1.10: Bonding in the Methyl Cation, the Methyl Radical, and the Methyl Anion
- Page ID
- 16303
17.1A: The geometry and relative stability of carbon radicals
As organic chemists, we are particularly interested in radical intermediates in which the unpaired electron resides on a carbon atom. Experimental evidence indicates that the three bonds in a carbon radical have trigonal planar geometry, and therefore the carbon is considered to be sp2-hybridized with the unpaired electron occupying the perpendicular, unhybridized 2pzorbital. Contrast this picture with carbocation and carbanion intermediates, which are both also trigonal planar but whose 2pz orbitals contain zero or two electrons, respectively.

The trend in the stability of carbon radicals parallels that of carbocations (section 8.4B): tertiary radicals, for example, are more stable than secondary radicals, followed by primary and methyl radicals. This should make intuitive sense, because radicals, like carbocations, can be considered to be electron deficient, and thus are stabilized by the electron-donating effects of nearby alkyl groups. Benzylic and allylic radicals are more stable than alkyl radicals due to resonance effects - an unpaired electron can be delocalized over a system of conjugated pi bonds. An allylic radical, for example, can be pictured as a system of three parallel 2pz orbitals sharing three electrons.
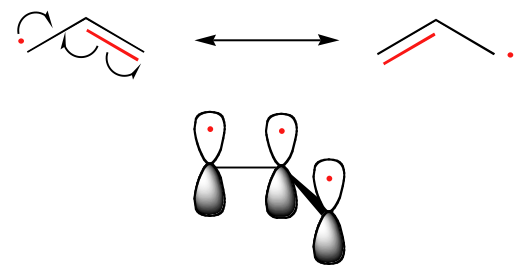

In this molecule, the already extensive resonance stabilization is further enhanced by the ability of the chlorine atoms to shield the radical center from external reagents. The radical is, in some sense, inside a protective 'cage'.
Exercise 17.2: Draw a resonance contributor of the structure above in which the unpaired electron is located on a chlorine atom.
17.1B: The diradical character of triplet oxygen
You may be surprised to learn that molecular oxygen (O2) often reacts like a radical species - or more accurately, like a diradical, with two separate unpaired valence electrons. This puzzling phenomenon is best explained by molecular orbital theory (you may want to go back to chapter 2 at this point to review basic MO theory).
In molecular orbital energy diagram form, the configuration of O2 looks like this:
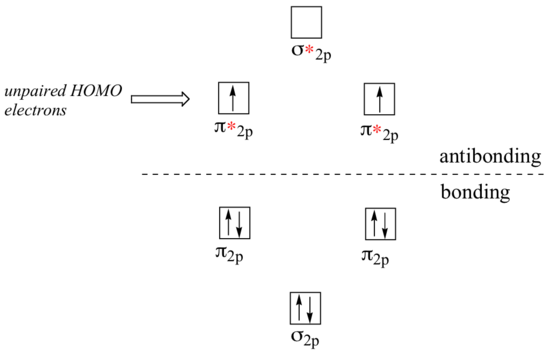
When the molecular orbitals of O2 are filled up with electrons according to Hund's rule (section 1.1C) , the HOMOs (Highest Occupied Molecular Orbitals) are the two antibonding π*2p orbitals, each holding a single, unpaired electron. This electron configuration, which describes oxygen in its lowest energy (ground) state, is referred to as the triplet state - the oxygen in the air around you istriplet oxygen. A higher energy state, in which the two highest energy electrons are paired in the same π*2p orbital, is called the singlet state of oxygen.
It is the last two unpaired electrons in triplet oxygen that react in radical-like fashion. For this reason, triplet O2 is often depicted by a Lewis structure as a diradical, with a single covalent bond, four lone pairs, and two unpaired electrons. The excited singlet state is often depicted as a doubly-bonded molecule.

These Lewis-dot depictions, while only approximations and unsatisfactory in many respects, can nonetheless be helpful in illustrating how O2 reacts. In any event, the molecular orbital picture of O2 is not simply an academic exercise: the diradical model for triplet oxygen is key to understanding its reactivity in many enzymatic and non-enzymatic contexts. We will see one example of triplet oxygen reactivity in section 17.2D.

