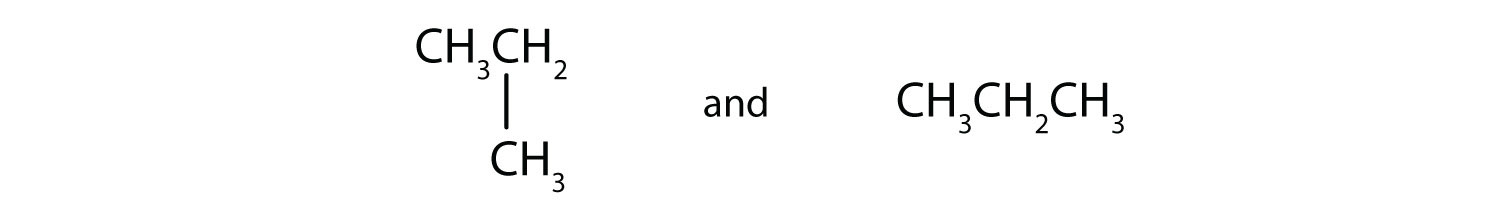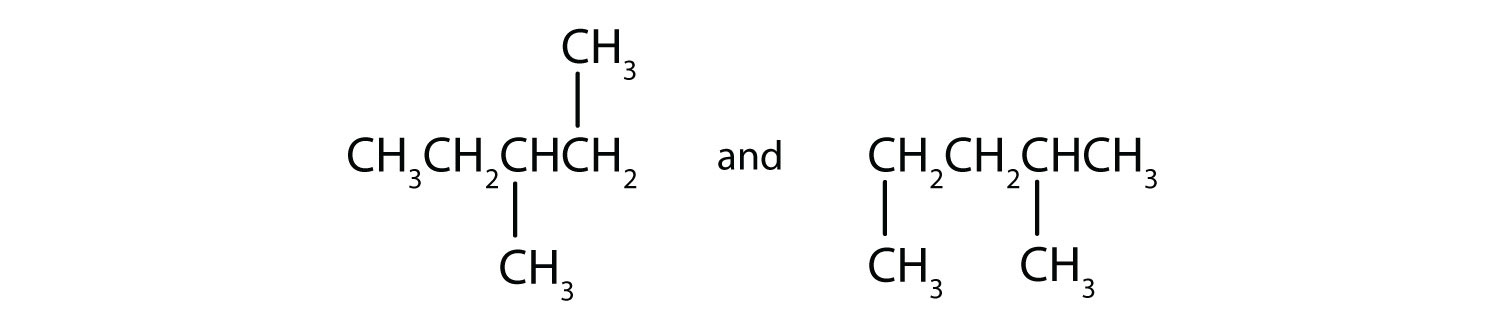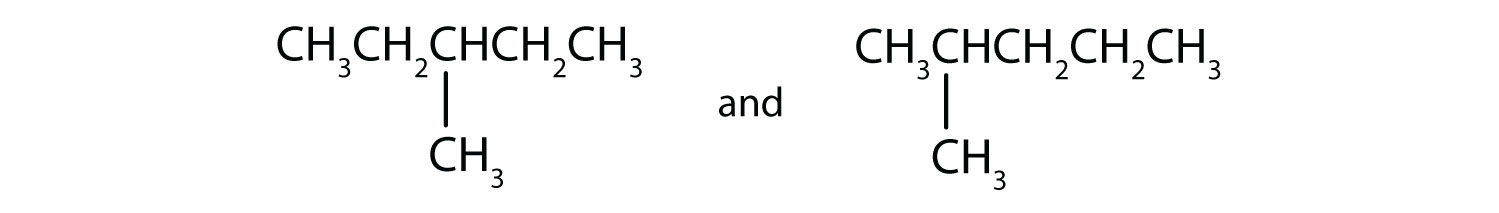6.E: Introduction to Organic Chemistry (Exercises)
- Page ID
- 521866
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Introduction to Organic Chemistry
Concept Review Exercises
-
Classify each compound as organic or inorganic.
- C3H8O
- CaCl2
- Cr(NH3)3Cl3
- C30H48O3N
-
Classify each compound as organic or inorganic.
- C6H10
- CoCl2
- C12H22O11
-
Classify each compound as organic or inorganic.
- CH3NH2
- NaNH2
- Cu(NH3)6Cl2
-
What compounds contain fewer carbon atoms than C3H8 and are its homologs?
-
What compounds contain five to eight carbon atoms and are homologs of C4H10?
Answers
-
a. organic / b. inorganic / c. inorganic / d. organic
2.
- organic
- inorganic
- organic
4. CH4 and C2H6
Structures and Names of Alkanes
Concept Review Exercises
- In the homologous series of alkanes, what is the molecular formula for the member just above C8H18?
- Use the general formula for alkanes to write the molecular formula of the alkane with 12 carbon atoms.
Answers
-
C9H20
-
C12H26
Exercises
-
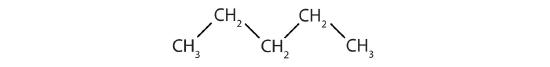
Write the condensed structural formula for each structural formula.
-
-
A condensed structural formula for isohexane can be written as (CH3)2CHCH2CH2CH3. Draw the line-angle formula for isohexane.
-
Draw a line-angle formula for the compound CH3CH2CH(CH3)CH2CH2CH3.
-
Give the structural formula for the compound represented by this line-angle formula:

