1.2: Standard Potentials
- Last updated
- Save as PDF
- Page ID
- 13314
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Learning Objectives
- To use redox potentials to predict whether a reaction is spontaneous.
- To balance redox reactions using half-reactions.
In a galvanic cell, current is produced when electrons flow externally through the circuit from the anode to the cathode because of a difference in potential energy between the two electrodes in the electrochemical cell. In the Zn/Cu system, the valence electrons in zinc have a substantially higher potential energy than the valence electrons in copper because of shielding of the s electrons of zinc by the electrons in filled d orbitals. Hence electrons flow spontaneously from zinc to copper(II) ions, forming zinc(II) ions and metallic copper. Just like water flowing spontaneously downhill, which can be made to do work by forcing a waterwheel, the flow of electrons from a higher potential energy to a lower one can also be harnessed to perform work.
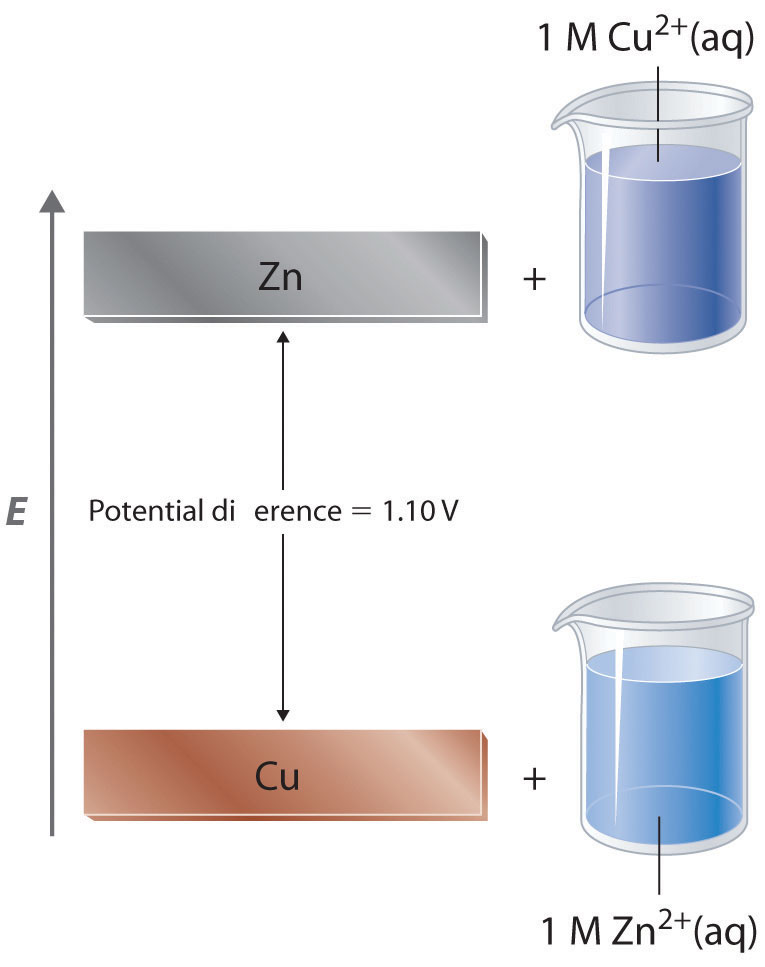
Because the potential energy of valence electrons differs greatly from one substance to another, the voltage of a galvanic cell depends partly on the identity of the reacting substances. If we construct a galvanic cell similar to the one in part (a) in Figure 19.3 but instead of copper use a strip of cobalt metal and 1 M Co2+ in the cathode compartment, the measured voltage is not 1.10 V but 0.51 V. Thus we can conclude that the difference in potential energy between the valence electrons of cobalt and zinc is less than the difference between the valence electrons of copper and zinc by 0.59 V.
The measured potential of a cell also depends strongly on the concentrations of the reacting species and the temperature of the system. To develop a scale of relative potentials that will allow us to predict the direction of an electrochemical reaction and the magnitude of the driving force for the reaction, the potentials for oxidations and reductions of different substances must be measured under comparable conditions. To do this, chemists use the standard cell potential (E°cell), defined as the potential of a cell measured under standard conditions—that is, with all species in their standard states (1 M for solutions,Concentrated solutions of salts (about 1 M) generally do not exhibit ideal behavior, and the actual standard state corresponds to an activity of 1 rather than a concentration of 1 M. Corrections for nonideal behavior are important for precise quantitative work but not for the more qualitative approach that we are taking here. 1 atm for gases, pure solids or pure liquids for other substances) and at a fixed temperature, usually 25°C.
Note
Measured redox potentials depend on the potential energy of valence electrons, the concentrations of the species in the reaction, and the temperature of the system.
Measuring Standard Electrode Potentials
It is physically impossible to measure the potential of a single electrode: only the difference between the potentials of two electrodes can be measured. (This is analogous to measuring absolute enthalpies or free energies. Recall that only differences in enthalpy and free energy can be measured.) We can, however, compare the standard cell potentials for two different galvanic cells that have one kind of electrode in common. This allows us to measure the potential difference between two dissimilar electrodes. For example, the measured standard cell potential (E°) for the Zn/Cu system is 1.10 V, whereas E° for the corresponding Zn/Co system is 0.51 V. This implies that the potential difference between the Co and Cu electrodes is 1.10 V − 0.51 V = 0.59 V. In fact, that is exactly the potential measured under standard conditions if a cell is constructed with the following cell diagram:
\[Co_{(s)} ∣ Co^{2+}(aq, 1 M)∥Cu^{2+}(aq, 1 M) ∣ Cu (s)\;\;\; E°=0.59\; V \label{19.9}\]
This cell diagram corresponds to the oxidation of a cobalt anode and the reduction of Cu2+ in solution at the copper cathode.
All tabulated values of standard electrode potentials by convention are listed for a reaction written as a reduction, not as an oxidation, to be able to compare standard potentials for different substances (Table P1). The standard cell potential (E°cell) is therefore the difference between the tabulated reduction potentials of the two half-reactions, not their sum:
\[E°_{cell} = E°_{cathode} − E°_{anode} \label{19.10}\]
In contrast, recall that half-reactions are written to show the reduction and oxidation reactions that actually occur in the cell, so the overall cell reaction is written as the sum of the two half-reactions. According to Equation \(\ref{19.10}\), when we know the standard potential for any single half-reaction, we can obtain the value of the standard potential of many other half-reactions by measuring the standard potential of the corresponding cell.
Note
The overall cell reaction is the sum of the two half-reactions, but the cell potential is the difference between the reduction potentials:
\[E°_{cell} = E°_{cathode} − E°_{anode}\]
Although it is impossible to measure the potential of any electrode directly, we can choose a reference electrode whose potential is defined as 0 V under standard conditions. The standard hydrogen electrode (SHE) is universally used for this purpose and is assigned a standard potential of 0 V. It consists of a strip of platinum wire in contact with an aqueous solution containing 1 M H+. The [H+] in solution is in equilibrium with H2 gas at a pressure of 1 atm at the Pt-solution interface (Figure \(\PageIndex{2}\)). Protons are reduced or hydrogen molecules are oxidized at the Pt surface according to the following equation:
\[2H^+_{(aq)}+2e^− \rightleftharpoons H_{2(g)} \label{19.11}\]
One especially attractive feature of the SHE is that the Pt metal electrode is not consumed during the reaction.
