7.9: Carbocation Structure and Stability
- Page ID
- 239153
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Objectives
After completing this section, you should be able to
- describe the geometry of a given carbocation.
- arrange a given series of carbocations in order of increasing or decreasing stability.
- explain the relative stability of methyl, primary, secondary and tertiary carbocations in terms of hyperconjugation and inductive effects.
Study Notes
Although hyperconjugation can be used to explain the relative stabilities of carbocations, this explanation is certainly not the only one, and is by no means universally accepted. A more common explanation, involving the concept of an inductive effect, is given below.
It is a general principle in chemistry that the more a charge is dispersed, the more stable is the species carrying the charge. Put simply, a species in which a positive charge is shared between two atoms would be more stable than a similar species in which the charge is borne wholly by a single atom. In a tertiary carbocation, the positively charged carbon atom attracts the bonding electrons in the three carbon-carbon sigma (σ) bonds, and thus creates slight positive charges on the carbon atoms of the three surrounding alkyl groups (and, indeed, on the hydrogen atoms attached to them). Chemists sometimes use an arrow to represent this inductive release:
Note: These diagrams do not reflect the geometry of the carbocation. The overall charge on the carbocation remains unchanged, but some of the charge is now carried by the alkyl groups attached to the central carbon atom; that is, the charge has been dispersed.
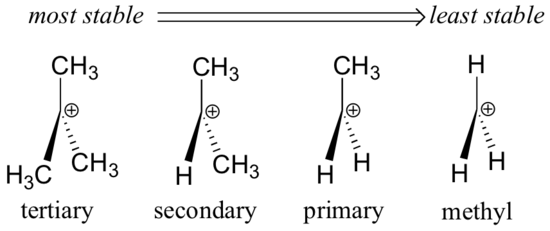
In the tertiary carbocation shown above, the three alkyl groups help to stabilize the positive charge. In a secondary carbocation, only two alkyl groups would be available for this purpose, while a primary carbocation has only one alkyl group available. Thus the observed order of stability for carbocations is as follows:
tertiary > secondary > primary > methyl.
The next step in understanding why Markovnikov's rule is often followed in electrophilic additions, involves understanding the structure and stability of the carboncation intermediate formed during the mechanism.
Carbocation Structure
Carbocations typically have three substituents which makes the carbon sp2 hybridized and gives the overall molecule a trigonal planar geometry. The carboncation's substituents are all in the same plane and have a bond angle of 120o between them. The carbon atom in the carbocation is electron deficient; it only has six valence electrons which are used to form three sigma covalent bonds with the substituents. The carbocation carbon has an unoccupied p orbital which is perpendicular to the plane created by the substituents. The p orbital can easily accept electron pairs during reactions making carbocations excellent Lewis acids.

Stability of Carbocation Intermediates
By being a reactive intermediate of the electrophilic addition mechanism, the stability of a carbocation has a direct effect on the reaction.
The critical question now becomes, what stabilizes a carbocation?
A positively charged species such as a carbocation is very electron-poor, and thus anything which donates electron density will help to stabilize it. Conversely, a carbocation will be destabilized by an electron withdrawing group.
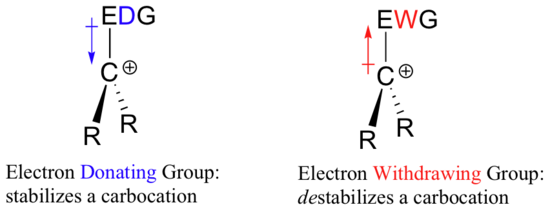
Extensive experimental evidence has shown that a carbocation becomes more stable as the number of alkyl substituents increases. Carbocations can be given a designation based on the number of alkyl groups attached to the carbocation carbon. Three alkyl groups is called a tertiary (3o) carbocation, 2 alkyl groups is called secondary (2o), and 1 alkyl group is called primary (1o). No alkyl groups are attached (3 hydrogen substituents) is called a methyl carbocation.