1. Write the molecular formula for the following compounds shown in their line bond structure.
a.

b.

c.

2. Draw an isomer of the following compounds.
a.
b.

3. Draw condensed formula for the following compounds shown in their line bond structure.
a.

b.

c.

4. Draw skeletal formula for the compounds in question 3 shown in their line bond structure.
5. The molecules shown below are in their condensed structural formula. Draw line bond formula, molecular formula, and skeletal formula of the compounds.
a. CH3CH2NH2
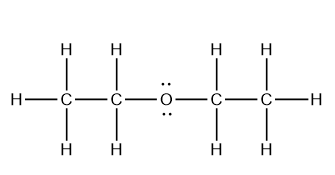
b. CH3OCH2CH2CH3
c. CH3CH2CH2Cl
d. CH3CH2CH2CH2CH3
-
Name each compound.
-
-
Write the structural formula for each compound.
- hexane
- octane
Answer
-
- CH3CH3
- CH3CH2CH3
- CH3CH2CH2CH2CH3
-
- pentane
- heptane
Branched-Chain Alkanes
Concept Review Exercises
-
In alkanes, can there be a two-carbon branch off the second carbon atom of a four-carbon chain? Explain.
-
A student is asked to write structural formulas for two different hydrocarbons having the molecular formula C5H12. She writes one formula with all five carbon atoms in a horizontal line and the other with four carbon atoms in a line, with a CH3 group extending down from the first attached to the third carbon atom. Do these structural formulas represent different molecular formulas? Explain why or why not.
Answers
-
No; the branch would make the longest continuous chain of five carbon atoms.
-
No; both are five-carbon continuous chains.
Exercises
-
Briefly identify the important distinctions between a straight-chain alkane and a branched-chain alkane.
-
How are butane and isobutane related? How do they differ?
-
Indicate whether the structures in each set represent the same compound or isomers.
-
- CH3CH2CH2CH3 and

- CH3CH2CH2CH2CH3 and

- CH3CH2CH2CH3 and
Answers
-
Straight-chain alkanes and branched-chain alkanes have different properties as well as different structures.
- no
- yes
IUPAC Nomenclature
Concept Review Exercises
- What is a CH3 group called when it is attached to a chain of carbon atoms—a substituent or a functional group?
- Which type of name uses numbers to locate substituents—common names or IUPAC names?
Answers
-
substituent
-
IUPAC names
Exercises
-
Briefly identify the important distinctions between an alkane and an alkyl group.
-
How many carbon atoms are present in each molecule?
- 2-methylbutane
- 3-ethylpentane
-
How many carbon atoms are present in each molecule?
- 2,3-dimethylbutane
- 3-ethyl-2-methylheptane
-
Draw the structure for each compound.
- 3-methylpentane
- 2,2,5-trimethylhexane
- 4-ethyl-3-methyloctane
-
Draw the structure for each compound.
- 2-methylpentane
- 4-ethyl-2-methylhexane
- 2,2,3,3-tetramethylbutane
-
Name each compound according to the IUPAC system.
-
-
Name each compound according to the IUPAC system.
-
-
What is a substituent? How is the location of a substituent indicated in the IUPAC system?
-
Briefly identify the important distinctions between a common name and an IUPAC name.
Answers
-
An alkane is a molecule; an alkyl group is not an independent molecule but rather a part of a molecule that we consider as a unit.
-
- 6
- 10
-
- 2,2,4,4-tetramethylpentane
- 3-ethylhexane
-
Common names are widely used but not very systematic; IUPAC names identify a parent compound and name other groups as substituents.
Cycloalkanes
Concept Review Exercises
- What is the molecular formula of cyclooctane?
- What is the IUPAC name for this compound?