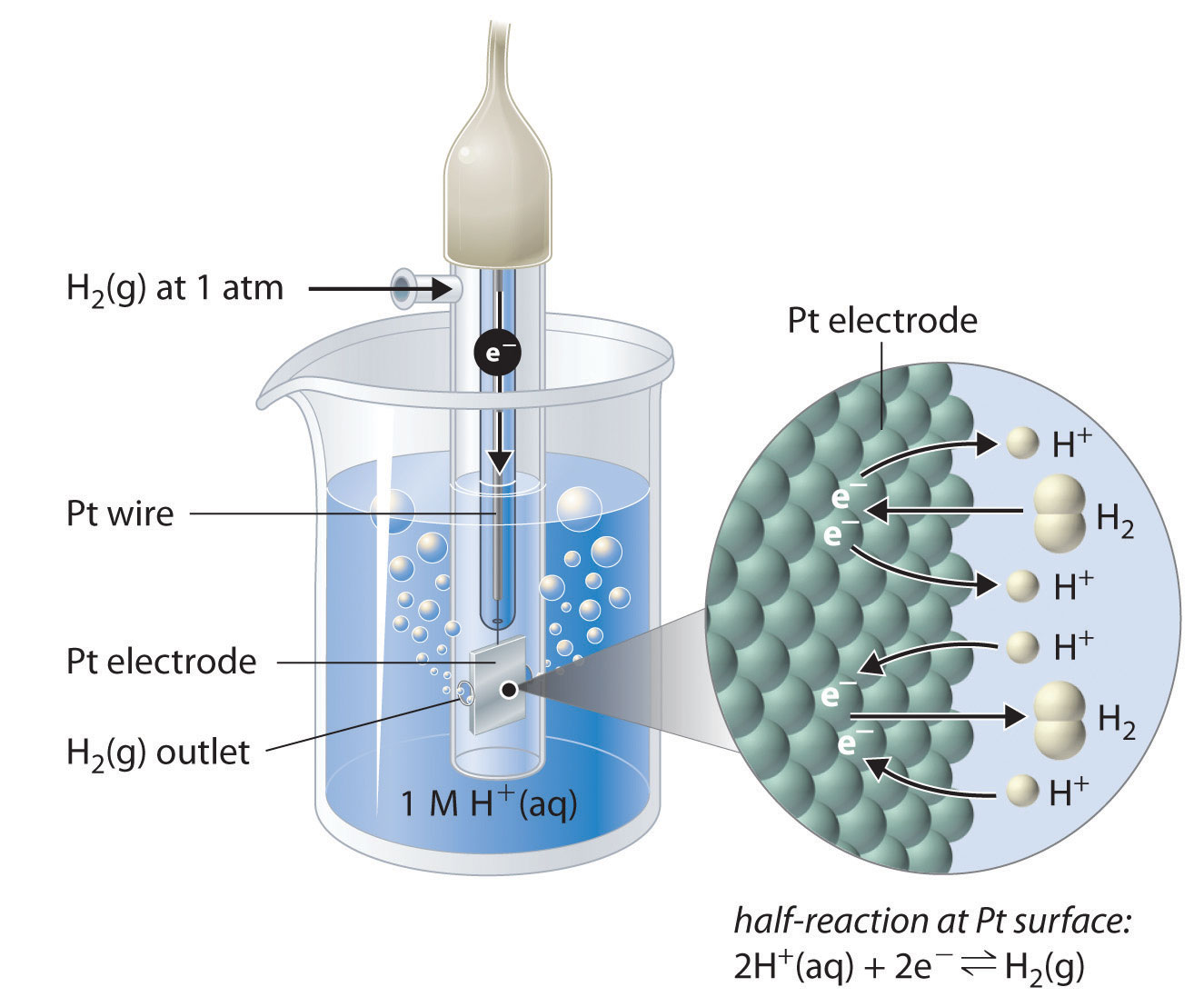
Figure \(\PageIndex{3}\) shows a galvanic cell that consists of a SHE in one beaker and a Zn strip in another beaker containing a solution of Zn2+ ions. When the circuit is closed, the voltmeter indicates a potential of 0.76 V. The zinc electrode begins to dissolve to form Zn2+, and H+ ions are reduced to H2 in the other compartment. Thus the hydrogen electrode is the cathode, and the zinc electrode is the anode. The diagram for this galvanic cell is as follows:
\[Zn_{(s)}∣Zn^{2+}_{(aq)}∥H^+(aq, 1 M)∣H_2(g, 1 atm)∣Pt_{(s)} \label{19.12}\]
The half-reactions that actually occur in the cell and their corresponding electrode potentials are as follows:
- cathode: \[2H^+_{(aq)} + 2e^− \rightarrow H_{2(g)}\;\;\; E°_{cathode}=0 V \label{19.13}\]
- anode: \[Zn_{(s)} \rightarrow Zn^{2+}_{(aq)}+2e^−\;\;\; E°_{anode}=−0.76\; V \label{19.14}\]
- overall: \[Zn_{(s)}+2H^+_{(aq)} \rightarrow Zn^{2+}_{(aq)}+H_{2(g)} \label{19.15}\]
\[E°_{cell}=E°_{cathode}−E°_{anode}=0.76\; V\]
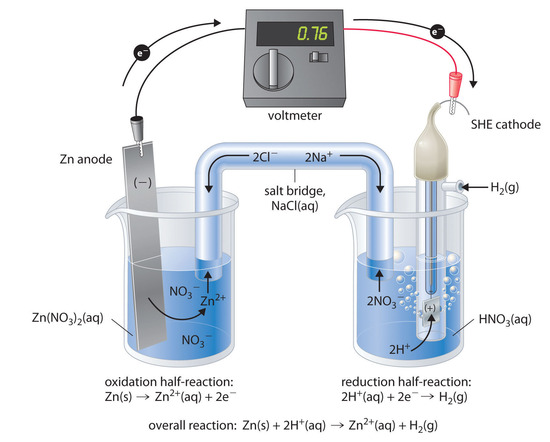
Although the reaction at the anode is an oxidation, by convention its tabulated E° value is reported as a reduction potential. The potential of a half-reaction measured against the SHE under standard conditions is called the standard electrode potential for that half-reaction.In this example, the standard reduction potential for Zn2+(aq) + 2e− → Zn(s) is −0.76 V, which means that the standard electrode potential for the reaction that occurs at the anode, the oxidation of Zn to Zn2+, often called the Zn/Zn2+ redox couple, or the Zn/Zn2+ couple, is −(−0.76 V) = 0.76 V. We must therefore subtract E°anode from E°cathode to obtain E°cell: 0 − (−0.76 V) = 0.76 V.