The overall order of stability is as follows:
Alkyl groups stabilized carbocations for two reasons. The first is through inductive effects. As discussed in Section 2-1, inductive effects occur when the electrons in covalent bonds are shifted towards an nearby atom with a higher electronegativity. In this case, the positively charged carbocation draws in electron density from the surrounding stubstituents thereby gaining stabilization by slightly reducing its positive charge. Alkyl groups are more effective at inductively donating electron density than a hydrogen because they are larger, more polarizable, and contain more bonding electrons. As more alkyl groups are attached to the carbocation more inductive electron donation occurs and the carbocation becomes more stable.
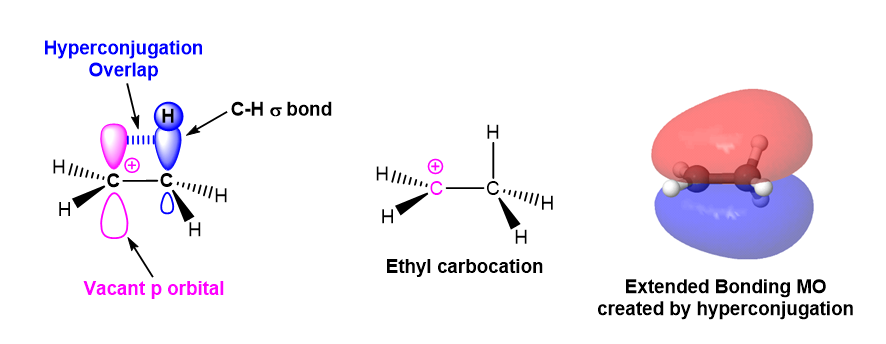
The second reason alkyl groups stabilize carbocations is through hyperconjugation. As previously discussed in Section 7.6, hyperconjugation is an electron donation that occurs from the parallel overlap of p orbitals with adjacent hybridized orbitals participating in sigma bonds. This electron donation serves to stabilize the carbocation. As the number of alkyl substituents increases, the number of sigma bonds available for hyperconjugation increases, and the carbocation tends to become more stabilized.
In the example of ethyl carbocation shown below, the p orbital from a sp2 hybridized carbocation carbon involved interacts with a sp3 hybridized orbital participating in an adjacent C-H sigma bond. Electron density from the C-H sigma bond is donated into carbocation's p orbtial providing stabilization.
The molecular orbital of the ethyl carbocation shows the interaction of electrons in methyl group's C-H sigma bonds with the adjacent empty p orbital from the carbocation. The interaction creates a bonding molecular orbital which extends over the three atom chain (C-C-H) involved in hyperconjugation. The expanded molecular orbital helps to stabilize the carbocation.
It is not accurate to say, however, that carbocations with higher substitution are always more stable than those with less substitution. Just as electron-donating groups can stabilize a carbocation, electron-withdrawing groups act to destabilize carbocations. Carbonyl groups are electron-withdrawing by inductive effects, due to the polarity of the C=O double bond. It is possible to demonstrate in the laboratory that carbocation A below is more stable than carbocation B, even though A is a primary carbocation and B is secondary.
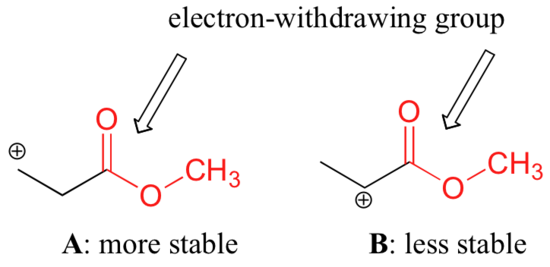
The difference in stability can be explained by considering the electron-withdrawing inductive effect of the ester carbonyl. Recall that inductive effects - whether electron-withdrawing or donating - are relayed through covalent bonds and that the strength of the effect decreases rapidly as the number of intermediary bonds increases. In other words, the effect decreases with distance. In species B the positive charge is closer to the carbonyl group, thus the destabilizing electron-withdrawing effect is stronger than it is in species A.