Answers
- C8H16
- ethylcyclopropane
Exercises
- Draw the structure for each compound.
- ethylcyclobutane
- propylcyclopropane
- Draw the structure for each compound.
- methylcyclohexane
- butylcyclobutane
- Cycloalkyl groups can be derived from cycloalkanes in the same way that alkyl groups are derived from alkanes. These groups are named as cyclopropyl, cyclobutyl, and so on. Name each cycloalkyl halide.
-
- Halogenated cycloalkanes can be named by the IUPAC system. As with alkyl derivatives, monosubstituted derivatives need no number to indicate the position of the halogen. To name disubstituted derivatives, the carbon atoms are numbered starting at the position of one substituent (C1) and proceeding to the second substituted atom by the shortest route. Name each compound.
-
Answers
-
- cyclopentyl bromide
- cyclohexyl chloride
Halogenated Hydrocarbons
Concept Review Exercises
- What is the IUPAC name for the HFC that has the formula CH2FCF3? (Hint: you must use a number to indicate the location of each substituent F atom.)
- What is the IUPAC name for the HCFC that has the formula CHCl2CF3?
Answers
- 1,1,1,2-tetrafluoroethane
- 1,1,1-trifluoro-2,2-dichloroethane
Exercises
- Write the condensed structural formula for each compound.
- methyl chloride
- chloroform
- Write the condensed structural formula for each compound.
- ethyl bromide
- carbon tetrachloride
- Write the condensed structural formulas for the two isomers that have the molecular formula C3H7Br. Give the common name and the IUPAC name of each.
- Write the condensed structural formulas for the four isomers that have the molecular formula C4H9Br. Give the IUPAC name of each.
- What is a CFC? How are CFCs involved in the destruction of the ozone layer?
- Explain why each compound is less destructive to the ozone layer than are CFCs.
- fluorocarbons
- HCFCs
Answers
-
- CH3Cl
- CHCl3
- CH3CH2CH2Br, propyl bromide, 1-bromopropane; CH3CHBrCH3, isopropyl bromide, 2-bromopropane
- compounds containing Cl, F, and C; by releasing Cl atoms in the stratosphere
Classifying Organic Molecules
Concept Review Exercises
- What is a functional group? Give at least two examples of functional groups.
Answers
- A functional group is a specific structural arrangement of atoms or bonds that imparts a characteristic chemical reactivity to the molecule; alcohol group and carboxylic group (answers will vary).
Exercises
-
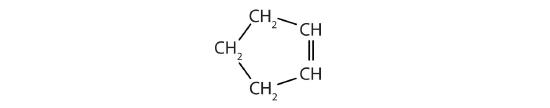
Classify each compound as saturated or unsaturated.
-

- CH3C≡CCH3
-
-
Classify each compound as saturated or unsaturated.
-
-
Identify the type of hydrocarbon in each structure.
-
-
-
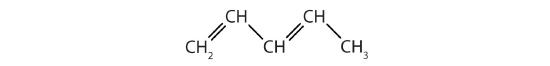
-