Because electrical potential is the energy needed to move a charged particle in an electric field, standard electrode potentials for half-reactions are intensive properties and do not depend on the amount of substance involved. Consequently, E° values are independent of the stoichiometric coefficients for the half-reaction, and, most important, the coefficients used to produce a balanced overall reaction do not affect the value of the cell potential.
Note
E° values do NOT depend on the stoichiometric coefficients for a half-reaction, because it is an intensive property.
Standard Electrode Potentials
To measure the potential of the Cu/Cu2+ couple, we can construct a galvanic cell analogous to the one shown in Figure \(\PageIndex{3}\) but containing a Cu/Cu2+ couple in the sample compartment instead of Zn/Zn2+. When we close the circuit this time, the measured potential for the cell is negative (−0.34 V) rather than positive. The negative value of E°cell indicates that the direction of spontaneous electron flow is the opposite of that for the Zn/Zn2+ couple. Hence the reactions that occur spontaneously, indicated by a positive E°cell, are the reduction of Cu2+ to Cu at the copper electrode. The copper electrode gains mass as the reaction proceeds, and H2 is oxidized to H+ at the platinum electrode. In this cell, the copper strip is the cathode, and the hydrogen electrode is the anode. The cell diagram therefore is written with the SHE on the left and the Cu2+/Cu couple on the right:
\[Pt_{(s)}∣H_2(g, 1 atm)∣H^+(aq, 1\; M)∥Cu^{2+}(aq, 1 M)∣Cu_{(s)} \label{19.16}\]
The half-cell reactions and potentials of the spontaneous reaction are as follows:
- Cathode: \[Cu^{2+}{(aq)} + 2e^− \rightarrow Cu_{(g)}\;\;\; E°_{cathode} = 0.34\; V \label{19.17}\]
- Anode: \[H_{2(g)} \rightarrow 2H^+_{(aq)} + 2e^−\;\;\; E°_{anode} = 0\; V \label{19.18}\]
- Overall: \[H_{2(g)} + Cu^{2+}_{(aq)} \rightarrow 2H^+_{(aq)} + Cu_{(s)} \label{19.19}\]
\[E°_{cell} = E°_{cathode}− E°_{anode} = 0.34\; V\]
Thus the standard electrode potential for the Cu2+/Cu couple is 0.34 V.
Electrode Potentials and ECell: https://youtu.be/zeeAXleT1c0
Balancing Redox Reactions Using the Half-Reaction Method
Previously, we described a method for balancing redox reactions using oxidation numbers. Oxidation numbers were assigned to each atom in a redox reaction to identify any changes in the oxidation states. Here we present an alternative approach to balancing redox reactions, the half-reaction method, in which the overall redox reaction is divided into an oxidation half-reaction and a reduction half-reaction, each balanced for mass and charge. This method more closely reflects the events that take place in an electrochemical cell, where the two half-reactions may be physically separated from each other.
We can illustrate how to balance a redox reaction using half-reactions with the reaction that occurs when Drano, a commercial solid drain cleaner, is poured into a clogged drain. Drano contains a mixture of sodium hydroxide and powdered aluminum, which in solution reacts to produce hydrogen gas:
\[Al_{(s)} + OH^−_{(aq)} \rightarrow Al(OH)^−_{4(aq)} + H_{2(g)} \label{19.20}\]
In this reaction, \(Al_{(s)}\) is oxidized to Al3+, and H+ in water is reduced to H2 gas, which bubbles through the solution, agitating it and breaking up the clogs.
The overall redox reaction is composed of a reduction half-reaction and an oxidation half-reaction. From the standard electrode potentials listed Table P1, we find the corresponding half-reactions that describe the reduction of H+ ions in water to H2and the oxidation of Al to Al3+ in basic solution:
- reduction: \[2H_2O_{(l)} + 2e^− \rightarrow 2OH^−_{(aq)} + H_{2(g)} \label{19.21}\]
- oxidation: \[Al_{(s)} + 4OH^−_{(aq)} \rightarrow Al(OH)^−_{4(aq)} + 3e^− \label{19.22}\]
The half-reactions chosen must exactly reflect the reaction conditions, such as the basic conditions shown here. Moreover, the physical states of the reactants and the products must be identical to those given in the overall reaction, whether gaseous, liquid, solid, or in solution.
In Equation \(\ref{19.21}\), two H+ ions gain one electron each in the reduction; in Equation \(\ref{19.22}\), the aluminum atom loses three electrons in the oxidation. The charges are balanced by multiplying the reduction half-reaction (Equation \(\ref{19.21}\)) by 3 and the oxidation half-reaction (Equation \(\ref{19.22}\)) by 2 to give the same number of electrons in both half-reactions:
- Adding the two half-reactions,
\[6H_2O_{(l)} + 2Al_{(s)} + 8OH^−_{(aq)} \rightarrow 2Al(OH)^−{4(aq)} + 3H_{2(g)} + 6OH^−_{(aq)} \label{19.25}\]
Simplifying by canceling substances that appear on both sides of the equation,
\[6H_2O_{(l)} + 2Al_{(s)} + 2OH^−_{(aq)} \rightarrow 2Al(OH)^−_{4(aq)} + 3H_{2(g)} \label{19.26}\]
We have a −2 charge on the left side of the equation and a −2 charge on the right side. Thus the charges are balanced, but we must also check that atoms are balanced:
\[2Al + 8O + 14H = 2Al + 8O + 14H \label{19.27}\]
The atoms also balance, so Equation \(\ref{19.26}\) is a balanced chemical equation for the redox reaction depicted in Equation \(\ref{19.20}\).
Note
The half-reaction method requires that half-reactions exactly reflect reaction conditions, and the physical states of the reactants and the products must be identical to those in the overall reaction.
We can also balance a redox reaction by first balancing the atoms in each half-reaction and then balancing the charges. With this alternative method, we do not need to use the half-reactions listed in Table P1 but instead focus on the atoms whose oxidation states change, as illustrated in the following steps:
Step 1: Write the reduction half-reaction and the oxidation half-reaction.
For the reaction shown in Equation \(\ref{19.20}\), hydrogen is reduced from H+ in OH− to H2, and aluminum is oxidized from Al° to Al3+:
- Step 2: Balance the atoms by balancing elements other than O and H. Then balance O atoms by adding H2O and balance H atoms by adding H+.
Elements other than O and H in the previous two equations are balanced as written, so we proceed with balancing the O atoms. We can do this by adding water to the appropriate side of each half-reaction:
- Balancing H atoms by adding H+, we obtain the following:
- We have now balanced the atoms in each half-reaction, but the charges are not balanced.
Step 3: Balance the charges in each half-reaction by adding electrons.
Two electrons are gained in the reduction of H+ ions to H2, and three electrons are lost during the oxidation of Al° to Al3+:
- Step 4: Multiply the reductive and oxidative half-reactions by appropriate integers to obtain the same number of electrons in both half-reactions.
In this case, we multiply Equation \(\ref{19.34}\) (the reductive half-reaction) by 3 and Equation \(\ref{19.35}\) (the oxidative half-reaction) by 2 to obtain the same number of electrons in both half-reactions:
- Step 5: Add the two half-reactions and cancel substances that appear on both sides of the equation.