Stabilization of a carbocation can also occur through resonance effects, and as we have already discussed in Section 2.4, resonance effects as a rule are more powerful than inductive effects. Consider the simple case of a benzylic carbocation:
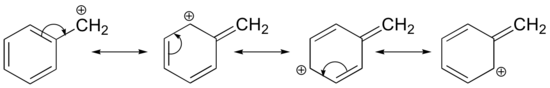
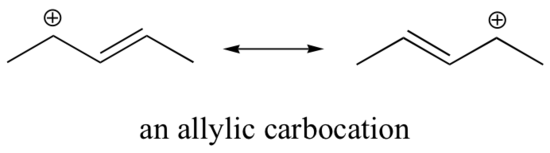
This carbocation is comparatively stable. In this case, electron donation is a resonance effect. Three additional resonance structures can be drawn for this carbocation in which the positive charge is located on one of three aromatic carbons. The positive charge is not isolated on the benzylic carbon, rather it is delocalized around the aromatic structure: this delocalization of charge results in significant stabilization. As a result, benzylic and allylic carbocations (where the positively charged carbon is conjugated to one or more non-aromatic double bonds) are significantly more stable than even tertiary alkyl carbocations.
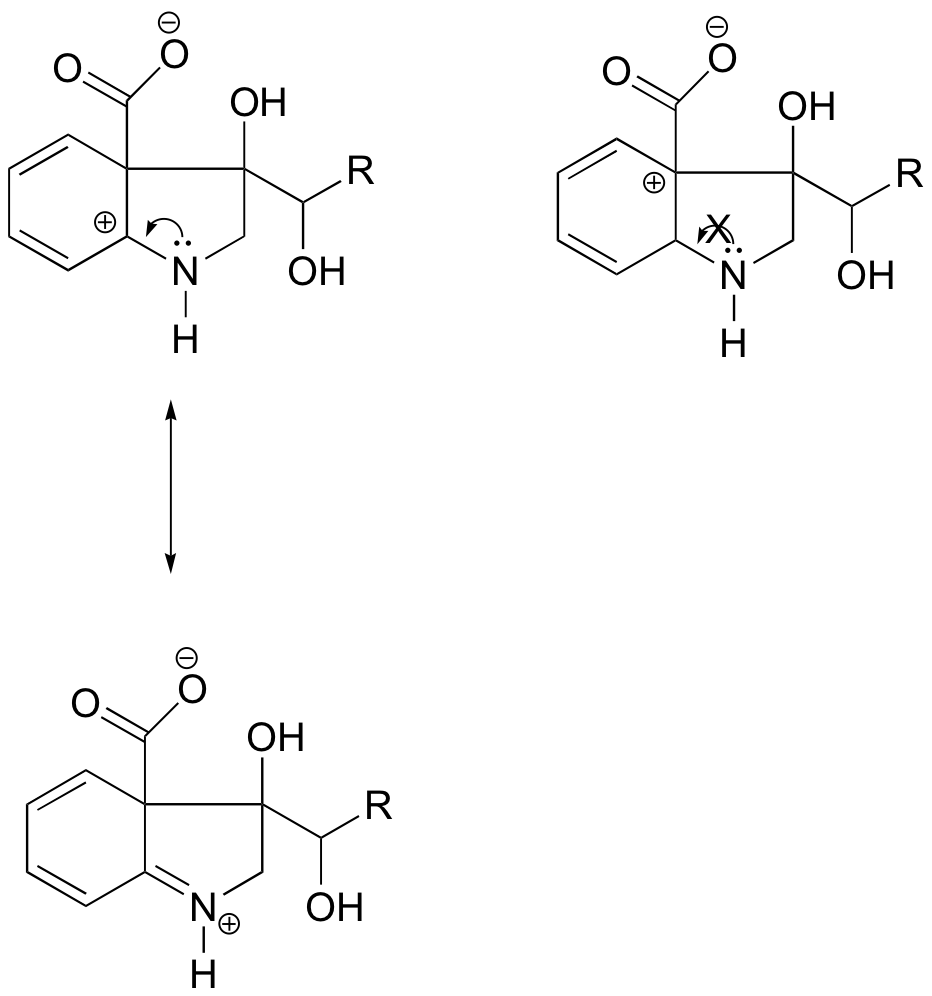
Because heteroatoms such as oxygen and nitrogen are more electronegative than carbon, you might expect that they would by definition be electron withdrawing groups that destabilize carbocations. In fact, the opposite is often true: if the oxygen or nitrogen atom is in the correct position, the overall effect is carbocation stabilization. This is due to the fact that these atoms can donate electron pairs by resonance to create a structure which obeys the octet rule. Carbocations are highly unstable because they are electron deficient and violate the octet rule. By filling carbon's octet, adjacent heteroatoms can greatly stabilize carbocations.
