Recrystallization
- Page ID
- 1588
Recrystallization, also known as fractional crystallization, is a procedure for purifying an impure compound in a solvent. The method of purification is based on the principle that the solubility of most solids increases with increased temperature. This means that as temperature increases, the amount of solute that can be dissolved in a solvent increases.
Introduction
An impure compound is dissolved (the impurities must also be soluble in the solvent), to prepare a highly concentrated solution at a high temperature. The solution is cooled. Decreasing the temperature causes the solubility of the impurities in the solution and the substance being purified to decrease. The impure substance then crystallizes before the impurities- assuming that there was more impure substance than there were impurities. The impure substance will crystallize in a purer form because the impurities won't crystallize yet, therefore leaving the impurities behind in the solution. A filtration process must be used to separate the more pure crystals at this point. The procedure can be repeated. Solubility curves can be used to predict the outcome of a recrystallization procedure.
Recrystallization works best when
- the quantity of impurities is small
- the solubility curve of the desired solute rises rapidly with temperature
The slower the rate of cooling, the larger the crystals are that form. The disadvantage of recrystallization is that it takes a long time. Also, it is very important that the proper solvent is used. This can only be determined by trial and error, based on predictions and observations. The solution must be soluble at high tempratures and insoluble at low temperatures. The advantage or recrystallization is that, when carried out correctly, it is a very effective way of obtaining a pure sample of some product, or precipitate.
Procedure
These are the important steps to the recrsytallization process.
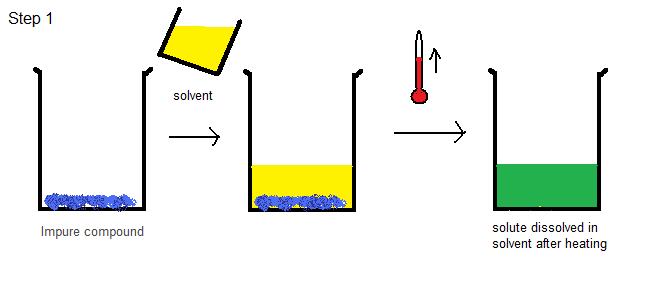
- Dissolve the solute in the solvent: Add boiling solvent to a beaker containing the impure compound. Heat the beaker and keep adding solvent until the solute is completely dissolved. See Figure 1
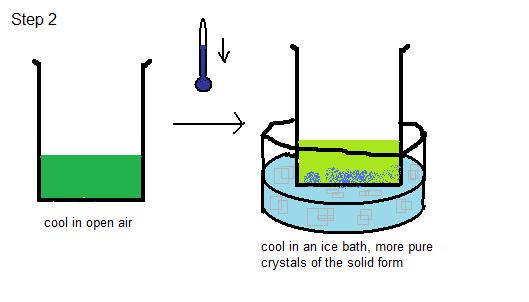
- Cool the Solution: The solution is cooled in open air first, and then cooled in an ice bath. Slow cooling often leads to purer crystals. Crystals should form on the bottom of the beaker. The process of "seeding" can be used to aid the formation of crystals- this means adding a pure crystal of the compound. The pure crystal forms a surface for the solute to crystallize upon. See Figure 2
- Obtain the crystals from the solute: The purer crystals precipitated from the solute are the desirable part of the mixture, and so they must be removed from the solvent. The process used for isolating the crystals that remain in the beaker still is called vacuum filtration. Suction is created using an aspirator, and whatever remains in the beaker is poured though a Buchner funnel. If for some reason there are no crystals visible, a gravity filtration can be performed. Activated carbon is added to the solution, the mixture is boiled, and a funnel system is used to transfer the new mixture to a new beaker of boiling solvent. Filter paper is used in the funnel to remove excess carbon. After this mixture cools slowly there should be large crystals present.
- Dry the resulting crystals: The crystals are dried by leaving them in the aspirator and then by removing them to a glass dish to wait a while longer. The purity of the crystals can be tested by performing a "melting point determination".
References
- Petrucci, Harwood, Herring, Madura. General Chemistry: Principles & Modern Applications, Ninth Ed. Upper Saddle River, NJ: Pearson Education, Inc., 2007.