Adding and, in this case, canceling 8H+, 3H2O, and 6e−,
\[2Al_{(s)} + 5H_2O_{(l)} + 3OH^−_{(aq)} + H^+_{(aq)} \rightarrow 2Al(OH)^−_{4(aq)} + 3H_{2(g)} \label{19.38}\]
We have three OH− and one H+ on the left side. Neutralizing the H+ gives us a total of 5H2O + H2O = 6H2O and leaves 2OH− on the left side:
\[2Al_{(s)} + 6H_2O_{(l)} + 2OH^−_{(aq)} \rightarrow 2Al(OH)^−_{4(aq)} + 3H_{2(g)} \label{19.39}\]
Step 6: Check to make sure that all atoms and charges are balanced.
Equation \(\ref{19.39}\) is identical to Equation \(\ref{19.26}\), obtained using the first method, so the charges and numbers of atoms on each side of the equation balance.
Example \(\PageIndex{1}\)
In acidic solution, the redox reaction of dichromate ion (\(Cr_2O_7^{2−}\)) and iodide (\(I^−\)) can be monitored visually. The yellow dichromate solution reacts with the colorless iodide solution to produce a solution that is deep amber due to the presence of a green \(Cr^{3+}_{(aq)}\) complex and brown I2(aq) ions (Figure \(\PageIndex{4}\)):
\[Cr_2O^{2−}_{7(aq)} + I^−_{(aq)} \rightarrow Cr^{3+}_{(aq)} + I_{2(aq)}\]
Balance this equation using half-reactions.
Given: redox reaction and Table P1
Asked for: balanced chemical equation using half-reactions
Strategy:
Follow the steps to balance the redox reaction using the half-reaction method.
Solution
From the standard electrode potentials listed in Table P1 we find the half-reactions corresponding to the overall reaction:
- reduction: \[Cr_2O^{2−}_{7(aq)} + 14H^+_{(aq)} + 6e^− \rightarrow 2Cr^{3+}(_{(aq)} + 7H_2O_{(l)}\]
- oxidation: \[2I^−_{(aq)} \rightarrow I_{2(aq)} + 2e^−\]
Balancing the number of electrons by multiplying the oxidation reaction by 3,
- oxidation: \[6I^−_{(aq)} \rightarrow 3I_{2(aq)} + 6e^−\]
Adding the two half-reactions and canceling electrons,
\[Cr_2O^{2−}_{7(aq)} + 14H^+_{(aq)} + 6I^−_{(aq)} \rightarrow 2Cr^{3+}_{(aq)} + 7H_2O_{(l)} + 3I_{2(aq)}\]
We must now check to make sure the charges and atoms on each side of the equation balance:
(−2) + 14 + (−6) = +6+6 = +62Cr + 7O+ 14H+ 6I = 2Cr + 7O + 14H + 6IThe charges and atoms balance, so our equation is balanced.
We can also use the alternative procedure, which does not require the half-reactions listed in Table P1.
Step 1: Chromium is reduced from \(Cr^{6+}\) in \(Cr_2O_7^{2−}\) to \(Cr^{3+}\), and \(I^−\) ions are oxidized to \(I_2\). Dividing the reaction into two half-reactions,
- reduction: \[Cr_2O^{2−}_{7(aq)} \rightarrow Cr^{3+}_{(aq)}\]
- oxidation: \[I^−_{(aq)} \rightarrow I_{2(aq)}\]
Step 2: Balancing the atoms other than oxygen and hydrogen,
- reduction: \[Cr_2O^{2−}_{7(aq)} \rightarrow 2Cr^{3+}_{(aq)}\]
- oxidation: \[2I^−_{(aq)} \rightarrow I_{2(aq)}\]
We now balance the O atoms by adding H2O—in this case, to the right side of the reduction half-reaction. Because the oxidation half-reaction does not contain oxygen, it can be ignored in this step.
- reduction: \[Cr_2O^{2−}_{7(aq)} \rightarrow 2Cr^{3+}_{(aq)} + 7H_2O_{(l)}\]
Next we balance the H atoms by adding H+ to the left side of the reduction half-reaction. Again, we can ignore the oxidation half-reaction.
- reduction: \[Cr_2O^{2−}_{7(aq)} + 14H^+_{(aq)} \rightarrow 2Cr^{3+}_{(aq)} + 7H_2O_{(l)}\]
Step 3: We must now add electrons to balance the charges. The reduction half-reaction (2Cr+6 to 2Cr+3) has a +12 charge on the left and a +6 charge on the right, so six electrons are needed to balance the charge. The oxidation half-reaction (2I− to I2) has a −2 charge on the left side and a 0 charge on the right, so it needs two electrons to balance the charge:
- reduction: Cr2O72−(aq) + 14H+(aq) + 6e− → 2Cr3+(aq) + 7H2O(l)
- oxidation: 2I−(aq) → I2(aq) + 2e−
Step 4: To have the same number of electrons in both half-reactions, we must multiply the oxidation half-reaction by 3:
- oxidation: 6I−(aq) → 3I2(s) + 6e−
Step 5: Adding the two half-reactions and canceling substances that appear in both reactions,
- Cr2O72−(aq) + 14H+(aq) + 6I−(aq) → 2Cr3+(aq) + 7H2O(l) + 3I2(aq)
Step 6: This is the same equation we obtained using the first method. Thus the charges and atoms on each side of the equation balance.
Exercise \(\PageIndex{1}\)
Copper is found as the mineral covellite (\(CuS\)). The first step in extracting the copper is to dissolve the mineral in nitric acid (\(HNO_3\)), which oxidizes sulfide to sulfate and reduces nitric acid to \(NO\):
\[CuS_{(s)} + HNO_{3(aq)} \rightarrow NO_{(g)} + CuSO_{4(aq)}\]
Balance this equation using the half-reaction method.

Figure by Didier Descouens. Image used by permission of Wikipedia Answer \[3CuS_{(s)} + 8HNO{3(aq)} \rightarrow 8NO_{(g)} + 3CuSO_{4(aq)} + 4H_2O_{(l)}\]
- Step 5: Add the two half-reactions and cancel substances that appear on both sides of the equation.
- Step 4: Multiply the reductive and oxidative half-reactions by appropriate integers to obtain the same number of electrons in both half-reactions.
- We have now balanced the atoms in each half-reaction, but the charges are not balanced.
- Balancing H atoms by adding H+, we obtain the following:
- Step 2: Balance the atoms by balancing elements other than O and H. Then balance O atoms by adding H2O and balance H atoms by adding H+.
