4.7: The Nuclear Atom
- Page ID
- 49313
It should be clear by now that elements follow a periodic law, but atomic weight is inadequate for fully explaining this phenomenon. If ordered by atomic weight, chemical and physical properties will repeat at regular intervals, but there are clear exceptions to this behavior, for instance, K has a smaller atomic weight than Ar, but its properties would place it after Ar in terms of periodic behavior. All of this implies that there is another aspect to atomic structure to explain this law. Today, we know that such properties arise from a nuclear model of organization for atomic structure. The atom is not indivisible as Dalton suggested, but rather made up of a small, dense nucleus containing neutrons and protons, as well as rapidly moving electrons which take up the greater volume of an atom.
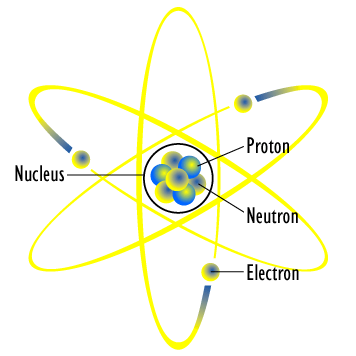
What led to this model? Beginning in the late 19th century a number of major experiments were performed. The results of these experiments helped scientists to formulate the current view of atomic structure. From this model of atomic structure, the origin of the periodicity of the elements can be more deeply explained. The order of the elements in the periodic table is dictated mostly by atomic weight which is directly related to the number of protons in the nucleus and the number of electrons surrounding the nucleus. The model of the Nuclear Atom reveals how the arrangement of electrons dictates the Periodic Law.