Identify the type of hydrocarbon in each structure.
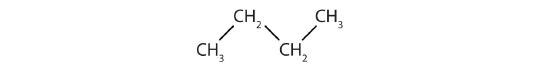
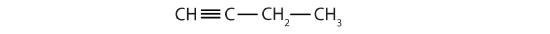
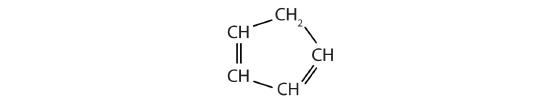
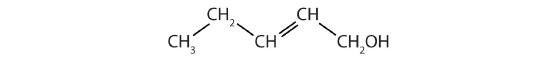
Identify the functional group(s) in each molecule.
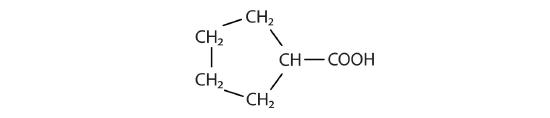
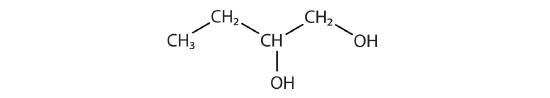
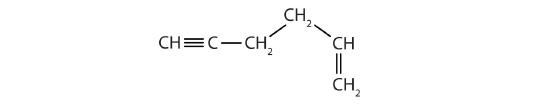
Identify the functional group(s) in each molecule.
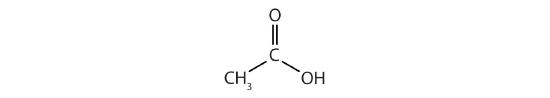
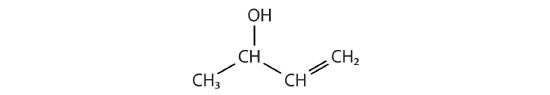
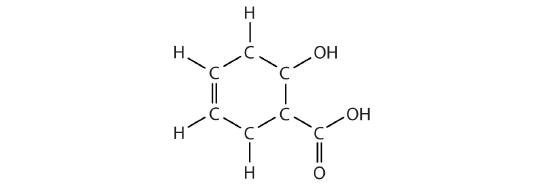
How many functional groups described in this section contain carbon and hydrogen atoms only? Name them.
What is the difference in the ways the two oxygen atoms in the carboxyl group are bonded to the carbon atom?
Answers
-
- unsaturated
- unsaturated
4.
↵5.
6.
a. a carbon-carbon double bond and alcohol
b. carboxyl group
c. carbon-carbon double bond and alcohol
d. carbon-carbon double bond; alcohol and carboxyl group
7. two; carbon-carbon double bonds and carbon-carbon triple bonds
8. There are two oxygen atoms in a carboxyl group: one is double-bonded while the other is OH, single bonded to the same carbon atom.
Classification of Alcohols
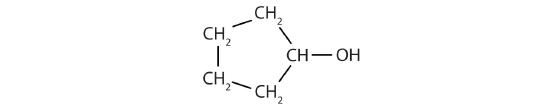
PROBLEM 6- Identify each alcohol as being 1o, 2o, or 3o.
a-
b-
Alkenes- Structures and Names
Concept Review Exercises
- Briefly identify the important distinctions between a saturated hydrocarbon and an unsaturated hydrocarbon.
- Briefly identify the important distinctions between an alkene and an alkane.
- Classify each compound as saturated or unsaturated. Identify each as an alkane, an alkene, or an alkyne.
-
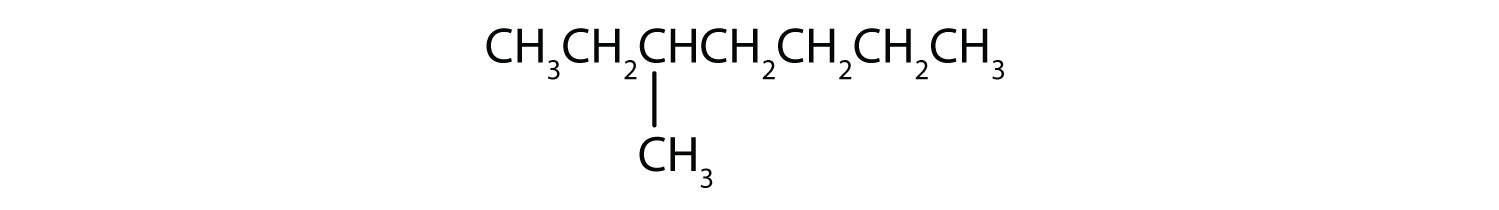
- CH3CH2C≡CCH3
-
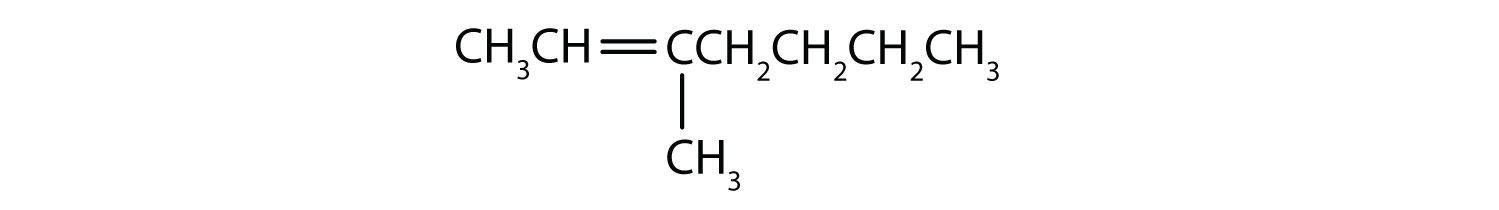
-
Answers
- Unsaturated hydrocarbons have double or triple bonds and are quite reactive; saturated hydrocarbons have only single bonds and are rather unreactive.
- An alkene has a double bond; an alkane has single bonds only.
-
- saturated; alkane
- unsaturated; alkyne
- unsaturated; alkene
Exercises
- Draw the structure for each compound.
