Isotopes: 13C
- Page ID
- 4181
Isotopomers or isotopic isomers are isomers with isotopic atoms, having the same number of each isotope of each element but differing in their positions.
Isotopomers
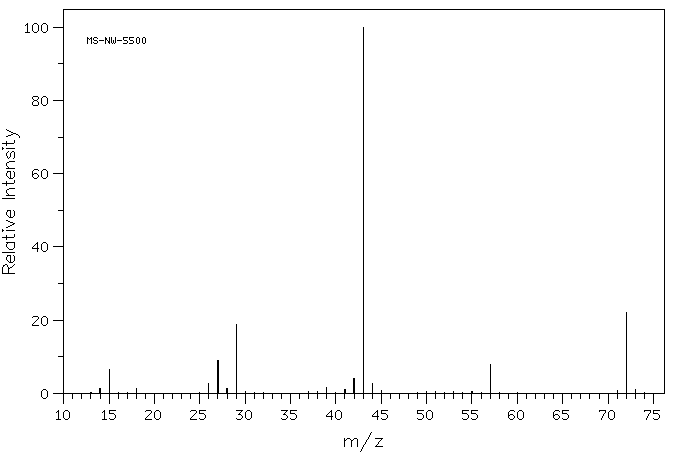
If you look closely at the mass spectrum of an organic compound, 2-butanone, you see a line at m/z 72, which corresponds to 4 carbons, an oxygen and 8 hydrogens.
- Usually, whole numbers are used for molecular weights in mass spectrometry.
- The atomic masses in the periodic table, out to 4 decimal places, are the average masses including different possible isotopes.
- Because mass spectrometry examines individual molecules, individual atomic masses are needed, not average ones. Usually that means using a whole number.
In addition, there are a number of other lines at lower values of m/z; these correspond to the masses of smaller pieces of those 2-butanone molecules that fall apart during the experiment. We won't look too closely at how those arise until we get to radical reactions later in the course. However, we will look at some factors that make cations stable later in this chapter.
If you look closely at the mass spectrum of 2-butanone, you'll also see another little peak at m/z 73. This is referred to as the M+1 peak (one greater than the molecular ion), and it arises because of 13C. This compound is referred to as an isotopomer; that means the same compound with a different isotope.
- 12C is about 99% abundant; 99% of carbon atoms have a mass of 12 amu.
- 13C is about 1% abundant; 1% of carbon atoms have a mass of 13 amu.
- Compounds that contain a 13C atom have a mass one larger than expected.
The chance that a molecule in a sample contains a 13C atom is related to the number of carbons present. If there is just one carbon atom in the molecule, it has a 1% chance of being a 13C. That means the M+1 peak would be only 1/100th as tall as M+, the peak for the molecular ion.
- The M+1 peak from a 13C atom is very small.
- The more carbons there are in a molecule, the bigger the M+1 peak.
- If there are 10 carbon atoms in the molecule, there is a 10% chance of a 13C atom being present. The M+1 peak is about 1/10th the size of the M+ peak.
- If there are 100 carbons in the molecule, there is a very good chance that a 13C atom is present. At that point, the M+1 peak is actually much larger than the M+. peak