Balancing a Redox Reaction in Acidic Conditions: https://youtu.be/IB-fWLsI0lc
Calculating Standard Cell Potentials
The standard cell potential for a redox reaction (E°cell) is a measure of the tendency of reactants in their standard states to form products in their standard states; consequently, it is a measure of the driving force for the reaction, which earlier we called voltage. We can use the two standard electrode potentials we found earlier to calculate the standard potential for the Zn/Cu cell represented by the following cell diagram:
\[ Zn{(s)}∣Zn^{2+}(aq, 1 M)∥Cu^{2+}(aq, 1 M)∣Cu_{(s)} \label{19.40}\]
We know the values of E°anode for the reduction of Zn2+ and E°cathode for the reduction of Cu2+, so we can calculate E°cell:
\[E°_{cell} = E°_{cathode} − E°_{anode} = 1.10\; V\]
This is the same value that is observed experimentally. If the value of E°cell is positive, the reaction will occur spontaneously as written. If the value of E°cell is negative, then the reaction is not spontaneous, and it will not occur as written under standard conditions; it will, however, proceed spontaneously in the opposite direction. As we shall see, this does not mean that the reaction cannot be made to occur at all under standard conditions. With a sufficient input of electrical energy, virtually any reaction can be forced to occur. Example 4 and its corresponding exercise illustrate how we can use measured cell potentials to calculate standard potentials for redox couples.
Note
A positive E°cell means that the reaction will occur spontaneously as written. A negative E°cell means that the reaction will proceed spontaneously in the opposite direction.
Example \(\PageIndex{2}\)
A galvanic cell with a measured standard cell potential of 0.27 V is constructed using two beakers connected by a salt bridge. One beaker contains a strip of gallium metal immersed in a 1 M solution of GaCl3, and the other contains a piece of nickel immersed in a 1 M solution of NiCl2. The half-reactions that occur when the compartments are connected are as follows:
cathode: Ni2+(aq) + 2e− → Ni(s)
anode: Ga(s) → Ga3+(aq) + 3e−
If the potential for the oxidation of Ga to Ga3+ is 0.55 V under standard conditions, what is the potential for the oxidation of Ni to Ni2+?
Given: galvanic cell, half-reactions, standard cell potential, and potential for the oxidation half-reaction under standard conditions
Asked for: standard electrode potential of reaction occurring at the cathode
Strategy:
- Write the equation for the half-reaction that occurs at the anode along with the value of the standard electrode potential for the half-reaction.
- Use Equation \(\ref{19.10}\) to calculate the standard electrode potential for the half-reaction that occurs at the cathode. Then reverse the sign to obtain the potential for the corresponding oxidation half-reaction under standard conditions.
Solution
A We have been given the potential for the oxidation of Ga to Ga3+ under standard conditions, but to report the standard electrode potential, we must reverse the sign. For the reduction reaction Ga3+(aq) + 3e− → Ga(s), E°anode = −0.55 V.
B Using the value given for E°cell and the calculated value of E°anode, we can calculate the standard potential for the reduction of Ni2+ to Ni from Equation \(\ref{19.10}\):
This is the standard electrode potential for the reaction Ni2+(aq) + 2e− → Ni(s). Because we are asked for the potential for the oxidation of Ni to Ni2+ under standard conditions, we must reverse the sign of E°cathode. Thus E° = −(−0.28 V) = 0.28 V for the oxidation. With three electrons consumed in the reduction and two produced in the oxidation, the overall reaction is not balanced. Recall, however, that standard potentials are independent of stoichiometry.
Exercise \(\PageIndex{2}\)
A galvanic cell is constructed with one compartment that contains a mercury electrode immersed in a 1 M aqueous solution of mercuric acetate \(Hg(CH_3CO_2)_2\) and one compartment that contains a strip of magnesium immersed in a 1 M aqueous solution of \(MgCl_2\). When the compartments are connected, a potential of 3.22 V is measured and the following half-reactions occur:
- cathode: Hg2+(aq) + 2e− → Hg(l)
- anode: Mg(s) → Mg2+(aq) + 2e−
If the potential for the oxidation of Mg to Mg2+ is 2.37 V under standard conditions, what is the standard electrode potential for the reaction that occurs at the anode?
Answer 0.85 V
We can use this procedure described to measure the standard potentials for a wide variety of chemical substances, some of which are listed in Table P2. These data allow us to compare the oxidative and reductive strengths of a variety of substances. The half-reaction for the standard hydrogen electrode (SHE) lies more than halfway down the list in Table \(\PageIndex{1}\). All reactants that lie below the SHE in the table are stronger oxidants than H+, and all those that lie above the SHE are weaker. The strongest oxidant in the table is F2, with a standard electrode potential of 2.87 V. This high value is consistent with the high electronegativity of fluorine and tells us that fluorine has a stronger tendency to accept electrons (it is a stronger oxidant) than any other element.
| Half-Reaction | E° (V) |
|---|---|
| Li+(aq) + e− \(\rightleftharpoons\) Li(s) | –3.040 |
| Be2+(aq) + 2e− \(\rightleftharpoons\) Be(s) | –1.99 |
| Al3+(aq) + 3e− \(\rightleftharpoons\) Al(s) | –1.676 |
| Zn2+(aq) + 2e− \(\rightleftharpoons\) Zn(s) | –0.7618 |
| Ag2S(s) + 2e− \(\rightleftharpoons\) 2Ag(s) + S2−(aq) | –0.71 |
| Fe2+(aq) + 2e− \(\rightleftharpoons\) Fe(s) | –0.44 |
| Cr3+(aq) + e− \(\rightleftharpoons\) Cr2+(aq) | –0.424 |
| Cd2+(aq) + 2e− \(\rightleftharpoons\) Cd(s) | –0.4030 |
| PbSO4(s) + 2e− \(\rightleftharpoons\) Pb(s) + SO42−(aq) | –0.356 |
| Ni2+(aq) + 2e− \(\rightleftharpoons\) Ni(s) | –0.257 |
| 2SO42−(aq) + 4H+(aq) + 2e− \(\rightleftharpoons\) S2O62−(aq) + 2H2O(l) | –0.25 |
| Sn2+(aq) + 2e− \(\rightleftharpoons\) Sn(s) | −0.14 |
| 2H+(aq) + 2e− \(\rightleftharpoons\) H2(g) | 0.00 |
| Sn4+(aq) + 2e− \(\rightleftharpoons\) Sn2+(aq) | 0.154 |
| Cu2+(aq) + e− \(\rightleftharpoons\) Cu+(aq) | 0.159 |
| AgCl(s) + e− \(\rightleftharpoons\) Ag(s) + Cl−(aq) | 0.2223 |
| Cu2+(aq) + 2e− \(\rightleftharpoons\) Cu(s) | 0.3419 |
| O2(g) + 2H2O(l) + 4e− \(\rightleftharpoons\) 4OH−(aq) | 0.401 |
| H2SO3(aq) + 4H+(aq) + 4e− \(\rightleftharpoons\) S(s) + 3H2O(l) | 0.45 |
