do not have sufficient scientific background to understand the detailed science. However, especially because environmental issues are rarely black-and-white, in order to make informed decisions the public needs to be able to discern between fact and political hyperbole.


