9.19: Hydrogen Bonding
- Page ID
- 53759
↵
What's the difference between these two molecules?
A rough rule of thumb is that higher molecular-weight materials have higher boiling points than their lower molecular-weight counterparts. More energy is needed to move the larger molecule from the liquid state to the vapor state. However, ammonia has a boiling point of \(-33.34^\text{o} \text{C}\) and a molecular weight of 17, while nitrogen (molecular weight 28) has a boiling point of \(-195.8^\text{o} \text{C}\). The lighter ammonia molecule must have other factors that influence its physical properties.
Hydrogen Bonding
The attractive force between water molecules is a dipole interaction. The hydrogen atoms are bound to the highly electronegative oxygen atom (which also possesses two lone pair sets of electrons, making for a very polar bond). The partially positive hydrogen atom of one molecule is then attracted to the oxygen atom of a nearby water molecule (see figure below).
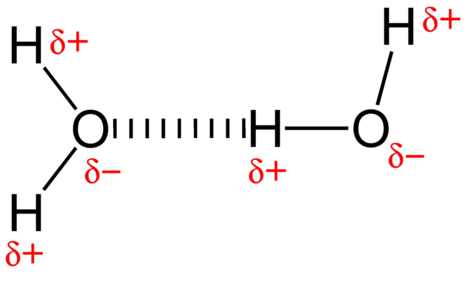
A hydrogen bond is an intermolecular attractive force in which a hydrogen atom that is covalently bonded to a small, highly electronegative atom is attracted to a lone pair of electrons on an atom in a neighboring molecule. Hydrogen bonds are very strong compared to other dipole interactions. The strength of a typical hydrogen bond is about \(5\%\) of that of a covalent bond.
Hydrogen bonding occurs only in molecules where hydrogen is covalently bonded to one of three elements: fluorine, oxygen, or nitrogen. These three elements are so electronegative that they withdraw the majority of the electron density in the covalent bond with hydrogen, leaving the \(\ce{H}\) atom very electron-deficient. The \(\ce{H}\) atom nearly acts as a bare proton, leaving it very attracted to lone pair electrons on a nearby atom.
The hydrogen bonding that occurs in water leads to some unusual, but very important, properties. Most molecular compounds that have a mass similar to water are gases at room temperature. Because of the strong hydrogen bonds, water molecules are able to stay condensed in the liquid state. The figure below shows how the bent shape, and two hydrogen atoms per molecule, allows each water molecule to be able to hydrogen bond to two other molecules.
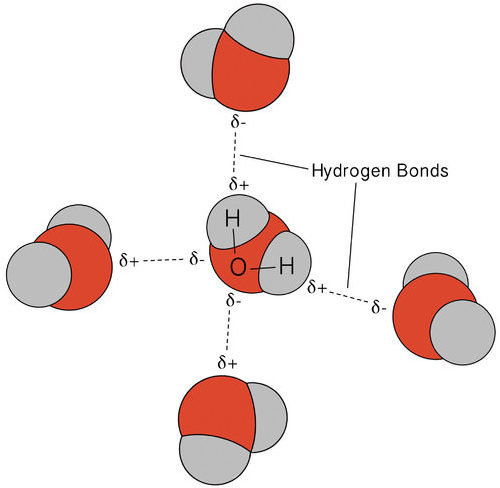
In the liquid state, the hydrogen bonds of water can break and reform as the molecules flow from one place to another. When water is cooled, the molecules begin to slow down. Eventually, when water is frozen to ice, the hydrogen bonds become permanent and form a very specific network (see figure below).
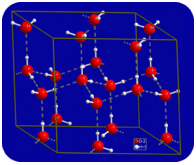
The bent shape of the molecules leads to gaps in the hydrogen bonding network of ice. Ice has a very unusual property—its solid state is less dense than its liquid state. Ice floats in water. Virtually all other substances are denser in the solid state than in the liquid state. Hydrogen bonds play a very important biological role in the physical structures of proteins and nucleic acids.
Why does water form droplets? Hint: it has to do with the attraction between water molecules. Learn more (and see how many drops of water you can get a penny can hold) in this simulation.
Summary
- Hydrogen bonds form when a \(\ce{H}\) attached to a \(\ce{N}\), \(\ce{O}\), or \(\ce{F}\) atom interacts with another \(\ce{N}\), \(\ce{O}\), or \(\ce{F}\) atom.
Review
- How strong is a hydrogen bond?
- What happens when H is covalently bonded to N, O, or F?
- How does the shape of the water molecule affect the properties of ice?