| I2(s) + 2e− \(\rightleftharpoons\) 2I−(aq) | 0.5355 |
| MnO42−(aq) + 2H2O(l) + 2e− \(\rightleftharpoons\) MnO2(s) + 4OH−(aq) | 0.60 |
| O2(g) + 2H+(aq) + 2e− \(\rightleftharpoons\) H2O2(aq) | 0.695 |
| H2SeO3(aq) + 4H+ + 4e− \(\rightleftharpoons\) Se(s) + 3H2O(l) | 0.74 |
| Fe3+(aq) + e− \(\rightleftharpoons\) Fe2+(aq) | 0.771 |
| Ag+(aq) + e− \(\rightleftharpoons\) Ag(s) | 0.7996 |
| NO3−(aq) + 3H+(aq) + 2e− \(\rightleftharpoons\) HNO2(aq) + H2O(l) | 0.94 |
| Br2(aq) + 2e− \(\rightleftharpoons\) 2Br−(aq) | 1.087 |
| MnO2(s) + 4H+(aq) + 2e− \(\rightleftharpoons\) Mn2+(aq) + 2H2O(l) | 1.23 |
| O2(g) + 4H+(aq) + 4e− \(\rightleftharpoons\) 2H2O(l) | 1.229 |
| Cr2O72−(aq) + 14H+(aq) + 6e− \(\rightleftharpoons\) 2Cr3+(aq) + 7H2O(l) | 1.36 |
| Cl2(g) + 2e− \(\rightleftharpoons\) 2Cl−(aq) | 1.396 |
|
\[Ce^{4+}(aq) + e^− \rightleftharpoons Ce^{3+}(aq)\] |
1.44 |
| PbO2(s) + HSO4−(aq) + 3H+(aq) + 2e− \(\rightleftharpoons\) PbSO4(s) + 2H2O(l) | 1.690 |
| H2O2(aq) + 2H+(aq) + 2e− \(\rightleftharpoons\) 2H2O(l) | 1.763 |
| F2(g) + 2e−\(\rightleftharpoons\) 2F−(aq) | 2.87 |
Similarly, all species in Table \(\PageIndex{1}\) that lie above H2 are stronger reductants than H2, and those that lie below H2 are weaker. The strongest reductant in the table is thus metallic lithium, with a standard electrode potential of −3.04 V. This fact might be surprising because cesium, not lithium, is the least electronegative element. The apparent anomaly can be explained by the fact that electrode potentials are measured in aqueous solution, where intermolecular interactions are important, whereas ionization potentials and electron affinities are measured in the gas phase. Due to its small size, the Li+ ion is stabilized in aqueous solution by strong electrostatic interactions with the negative dipole end of water molecules. These interactions result in a significantly greater ΔHhydration for Li+ compared with Cs+. Lithium metal is therefore the strongest reductant (most easily oxidized) of the alkali metals in aqueous solution.
Note
Species in Talbe Table \(\PageIndex{1}\) (or Table P2) that lie above H2 are stronger reducing agents (more easily oxidized) than H2. Species that lie below H2 are stronger oxidizing agents.
Because the half-reactions shown in Table \(\PageIndex{1}\) are arranged in order of their E° values, we can use the table to quickly predict the relative strengths of various oxidants and reductants. Any species on the left side of a half-reaction will spontaneously oxidize any species on the right side of another half-reaction that lies below it in the table. Conversely, any species on the right side of a half-reaction will spontaneously reduce any species on the left side of another half-reaction that lies above it in the table. We can use these generalizations to predict the spontaneity of a wide variety of redox reactions (E°cell > 0), as illustrated below.
Example \(\PageIndex{3}\)
The black tarnish that forms on silver objects is primarily Ag2S. The half-reaction for reversing the tarnishing process is as follows:
- Referring to Table \(\PageIndex{1}\), predict which species—H2O2(aq), Zn(s), I−(aq), Sn2+(aq)—can reduce Ag2S to Ag under standard conditions.
- Of these species—H2O2(aq), Zn(s), I−(aq), Sn2+(aq), identify which is the strongest reducing agent in aqueous solution and thus the best candidate for a commercial product.
- From the data in Table \(\PageIndex{1}\), suggest an alternative reducing agent that is readily available, inexpensive, and possibly more effective at removing tarnish.

Given: reduction half-reaction, standard electrode potential, and list of possible reductants
Asked for: reductants for Ag2S, strongest reductant, and potential reducing agent for removing tarnish
Strategy:
A From their positions inTable \(\PageIndex{1}\), decide which species can reduce Ag2S. Determine which species is the strongest reductant.
B Use Table \(\PageIndex{1}\) to identify a reductant for Ag2S that is a common household product.
Solution
We can solve the problem in one of two ways: (1) compare the relative positions of the four possible reductants with that of the Ag2S/Ag couple in Table \(\PageIndex{1}\) or (2) compare E° for each species with E° for the Ag2S/Ag couple (−0.69 V).
- A The species in Table \(\PageIndex{1}\) are arranged from top to bottom in order of increasing reducing strength. Of the four species given in the problem, I−(aq), Sn2+(aq), and H2O2(aq) lie above Ag2S, and one [Zn(s)] lies below it. We can therefore conclude that Zn(s) can reduce Ag2S(s) under standard conditions, whereas I−(aq), Sn2+(aq), and H2O2(aq) cannot. Sn2+(aq) and H2O2(aq) appear twice in the table: on the left side (oxidant) in one half-reaction and on the right side (reductant) in another.
- The strongest reductant is Zn(s), the species on the right side of the half-reaction that lies closer to the bottom of Table \(\PageIndex{1}\) than the half-reactions involving I−(aq), Sn2+(aq), and H2O2(aq). (Commercial products that use a piece of zinc are often marketed as a “miracle product” for removing tarnish from silver. All that is required is to add warm water and salt for electrical conductivity.)
- B Of the reductants that lie below Zn(s) in Table \(\PageIndex{1}\), and therefore are stronger reductants, only one is commonly available in household products: Al(s), which is sold as aluminum foil for wrapping foods.
Example \(\PageIndex{4}\)
Use the data in Table \(\PageIndex{1}\) to determine whether each reaction is likely to occur spontaneously under standard conditions:
- Sn(s) + Be2+(aq) → Sn2+(aq) + Be(s)
- MnO2(s) + H2O2(aq) + 2H+(aq) → O2(g) + Mn2+(aq) + 2H2O(l)
Given: redox reaction and list of standard electrode potentials (Table P2 )
Asked for: reaction spontaneity
Strategy:
- Identify the half-reactions in each equation. Using Table \(\PageIndex{1}\), determine the standard potentials for the half-reactions in the appropriate direction.
- Use Equation \(\ref{19.10}\) to calculate the standard cell potential for the overall reaction. From this value, determine whether the overall reaction is spontaneous.
Solution
- A Metallic tin is oxidized to Sn2+(aq), and Be2+(aq) is reduced to elemental beryllium. We can find the standard electrode potentials for the latter (reduction) half-reaction (−1.85 V) and for the former (oxidation) half-reaction (−0.14 V) directly from Table \(\PageIndex{1}\).
