Diastereomers and Optical Resolution
- Page ID
- 4120
- Two enantiomers have the same physical properties. They cannot be separated easily by standard laboratory techniques.
- Two diastereomers have different physical properties and can often be separated via standard laboratory techniques.
- If an additional chiral center can be incorporated into a pair of enantiomers so that they become diastereomers, they can be separated.
An example of this method of obtaining one isomer of a compound involves the formation of a diastereomeric salt. The salt has multiple chiral centers, and so diastereomers are possible.
If a racemic mixture of phenylsuccinic acid is mixed with a pure sample of (-)-proline (a naturally available amino acid), a proton transfer (or Bronsted acid-base) reaction occurs. Two protons are transferred from the phenylsuccinic acid to the proline. One proline has a position available for one extra proton, so the two protons end up on two different prolines. Because protons have +1 charge, each proline is cationic. The phenylsuccinic acid gave up two positives, so it is a dianion. Together, these three ions form a salt.
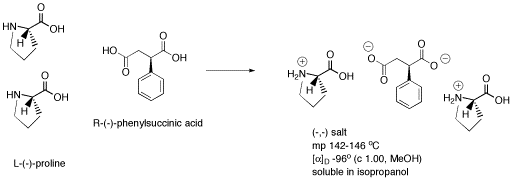
Figure SC14.1. Formation of a salt containing three chiral centers.
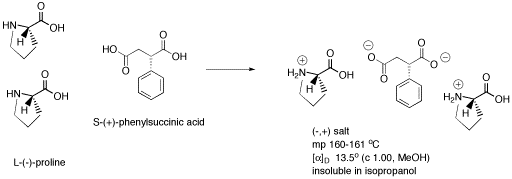
Figure SC14.2. Formation of a salt containing three chiral centers. This one is a diastereomer of the salt in the previous picture.
If a racemic mixture of phenylsuccinic acid is used, but pure (-)-proline is added, two possible diastereomers result. One chiral center, in the proline, is always the same. The other chiral center, in the phenylsuccinic acid, can be in two different configurations. As a result, cations and anions may pack differently together in each case, so different melting points and solubilities result. One salt precipitates or forms a solid from the solution, but the other stays dissolved. The two diastereomers can be separated by filtration.
Problem SC14.1.
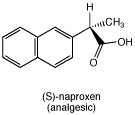
Suppose you have a pure sample of L-phenylalanine. Write equations for reactions, using structures, that show how you could use it to obtain a sample of (S)-naproxen, an analgesic.