18.12: Heterogeneous Catalysis
- Page ID
- 49634
Our previous discussion has concentrated on catalysts which are in the same phase as the reaction being catalyzed. This kind of catalysis is called homogeneous catalysis. Many important industrial processes rely on heterogeneous catalysis, in which the catalyst is in a different phase. Usually the catalyst is a solid and the reactants are gases, and so the rate-limiting step occurs at the solid surface. Thus heterogeneous catalysis is also referred to as surface catalysis.
The detailed mechanisms of most heterogeneous reactions are not yet understood, but certain sites on the catalyst surface appear to be able to weaken or break bonds in reactant molecules. These are called active sites. One example of heterogeneous catalysis is hydrogenation of an unsaturated organic compound such as ethane (C2H4) by metal catalysts such as Pt or Ni:
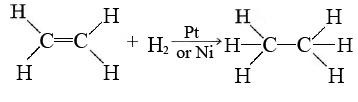 The currently accepted mechanism for this reaction involves weak bonding of both H2 and C2H4 to atoms on the metal surface. This is called adsorption. The H2 molecules dissociate to individual H atoms, each of which is weakly bonded to a Pt atom:
The currently accepted mechanism for this reaction involves weak bonding of both H2 and C2H4 to atoms on the metal surface. This is called adsorption. The H2 molecules dissociate to individual H atoms, each of which is weakly bonded to a Pt atom:
-
- \[\ce{H2 + 2Pt (surface) -> 2H ---Pt(surface)} \nonumber \]
These adsorbed H atoms can move across the metal surface, and eventually they combine with a C2H4 molecule, completing the reaction. Because adsorption and dissociation of H2 on a Pt surface is exothermic (ΔHm° >= –160 kJ mol–1), it can provide H atoms for further reaction without a large activation energy. By contrast, dissociation of gaseous H2 molecules without a metal surface would require the full bond enthalpy (ΔHm° = +436 kJ mol–1). Clearly the metal surface makes a major contribution in lowering the activation energy.
Heterogeneous catalysts are used extensively in the petroleum industry. One example is the combination of SiO2 and Al2O3 used to speed up cracking of long-chain hydrocarbons into the smaller molecules needed for gasoline. Another is the Pt catalyst used to reform hydrocarbon chains into aromatic ring structures. This improves the octane rating of gasoline, making it more suitable for use in automobile engines. Other industries also make effective use of catalysts. SO2, obtained by burning sulfur (or even from burning coal), can be oxidized to SO3 over vanadium pentoxide, V2O5. This is an important step in manufacturing H2SO4.
Another important heterogeneous catalyst is used in the Haber process for synthesis of NH3 from N2 and H2. As with most industrial catalysts, its exact composition is a trade secret, but it is mainly Fe with small amounts of Al2O3 and K2O added.
The surface catalyst you are most likely to be familiar with is found in the exhaust systems of many automobiles constructed since 1976. Such a catalytic converter contains from 1 to 3 g Pt in a fine layer on the surface of a honeycomb-like structure or small beads made of Al2O3. The catalyst speeds up oxidation of unburned hydrocarbons and CO which would otherwise be emitted from the exhaust as air pollutants. It apparently does this by adsorbing and weakening the bond in the O2 molecule. Individual O atoms are then more readily transferred to CO or hydrocarbon molecules, producing CO2 and H2O. This action of the catalytic surface can be inhibited or poisoned if lead atoms (from tetraethyllead in leaded gasoline) react with the surface. Hence the prohibition of use of leaded fuel in cars equipped with catalytic converters.


