26.5 Amino Acid Analysis of Peptides
- Page ID
- 91060
Objectives
After completing this section, you should be able to describe, briefly, how the identity and amounts of each amino acid residue present in a peptide of unknown structure may be determined.
Make certain that you can define, and use in context, the key term below.
- amino acid analyzer
You need not memorize the reaction between ninhydrin and an α‑amino acid.
Ion-exchange chromatography
When a protein is to be analysed, it is first heated with acid to hydrolyse all the peptide bonds. When such a mixture of amino acids is to be purified and estimated quantitatively, ion-exchange chromatography is the technique of choice. Fully automated amino acid analyzers are now available, which are equipped with a solvent pump to deliver the required buffer(s) in a programmed manner. There is a column, filled with Dowex 50 resin (Fig 26.5.1). This solid support is made up of polymeric beads. Chemically speaking they are polymers bearing arylsulfonic acid groups. The cation exchange resin helps in the separation of amino acids. In a typical run (Fig 26.5.2), the eluent is a buffer. The pH value of the buffer could be varied as step elution or as gradient elution. The chromatogram shown in Fig 26.5.2 is a chromatogram run with gradient elution technique, using ninhydrin as the post column treatment. The detector is a UV detector scanning the wavelengths 570 nm and 440 nm.
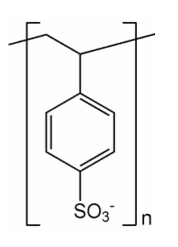
Fig 26.5.1: A Cation Rasin like Dowex 50 is a polymeric bead bearing aryl sulfonic acid groups
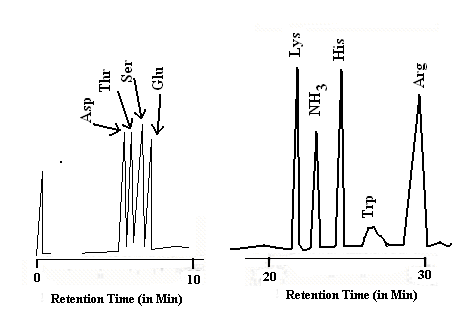
Fig 26.5.2: Some typical chromatograms from an amino acid analyzer
The Ninhydrin Reaction
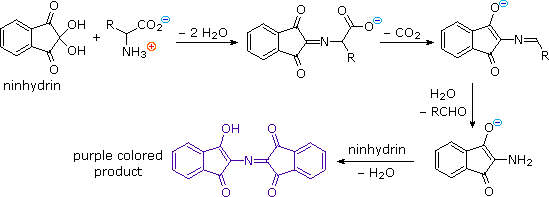
Alpha-amino acids show reactivity at their the carboxylic acid and amine sites typical of those functional groups. In addition to these common reactions of amines and carboxylic acids, common alpha-amino acids, except proline, undergo a unique reaction with the triketohydrindene hydrate known as ninhydrin. Among the products of this unusual reaction (shown on the left below) is a purple colored imino derivative, which provides as a useful color test for these amino acids, most of which are colorless. A common application of the ninhydrin test is the visualization of amino acids in paper chromatography. As shown in the graphic on the right, samples of amino acids or mixtures thereof are applied along a line near the bottom of a rectangular sheet of paper (the baseline). The bottom edge of the paper is immersed in an aqueous buffer, and this liquid climbs slowly toward the top edge. As the solvent front passes the sample spots, the compounds in each sample are carried along at a rate which is characteristic of their functionality, size and interaction with the cellulose matrix of the paper. Some compounds move rapidly up the paper, while others may scarcely move at all. The ratio of the distance a compound moves from the baseline to the distance of the solvent front from the baseline is defined as the retardation (or retention) factor Rf. Different amino acids usually have different Rf's under suitable conditions. In the example on the right, the three sample compounds (1, 2 & 3) have respective Rf values of 0.54, 0.36 & 0.78.
Exercise
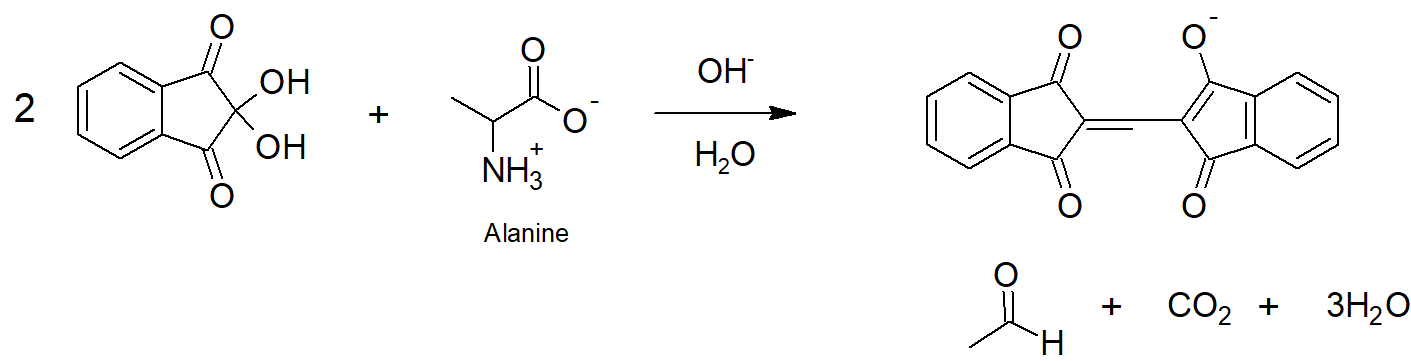
Show the structure of the products obtained of reaction of alanine with ninhydrin.
- Answer
-
Contributors and Attributions
Dr. Dietmar Kennepohl FCIC (Professor of Chemistry, Athabasca University)
Prof. Steven Farmer (Sonoma State University)
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry
- Prof. R Balaji Rao (Department of Chemistry, Banaras Hindu University, Varanasi) as part of Information and Communication Technology

