Physical Properties of Cycloalkanes
- Page ID
- 853
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
Cycloalkanes are types of alkanes that have one or more rings of carbon atoms in their structure. The physical properties of cycloalkanes are similar to those of alkanes, but they have higher boiling points, melting points and higher densities due to the greater number of London forces that they contain.
Introduction
Cycloalkanes consist of carbon and hydrogen atoms that are saturated because of the single carbon-carbon bond (meaning that no more hydrogen atoms can be added). Cycloalkanes are also non polar and do not have intermolecular hydrogen bonding; they are usually hydrophobic (meaning they do not dissolve in water) and are less dense than water. Cycloalkanes can also be used for many different purposes. These uses are typically classified by the number of carbons in the cycloalkane ring. Many cycloalkanes are used in motor fuel, natural gas, petroleum gas, kerosene, diesel, and many other heavy oils. There are 4 general groups of cycloalkanes:
- Small rings (cyclopropane, cyclobutane)
- Common rings (cyclopentane, cyclohexane, cycloheptane)
- Medium rings (from 8-12 membered)
- Large rings (13 membered and higher)
Cycloalkanes can be substituted and named as cycloalkyl derivatives, and disubstituted cycloalkanes can be stereoisomers. In one isomer, if two substituents are placed on the same face or side of the ring, they are called cis. If the two substituents are on opposite faces, they are called trans. Substituents can also be either equitorial or axial on certain cycloalkanes, such as cyclohexane.
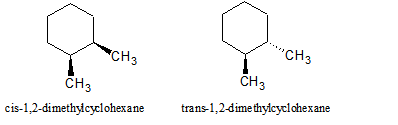
Figure 1: The cis and trans isomers are stereoisomers, meaning that they have identicial connectivities but a different arrangement of their atoms.
Generally, the melting point, the boiling point and the density of cycloalkanes increase as the number of carbons increases. This trend occurs because of the greater number of bonds that are in higher membered rings, thus making the bonds harder to break.
London Dispersion Forces and Cycloalkanes
Although alkanes are similar to cycloalkanes, they have higher London Dispersion forces because the ring shape allows for a greater area of contact. Ring strain also causes certain cylcoalkanes to be more reactive. London Dispersion Forces are the attractive or repulsive forces between molecules or between parts of the same molecule. For cycloalkanes, London dispersion forces refer to the repulsive forces between the molecules that cause ring strain.
Ring Strain in Cycloalkanes
Ring Strain occurs because the carbons in cycloalkanes are sp3 hybridized, which means that they do not have the expected ideal bond angle of 109.5o ; this causes an increase in the potential energy because of the desire for the carbons to be at an ideal 109.5o. An example of ring strain can be seen in the diagram of cyclopropane below in which the bond angle is 60o between the carbons.
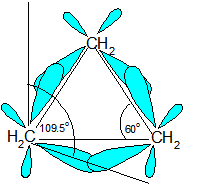
The reason for ring strain can be seen through the tetrahedral carbon model. The C-C-C bond angles in cyclopropane (diagram above) (60o) and cyclobutane (90o) are much different than the ideal bond angle of 109.5o. This bond angle causes cyclopropane and cyclobutane to have a high ring strain. However, molecules, such as cyclohexane and cyclopentane, would have a much lower ring strain because the bond angle between the carbons is much closer to 109.5o.
Below are some examples of cycloalkanes. Ring strain can be seen more prevalently in the cyclopropane and cyclobutane models.
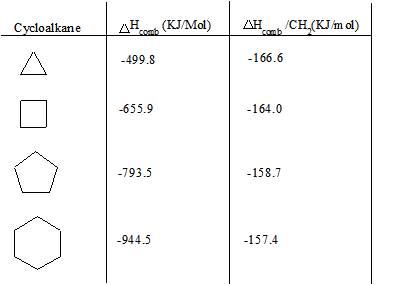
Below is a chart of cycloalkanes and their respective heats of combustion ( ΔHcomb). The ΔHcomb value increases as the number of carbons in the cycloalkane increases (higher membered ring), and the ΔHcomb/CH2 ratio decreases. The increase in ΔHcomb can be attributed to the greater amount of London Dispersion forces. However, the decrease in ΔHcomb/CH2can be attributed to a decrease in the ring strain.
How do cycloalkanes deal with ring strain?
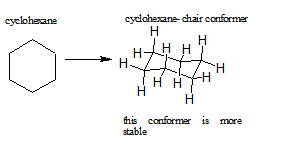
Certain cycloalkanes, such as cyclohexane, deal with ring strain by forming conformers. A conformer is a stereoisomer in which molecules of the same connectivity and formula exist as different isomers, in this case, to reduce ring strain. The ring strain is reduced in conformers due to the rotations around the sigma bonds. More about cyclohexane and its conformers can be seen here.
Different Types of Strain
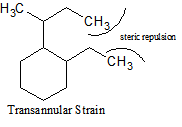
There are many different types of strain that occur with cycloalkanes. In addition to ring strain, there is also transannular strain, eclipsing, or torsional strain and bond angle strain.Transannular strain exists when there is steric repulsion between atoms. Eclipsing (torsional) strain exists when a cycloalkane is unable to adopt a staggered conformation around a C-C bond, and bond angle strain is the energy needed to distort the tetrahedral carbons enough to close the ring. The presence of angle strain in a molecule indicates that there are bond angles in that particular molecule that deviate from the ideal bond angles required (i.e., that molecule has conformers).


Outside links
References
- Vollhardt, K. Peter C., and Neil E. Schore. Organic Chemistry. 5th ed. New York: W.H. Freeman, 2007.
- John E. McMurry, and Eric E. Simanek. Fundamentals of Organic Chemistry. 6th Ed. Brooks Cole, 2006.
Problems
- Which has a higher melting point?
- cyclopentane
- cyclopropane
- cycloocatne
- they all have the same melting point.
- Why do cycloalkanes have different physical properties from normal alkanes?
- What is bond angle strain?
- What are London Dispersion Forces, and how do they play a part in cycloalkanes?
- What is the "ideal bond angle" for most cycloalkanes?
Answers
- C; as the number of carbons increases, so does the melting point. This happens because there are essentially more London dispersion forces acting upon the molecule which then makes it much harder to melt, or boil.
- Cycloalkanes have different physical properties from normal alkanes due to the greater number of London dispersion forces that they have. Cycloalkanes also have the ability to have more steric hindrance, thus increasing their relative energy level.
- Bond angle strain is is the energy needed to distort the tetrahedral carbons enough to close the ring
- London Dispersion Forces are the attractive or repulsive forces between molecules, or in between parts of the same molecule. They play a role in cycloalkanes because they refer to the intramolecular forces that causes ring strain.
- 109.5 o
Contributors
- Nicole Peiris

