5.16: Pauli Exclusion Principle
- Page ID
- 52974

Can you name one thing that easily distinguishes you from the rest of the world?
And we're not talking about DNA—that's a little expensive to sequence. For many people, it is their email address. My email address allows people all over the world to contact me. It does not belong to anyone else, but serves to identify me. Electrons also have a unique set of identifiers in the quantum numbers that describe their location and spin.
Pauli Exclusion Principle
When we look at the orbital possibilities for a given atom, we see that there are different arrangements of electrons for each different type of atom. Since each electron must maintain its unique identity, we intuitively sense that the four quantum numbers for any given electron must not match up exactly with the four quantum numbers for any other electron in that atom.
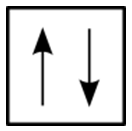
For the hydrogen atom, there is no problem since there is only one electron in the \(\ce{H}\) atom. However, for helium, we see that the first three quantum numbers for the two electrons are the same: same energy level, same spherical shape. What differentiates the two helium electrons is their spin. One of the electrons has a \(+\frac{1}{2}\) spin while the other electron has a \(-\frac{1}{2}\) spin. So, the two electrons in the \(1s\) orbital are each unique and distinct from one another because their spins are different. This observation leads to the Pauli exclusion principle, which states that no two electrons in an atom can have the same set of four quantum numbers. The energy of the electron is specified by the principal, angular momentum, and magnetic quantum numbers. If those three numbers are identical for two electrons, the spin numbers must be different in order for the two electrons to be differentiated from one another. The two values of the spin quantum number allow each orbital to hold two electrons. Figure \(\PageIndex{2}\) shows how the electrons are indicated in a diagram.
Summary
- The Pauli exclusion principle specifies limits on how identical quantum numbers can be for two electrons in the same atom.
Review
- What is the difference between the two helium electrons?
- What does the Pauli exclusion principle state?
- What does the two values for the spin quantum number allow?