B Adding the two half-reactions gives the overall reaction:
|
\(\textrm{cathode:} \; \mathrm{Be^{2+}(aq)} +\mathrm{2e^-} \rightarrow \mathrm{Be(s)}\) \(\textrm{anode:} \; \mathrm{Sn(s) \rightarrow \mathrm{Sn^{2+}}(s)} +\mathrm{2e^-} \) \(\textrm{total:} \; \mathrm{Sn(s)+ \mathrm{Be^{2+}(aq)} \rightarrow \mathrm{Sn^{2+}}(aq)} + \mathrm{Be(s)}\) |
\(E^\circ_{\textrm{cathode}}=\textrm{–1.99 V} \\ E^\circ_{\textrm{anode}}=\textrm{-0.14 V} \\ E^\circ_{\textrm{cell}}=E^\circ_{\textrm{cathode}}-E^\circ_{\textrm{anode}} \\ \hspace{5mm} =-\textrm{1.85 V}\) |
The standard cell potential is quite negative, so the reaction will not occur spontaneously as written. That is, metallic tin cannot reduce Be2+ to beryllium metal under standard conditions. Instead, the reverse process, the reduction of stannous ions (Sn2+) by metallic beryllium, which has a positive value of E°cell, will occur spontaneously.
- A MnO2 is the oxidant (Mn4+ is reduced to Mn2+), while H2O2 is the reductant (O2− is oxidized to O2). We can obtain the standard electrode potentials for the reduction and oxidation half-reactions directly from Table \(\PageIndex{1}\).
B The two half-reactions and their corresponding potentials are as follows
| \(\begin{align}\textrm{cathode:} & \mathrm{MnO_2(s)}+\mathrm{4H^+(aq)}+\mathrm{2e^-}\rightarrow\mathrm{Mn^{2+}(aq)}+\mathrm{2H_2O(l)} \nonumber \\ \textrm{anode:} & \mathrm{H_2O_2(aq)}\rightarrow\mathrm{O_2(g)}+\mathrm{2H^+(aq)}+\mathrm{2e^-} \nonumber\\ \textrm{overall:} & \mathrm{MnO_2(s)}+\mathrm{H_2O_2(aq)}+\mathrm{2H^+(aq)}\rightarrow\mathrm{O_2(g)}+\mathrm{Mn^{2+}(aq)}+\mathrm{2H_2O(l)} \nonumber\end{align}\) |
\(E^\circ_{\textrm{cathode}}=\textrm{1.22 V} \nonumber \\ E^\circ_{\textrm{anode}}=\textrm{0.70 V} \nonumber \\ E^\circ_{\textrm{cell}}=E^\circ_{\textrm{cathode}}-E^\circ_{\textrm{anode}} \nonumber \\ \hspace{5mm} =-\textrm{0.53 V}\) |
The standard potential for the reaction is positive, indicating that under standard conditions, it will occur spontaneously as written. Hydrogen peroxide will reduce MnO2, and oxygen gas will evolve from the solution.
Exercise \(\PageIndex{4}\)
Use the data in Table \(\PageIndex{1}\) to determine whether each reaction is likely to occur spontaneously under standard conditions:
- 2Ce4+(aq) + 2Cl−(aq) → 2Ce3+(aq) + Cl2(g)
- 4MnO2(s) + 3O2(g) + 4OH−(aq) → 4MnO4−(aq) + 2H2O
Answer
- spontaneous (E°cell = 0.36 V)
- nonspontaneous (E°cell = −0.20 V)
Although the sign of E°cell tells us whether a particular redox reaction will occur spontaneously under standard conditions, it does not tell us to what extent the reaction proceeds, and it does not tell us what will happen under nonstandard conditions. To answer these questions requires a more quantitative understanding of the relationship between electrochemical cell potential and chemical thermodynamics.
Reference Electrodes and Measuring Concentrations
When using a galvanic cell to measure the concentration of a substance, we are generally interested in the potential of only one of the electrodes of the cell, the so-called indicator electrode, whose potential is related to the concentration of the substance being measured. To ensure that any change in the measured potential of the cell is due to only the substance being analyzed, the potential of the other electrode, the reference electrode, must be constant. You are already familiar with one example of a reference electrode: the SHE. The potential of a reference electrode must be unaffected by the properties of the solution, and if possible, it should be physically isolated from the solution of interest. To measure the potential of a solution, we select a reference electrode and an appropriate indicator electrode. Whether reduction or oxidation of the substance being analyzed occurs depends on the potential of the half-reaction for the substance of interest (the sample) and the potential of the reference electrode.
Note
The potential of any reference electrode should not be affected by the properties of the solution to be analyzed, and it should also be physically isolated.
There are many possible choices of reference electrode other than the SHE. The SHE requires a constant flow of highly flammable hydrogen gas, which makes it inconvenient to use. Consequently, two other electrodes are commonly chosen as reference electrodes. One is the silver–silver chloride electrode, which consists of a silver wire coated with a very thin layer of AgCl that is dipped into a chloride ion solution with a fixed concentration. The cell diagram and reduction half-reaction are as follows:
\[Cl^−_{(aq)}∣AgCl_{(s)}∣Ag_{(s)} \label{19.44}\]
\[AgCl_{(s)}+e^− \rightarrow Ag_{(s)} + Cl^−_{(aq)}\]
If a saturated solution of KCl is used as the chloride solution, the potential of the silver–silver chloride electrode is 0.197 V versus the SHE. That is, 0.197 V must be subtracted from the measured value to obtain the standard electrode potential measured against the SHE.
A second common reference electrode is the saturated calomel electrode (SCE), which has the same general form as the silver–silver chloride electrode. The SCE consists of a platinum wire inserted into a moist paste of liquid mercury (Hg2Cl2; called calomel in the old chemical literature) and KCl. This interior cell is surrounded by an aqueous KCl solution, which acts as a salt bridge between the interior cell and the exterior solution (part (a) in Figure \(\PageIndex{4}\)). Although it sounds and looks complex, this cell is actually easy to prepare and maintain, and its potential is highly reproducible. The SCE cell diagram and corresponding half-reaction are as follows:
\[Pt_{(s)} ∣ Hg_2Cl_{2(s)}∣KCl_{(aq, sat)} \label{19.45}\]
\[Hg_2Cl_{2(s)} + 2e^− \rightarrow 2Hg_{(l)} + 2Cl^−{(aq)} \label{19.46}\]
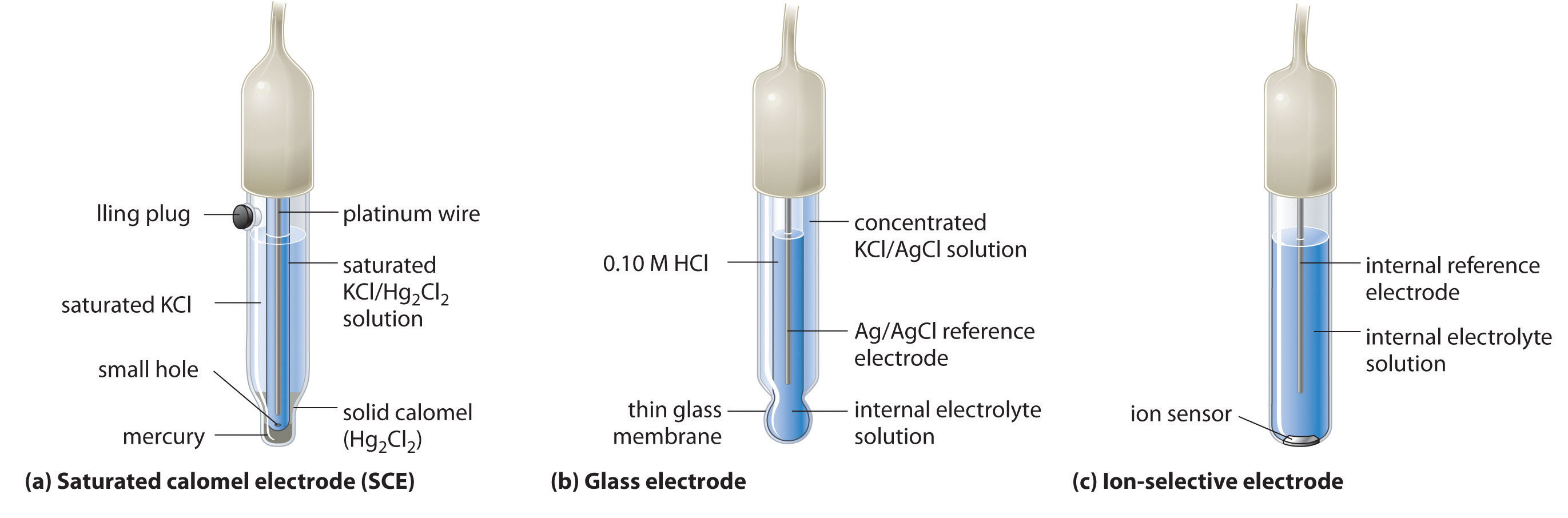
At 25°C, the potential of the SCE is 0.2415 V versus the SHE, which means that 0.2415 V must be subtracted from the potential versus an SCE to obtain the standard electrode potential.