- 2-methyl-2-pentene
- 2,3-dimethyl-1-butene
- cyclohexene
- Draw the structure for each compound.
- 5-methyl-1-hexene
- 3-ethyl-2-pentene
- 4-methyl-2-hexene
- Name each compound according to the IUPAC system.
-
- Name each compound according to the IUPAC system.
-
Answers
-
- 2-methyl-1-pentene
- 2-methyl-2-pentene
- 2,5-dimethyl-2-hexene
Cis-Trans Isomers (Geometric Isomers)
Concept Review Exercises
- What are cis-trans (geometric) isomers? What two types of compounds can exhibit cis-trans isomerism?
- Classify each compound as a cis isomer, a trans isomer, or neither.
-
-
Which compounds can exist as cis-trans (geometric) isomers? Draw the cis and trans isomers.
a. ClCH=CHBr
b. CH2=CBrCH3
c. (CH3)2C=CHCH2CH3
d. CH3CH=CHCH2CH3
e. CH2=CHCH2CH2CH3
f. CH3CH=CHCH2CH3
g. CH3CH2CH=CHCH2CH3
Answers
- Cis-trans isomers are compounds that have different configurations (groups permanently in different places in space) because of the presence of a rigid structure in their molecule. Alkenes and cyclic compounds can exhibit cis-trans isomerism.
-
- trans (the two hydrogen atoms are on opposite sides)
- cis (the two hydrogen atoms are on the same side, as are the two ethyl groups)
- cis (the two ethyl groups are on the same side)
- neither (flipping the bond does not change the molecule. There are no isomers for this molecule)
Exercises
- Draw the structures of the cis-trans isomers for each compound. Label them cis and trans. If no cis-trans isomers exist, write none.
- 2-bromo-2-pentene
- 3-hexene
- 4-methyl-2-pentene
- 1,1-dibromo-1-butene
- 2-butenoic acid (CH3CH=CHCOOH)
- Draw the structures of the cis-trans isomers for each compound. Label them cis and trans. If no cis-trans isomers exist, write none.
- 2,3-dimethyl-2-pentene
- 1,1-dimethyl-2-ethylcyclopropane
- 1,2-dimethylcyclohexane
- 5-methyl-2-hexene
- 1,2,3-trimethylcyclopropane
Answer
-
a: none. There are two distinct geometric isomers, but since there are there are four different groups off the double bond, these are both cis/trans isomers (they are technically E/Z isomers discussed elsewhere).b:

