Configurational Nomenclature
- Page ID
- 1251
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Designating the Configuration of Chiral Centers
Although enantiomers may be identified by their characteristic specific rotations, the assignment of a unique configuration to each has not yet been discussed. We have referred to the mirror-image configurations of enantiomers as "right-handed" and "left-handed", but deciding which is which is not a trivial task. An early procedure assigned a D prefix to enantiomers chemically related to a right-handed reference compound and a L prefix to a similarly related left-handed group of enantiomers. Although this notation is still applied to carbohydrates and amino acids, it required chemical transformations to establish group relationships, and proved to be ambiguous in its general application. A final solution to the vexing problem of configuration assignment was devised by three European chemists: R. S. Cahn, C. K. Ingold and V. Prelog. The resulting nomenclature system is sometimes called the CIP system or the R-S system.
In the CIP system of nomenclature, each chiral center in a molecule is assigned a prefix (R or S), according to whether its configuration is right- or left-handed. No chemical reactions or interrelationship are required for this assignment. The symbol R comes from the Latin rectus for right, and S from the Latin sinister for left. The assignment of these prefixes depends on the application of two rules: The Sequence Rule and The Viewing Rule. The sequence rule is the same as that used for assigning E-Z prefixes to double bond stereoisomers. Since most of the chiral stereogenic centers we shall encounter are asymmetric carbons, all four different substituents must be ordered in this fashion.
The Sequence Rule for Assignment of Configurations to Chiral Centers
Assign sequence priorities to the four substituents by looking at the atoms attached directly to the chiral center.
1. The higher the atomic number of the immediate substituent atom, the higher the priority.
For example, H– < C– < N– < O– < Cl–. (Different isotopes of the same element are assigned a priority according to their atomic mass.)
2. If two substituents have the same immediate substituent atom,
evaluate atoms progressively further away from the chiral center until a difference is found.
For example, CH3– < C2H5– < ClCH2– < BrCH2– < CH3O–.
3. If double or triple bonded groups are encountered as substituents, they are treated as an equivalent set of single-bonded atoms.
For example, C2H5– < CH2=CH– < HC≡C–
The Viewing Rule
Once the relative priorities of the four substituents have been determined, the chiral center must be viewed from the side opposite the lowest priority group. If we number the substituent groups from 1 to 4, with 1 being the highest and 4 the lowest in priority sequence, the two enantiomeric configurations are shown in the following diagram along with a viewers eye on the side opposite substituent #4.

Remembering the geometric implication of wedge and hatched bonds, an observer (the eye) notes whether a curved arrow drawn from the # 1 position to the # 2 location and then to the # 3 position turns in a clockwise or counter-clockwise manner. If the turn is clockwise, as in the example on the right, the configuration is classified R. If it is counter-clockwise, as in the left illustration, the configuration is S. Another way of remembering the viewing rule, is to think of the asymmetric carbon as a steering wheel. The bond to the lowest priority group (# 4) is the steering column, and the other bonds are spokes on the wheel. If the wheel is turned from group # 1 toward group # 2, which in turn moves toward group # 3, this would either negotiate a right turn (R) or a left turn (S). This model is illustrated below for a right-handed turn, and the corresponding (R)-configurations of lactic acid and carvone are shown to its right. The stereogenic carbon atom is colored magenta in each case, and the sequence priorities are shown as light blue numbers. Note that if any two substituent groups on a stereogenic carbon are exchanged or switched, the configuration changes to its mirror image.