One of the most common uses of electrochemistry is to measure the H+ ion concentration of a solution. A glass electrode is generally used for this purpose, in which an internal Ag/AgCl electrode is immersed in a 0.10 M HCl solution that is separated from the solution by a very thin glass membrane (part (b) in Figure \(\PageIndex{5}\)). The glass membrane absorbs protons, which affects the measured potential. The extent of the adsorption on the inner side is fixed because [H+] is fixed inside the electrode, but the adsorption of protons on the outer surface depends on the pH of the solution. The potential of the glass electrode depends on [H+] as follows (recall that pH = −log[H+]:
\[E_{glass} = E′ + (0.0591\; V \times \log[H^+]) = E′ − 0.0591\; V \times pH \label{19.47}\]
The voltage E′ is a constant that depends on the exact construction of the electrode. Although it can be measured, in practice, a glass electrode is calibrated; that is, it is inserted into a solution of known pH, and the display on the pH meter is adjusted to the known value. Once the electrode is properly calibrated, it can be placed in a solution and used to determine an unknown pH.
Ion-selective electrodes are used to measure the concentration of a particular species in solution; they are designed so that their potential depends on only the concentration of the desired species (part (c) in Figure \(\PageIndex{5}\)). These electrodes usually contain an internal reference electrode that is connected by a solution of an electrolyte to a crystalline inorganic material or a membrane, which acts as the sensor. For example, one type of ion-selective electrode uses a single crystal of Eu-doped \(LaF_3\) as the inorganic material. When fluoride ions in solution diffuse to the surface of the solid, the potential of the electrode changes, resulting in a so-called fluoride electrode. Similar electrodes are used to measure the concentrations of other species in solution. Some of the species whose concentrations can be determined in aqueous solution using ion-selective electrodes and similar devices are listed in Table \(\PageIndex{2}\).
| Species | Type of Sample |
|---|---|
| H+ | laboratory samples, blood, soil, and ground and surface water |
| NH3/NH4+ | wastewater and runoff water |
| K+ | blood, wine, and soil |
| CO2/HCO3− | blood and groundwater |
| F− | groundwater, drinking water, and soil |
| Br− | grains and plant extracts |
| I− | milk and pharmaceuticals |
| NO3− | groundwater, drinking water, soil, and fertilizer |
The Standard Hydrogen Electrode (SHE): https://youtu.be/GS-SE7IDDtY
Summary
The flow of electrons in an electrochemical cell depends on the identity of the reacting substances, the difference in the potential energy of their valence electrons, and their concentrations. The potential of the cell under standard conditions (1 M for solutions, 1 atm for gases, pure solids or liquids for other substances) and at a fixed temperature (25°C) is called the standard cell potential (E°cell). Only the difference between the potentials of two electrodes can be measured. By convention, all tabulated values of standard electrode potentials are listed as standard reduction potentials. The overall cell potential is the reduction potential of the reductive half-reaction minus the reduction potential of the oxidative half-reaction (E°cell = E°cathode − E°anode). The potential of the standard hydrogen electrode (SHE) is defined as 0 V under standard conditions. The potential of a half-reaction measured against the SHE under standard conditions is called its standard electrode potential. The standard cell potential is a measure of the driving force for a given redox reaction. All E° values are independent of the stoichiometric coefficients for the half-reaction. Redox reactions can be balanced using the half-reaction method, in which the overall redox reaction is divided into an oxidation half-reaction and a reduction half-reaction, each balanced for mass and charge. The half-reactions selected from tabulated lists must exactly reflect reaction conditions. In an alternative method, the atoms in each half-reaction are balanced, and then the charges are balanced. Whenever a half-reaction is reversed, the sign of E° corresponding to that reaction must also be reversed.
The oxidative and reductive strengths of a variety of substances can be compared using standard electrode potentials. Apparent anomalies can be explained by the fact that electrode potentials are measured in aqueous solution, which allows for strong intermolecular electrostatic interactions, and not in the gas phase.
If E°cell is positive, the reaction will occur spontaneously under standard conditions. If E°cell is negative, then the reaction is not spontaneous under standard conditions, although it will proceed spontaneously in the opposite direction. The potential of an indicator electrode is related to the concentration of the substance being measured, whereas the potential of the reference electrode is held constant. Whether reduction or oxidation occurs depends on the potential of the sample versus the potential of the reference electrode. In addition to the SHE, other reference electrodes are the silver–silver chloride electrode; the saturated calomel electrode (SCE); the glass electrode, which is commonly used to measure pH; and ion-selective electrodes, which depend on the concentration of a single ionic species in solution. Differences in potential between the SHE and other reference electrodes must be included when calculating values for E°.
Contributors and Attributions
- Anonymous
- cathode: \[Cu^{2+}_{(aq)} + 2e^− \rightarrow Cu_{(s)} \;\;\; E°_{cathode} = 0.34\; V \label{19.41}\]
- anode: \[Zn_{(s)} \rightarrow Zn^{2+}(aq, 1 M) + 2e^−\;\;\; E°_{anode} = −0.76\; V \label{19.42}\]
- overall: \[Zn_{(s)} + Cu^{2+}_{(aq)} \rightarrow Zn^{2+}_{(aq)} + Cu_{(s)} \label{19.43}\]
- Redox reactions can be balanced using the half-reaction method.
- The standard cell potential is a measure of the driving force for the reaction.
- The relative strengths of various oxidants and reductants can be predicted using E° values.