c:

d:
 e:
e:
Alkynes
Concept Review Exercises
- Briefly identify the important differences between an alkene and an alkyne. How are they similar?
- The alkene (CH3)2CHCH2CH=CH2 is named 4-methyl-1-pentene. What is the name of (CH3)2CHCH2C≡CH?
- Do alkynes show cis-trans isomerism? Explain.
Answers
- Alkenes have double bonds; alkynes have triple bonds. Both undergo addition reactions.
- 4-methyl-1-pentyne
- No; a triply bonded carbon atom can form only one other bond. It would have to have two groups attached to show cis-trans isomerism.
Exercises
- Draw the structure for each compound.
- acetylene
- 3-methyl-1-hexyne
- Draw the structure for each compound.
- 4-methyl-2-hexyne
- 3-octyne
- Name each alkyne.
- CH3CH2CH2C≡CH
- CH3CH2CH2C≡CCH3
Answers
-
- H–C≡C–H
-

-
- 1-pentyne
- 2-hexyne
Additional Exercises
-
You find an unlabeled jar containing a solid that melts at 48°C. It ignites readily and burns readily. The substance is insoluble in water and floats on the surface. Is the substance likely to be organic or inorganic?
-
Give the molecular formulas for methylcyclopentane, 2-methylpentane, and cyclohexane. Which are isomers?
-
What is wrong with each name? (Hint: first write the structure as if it were correct.) Give the correct name for each compound.
- 2-dimethylpropane
- 2,3,3-trimethylbutane
- 2,4-diethylpentane
- 3,4-dimethyl-5-propylhexane
-
What is the danger in swallowing a liquid alkane?
-
Distinguish between lighter and heavier liquid alkanes in terms of their effects on the skin.
-
Following is the line formula for an alkane. Draw its structure and give its name.
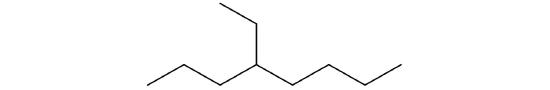
-
Write equations for the complete combustion of each compound.
- propane (a bottled gas fuel)
- octane (a typical hydrocarbon in gasoline).
-
The density of a gasoline sample is 0.690 g/mL. On the basis of the complete combustion of octane, calculate the amount in grams of carbon dioxide (CO2) and water (H2O) formed per gallon (3.78 L) of the gasoline when used in an automobile.
-
Draw the structures for the five isomeric hexanes (C6H14). Name each by the IUPAC system.
-
Indicate whether the structures in each set represent the same compound or isomers.
-
-
Consider the line-angle formulas shown here and answer the questions.
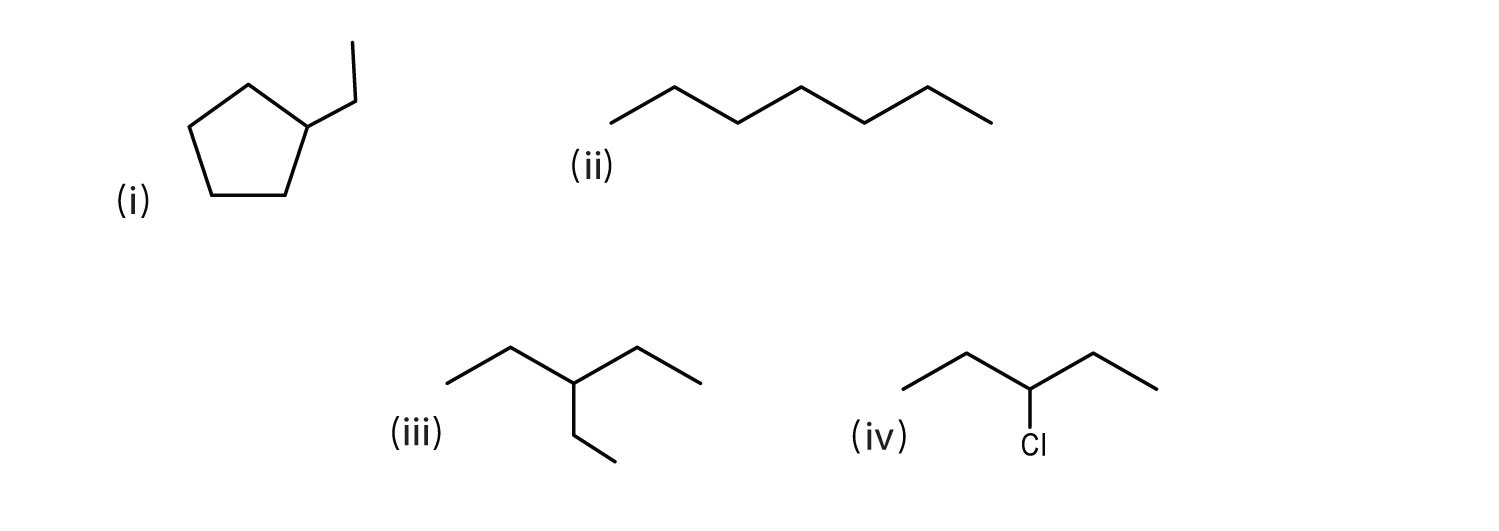
- Which pair of formulas represents isomers? Draw each structure.