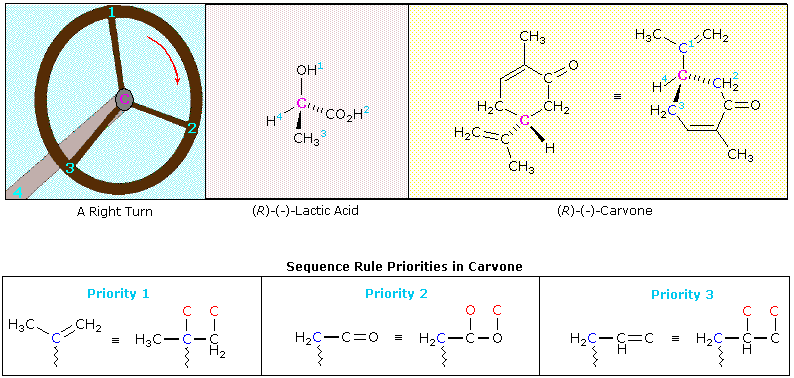
The sequence order of the substituent groups in lactic acid should be obvious, but the carvone example requires careful analysis. The hydrogen is clearly the lowest priority substituent, but the other three groups are all attached to the stereogenic carbon by bonds to carbon atoms (colored blue here). Two of the immediate substituent species are methylene groups (CH2), and the third is a doubly-bonded carbon. Rule # 3 of the sequence rules allows us to order these substituents. The carbon-carbon double bond is broken so as to give imaginary single-bonded carbon atoms (the phantom atoms are colored red in the equivalent structure). In this form the double bond assumes the priority of a 3º-alkyl group, which is greater than that of a methylene group. To establish the sequence priority of the two methylene substituents (both are part of the ring), we must move away from the chiral center until a point of difference is located. This occurs at the next carbon, which on one side is part of a carbonyl double bond (C=O), and on the other, part of a carbon-carbon double bond. Rule # 3 is again used to evaluate the two cases. The carbonyl group places two oxygens (one phantom) on the adjacent carbon atom, so this methylene side is ranked ahead of the other.
An interesting feature of the two examples shown here is that the R-configuration in both cases is levorotatory (-) in its optical activity. The mirror-image S-configurations are, of course, dextrorotatory (+). It is important to remember that there is no simple or obvious relationship between the R or S designation of a molecular configuration and the experimentally measured specific rotation of the compound it represents. In order to determine the true or "absolute" configuration of an enantiomer, as in the cases of lactic acid and carvone reported here, it is necessary either to relate the compound to a known reference structure, or to conduct a rather complex X-ray analysis on a single crystal of the sample.
| The module on the right provides examples of chiral and achiral molecules for analysis. These are displayed as three-dimensional structures which may be moved about and examined from various points of view. By using this resource the reader's understanding of configurational notation may be tested. This visualization makes use of the Jmol applet. With some browsers it may be necessary to click a button twice for action. |
|
||||||||
Configurational drawings of chiral molecules sometimes display structures in a way that does not permit an easy application of the viewing rule. In the example of carvone, shown above, the initial formula directed the lowest priority substituent (H) toward the viewer, requiring either a reorientation display or a very good sense of three-dimensional structure on the part of the reader. The Fischer projection formulas, described later, are another example of displays that challenge even experienced students. A useful mnemonic, suggested by Professor Michael Rathke, is illustrated below. Here a stereogenic tetrahedral carbon has four different substituents, designated 1, 2, 3 & 4. If we assume that these numbers represent the sequence priority of these substituents (1 > 2 > 3 > 4), then the R and S configurations are defined.
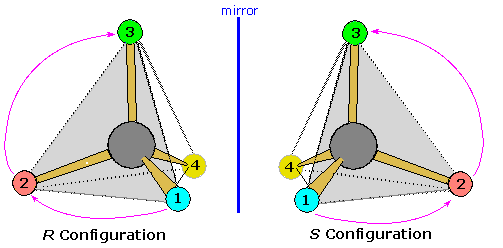
The viewing rule states that when the lowest priority substituent (4) is oriented behind the triangular face defined by the three higher priority substituents (shaded light gray here), a clockwise sequential arrangement of these substituents (1, 2 & 3) is defined as R, and a counter-clockwise sequence as S.
Now a tetrahedral structure may be viewed from any of the four triangular faces, and the symmetry of the system is such that a correct R/S assignment is made if the remote out-of plane group has an even number sequence priority (2 or 4), whereas the wrong assignment results when the out-of plane group has an odd priority (1 or 3). Once one recognizes this relationship, the viewing options are increased and a configurational assignment is more easily achieved. For an example, click on the diagram to see the 1:3:4-face, shaded light gray. oriented in front of substituent 2. Note that the R/S assignment is unchanged.
Contributors
- William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry