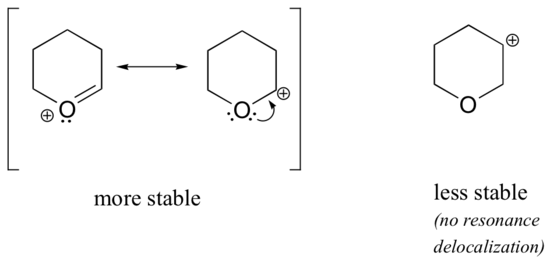
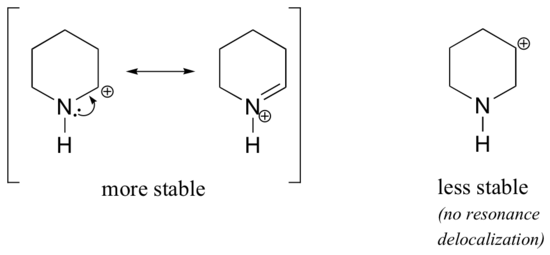
As seen in the structure above, the more stable carbocations are directly adjacent to heteroatoms which can donate lone pair electrons. This puts the positive charge on the heteroatom and give the carbon a full octet. In the less stable carbocations the positively-charged carbon is more than one bond away from the heteroatom, and thus no resonance effects are possible. In fact, in these carbocation species the heteroatoms actually destabilize the positive charge, because they are electron withdrawing by induction.
Example 7.9.1
In which of the structures below is the carbocation expected to be more stable? Explain.
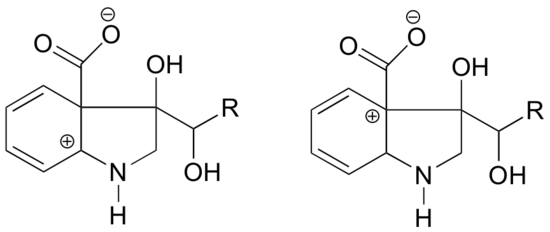
- Answer
-
In the carbocation on the left, the positive charge is located in a position relative to the nitrogen such that the lone pair of electrons on the nitrogen can be donated to fill the empty orbital. This is not possible for the carbocation species on the right.
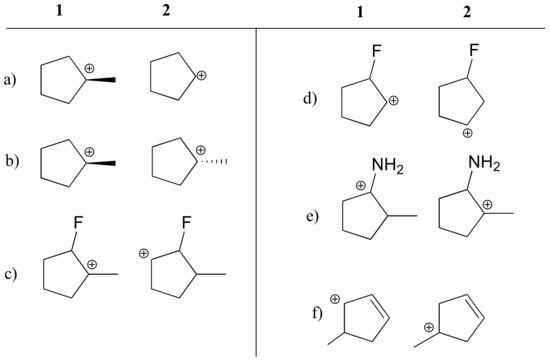
Exercise \(\PageIndex{1}\)
1) State which carbocation in each pair below is more stable, or if they are expected to be approximately equal. Explain your reasoning.
2) Draw the carbocation intermediates that are seen in the mechanism of the following reactions:
- Answer
-
1)
a) 1 (tertiary vs. secondary carbocation)
b) equal
c) 1 (tertiary vs. secondary carbocation)
d) 2 (positive charge is further from electron-withdrawing fluorine)
e) 1 (lone pair on nitrogen can donate electrons by resonance)
f) 1 (allylic carbocation – positive charge can be delocalized to a second carbon)
2)

Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)
Organic Chemistry With a Biological Emphasis by Tim Soderberg (University of Minnesota, Morris)