- Which formula represents an alkyl halide? Name the compound and write its condensed structural formula.
- Which formula represents a cyclic alkane? Name the compound and draw its structure.
- What is the molecular formula of the compound represented by (i)?
-
Give the molecular formula for each compound.
-
-
What is wrong with each name? Draw the structure and give the correct name for each compound.
- 2-methyl-4-heptene
- 2-ethyl-2-hexene
- 2,2-dimethyl-3-pentene
-
What is wrong with each name?
- 2-bromobenzene
- 3,3-dichlorotoluene
- 1,4-dimethylnitrobenzene
-
The following are line-angle formulas for three compounds. Draw the structure and give the name for each.
-
-
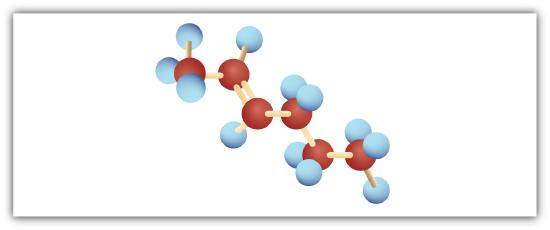
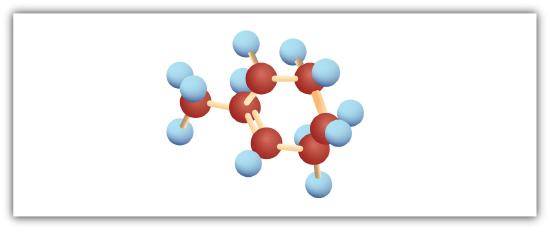
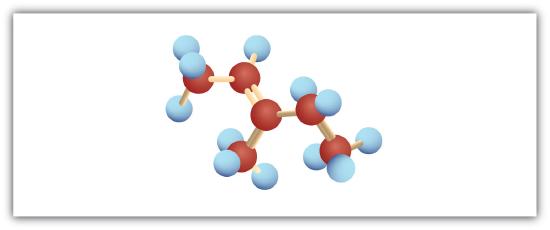
The following are ball-and-stick molecular models for three compounds (blue balls represent H atoms; red balls are C atoms). Write the condensed structural formula and give the name for each.
-
- Draw the structures of 9 compounds. Each of the 10 compounds should be an example of the functional group below with the correct # of carbon atoms specified.
a. An alkene with 4 carbons
b. An alcohol with 3 carbons
c. A thiol with 2 carbons.
e. An amine with 3 carbons. Label as primary, secondary, or tertiary.
f. A ketone with 3 carbons.
g. An aldehyde with 3 carbons.
h. A carboxylic acid with 3 carbons.
i. An ester with 3 carbons.
j. An amide with 2 carbons.
Answers
-
organic
-
- Two numbers are needed to indicate two substituents; 2,2-dimethylpropane.
- The lowest possible numbers were not used; 2,2,3-trimethylbutane.
- An ethyl substituent is not possible on the second carbon atom; 3,5-dimethylheptane.
- A propyl substituent is not possible on the fifth carbon atom; 3,4,5-trimethyloctane.
-
Lighter alkanes wash away protective skin oils; heavier alkanes form a protective layer.
-
- C3H8 + 5O2 → 3CO2 + 4H2O
- 2C8H18 + 25O2 → 16CO2 + 18H2O
-
CH3CH2CH2CH2CH2CH3; hexane




-
- ii and iii; CH3CH2CH2CH2CH2CH2CH3 and

- iv; 3-chloropentane; CH3CH2CHClCH2CH3
- i; ethylcyclopentane;

- C7H14
13.
- number not needed
- can’t have two groups on one carbon atom on a benzene ring
- can’t have a substituent on the same carbon atom as the nitro group
15.- CH3CH=CHCH2CH2CH3; 2-hexene
-

-

- ii and iii; CH3CH2CH2CH2CH2CH2CH3 and