1.2: The Classification of Matter
- Page ID
- 165409
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)
Skills to Develop
- Use physical and chemical properties, including phase, to describe matter.
- Identify a sample of matter as an element, a compound, or a mixture.
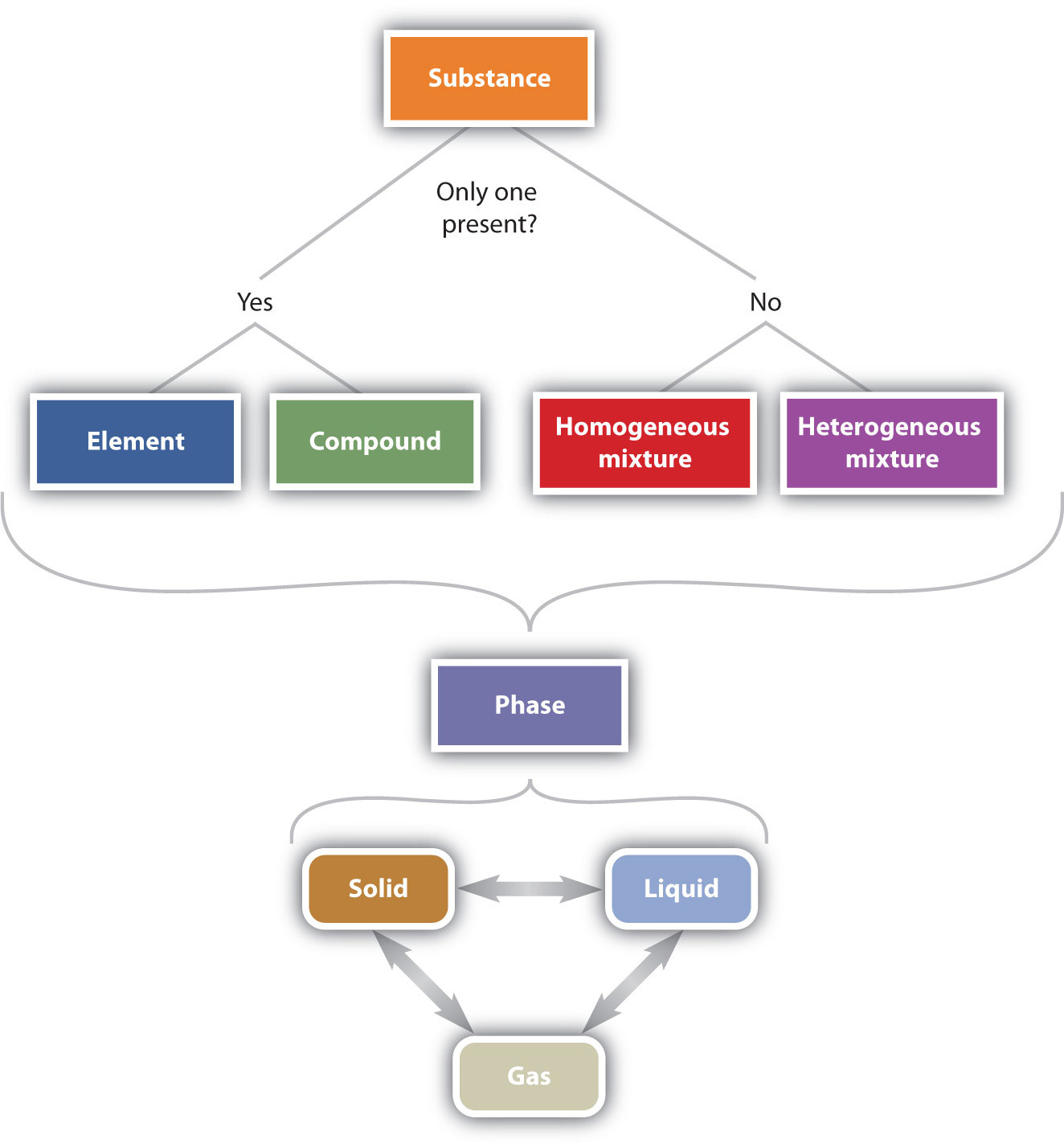
Part of understanding matter is being able to describe it. One way chemists describe matter is to assign different kinds of properties to different categories.
Elements and Compounds
Any sample of matter that has the same physical and chemical properties throughout the sample is called a substance. There are two types of substances. A substance that cannot be broken down into chemically simpler components is called an element. Aluminum, which is used in soda cans and is represented by the symbol Al, is an element. A substance that can be broken down into chemically simpler components (because it consists of more than one element) is called a compound. Water is a compound composed of the elements hydrogen and oxygen and is described by the chemical formula, H2O. Today, there are about 118 elements in the known universe. In contrast, scientists have identified tens of millions of different compounds to date.
Sometimes the word pure is used to describe a substance, but this is not absolutely necessary. By definition, any single substance, element or compound is pure.
The smallest part of an element that maintains the identity of that element is called an atom. Atoms are extremely tiny; to make a line of iron atoms that is 1 inch long, you would need approximately 217 million iron atoms. The smallest part of a compound that maintains the identity of that compound is called a molecule. Molecules are composed of two or more different atoms that are attached together and behave as a unit. Scientists usually work with millions and millions of atoms and molecules at a time. When a scientist is working with large numbers of atoms or molecules at a time, the scientist is studying the macroscopic viewpoint of the universe. However, scientists can also describe chemical events on the level of individual atoms or molecules, which is referred to as the microscopic viewpoint. We will see examples of both macroscopic and microscopic viewpoints throughout this book (Figure \(\PageIndex{1}\)).

Mixtures
A material composed of two or more substances is a mixture. In a mixture, the individual substances maintain their chemical identities. Many mixtures are obvious combinations of two or more substances, such as a mixture of sand and water. Such mixtures are called heterogeneous mixtures. In some mixtures, the components are so intimately combined that they act like a single substance (even though they are not). Mixtures with a consistent or uniform composition throughout are called homogeneous mixtures (or solutions). For example, when sugar is dissolved in water to form a liquid solution, the individual properties of the components cannot be distinguished. Other examples or homogenous mixtures include solid solutions, like the metal alloy steel, and gaseous solutions, like air which is a mixture of mainly nitrogen and oxygen.
Example \(\PageIndex{1}\)
How would a chemist categorize each example of matter?
- saltwater
- soil
- water
- oxygen
- Answer a
-
Saltwater acts as if it were a single substance even though it contains two substances—salt and water. Saltwater is a homogeneous mixture, or a solution.
- Answer b
-
Soil is composed of small pieces of a variety of materials, so it is a heterogeneous mixture.
- Answer c
-
Water is a substance; more specifically, because water is composed of hydrogen and oxygen, it is a compound.
- Answer d
-
Oxygen, a substance, is an element.
Exercise \(\PageIndex{1}\)
How would a chemist categorize each example of matter?
- coffee
- hydrogen
- an egg
- Answer
-
a. breakfast coffee is a homogeneous mixture or a solution. b. element c. heterogeneous mixture.
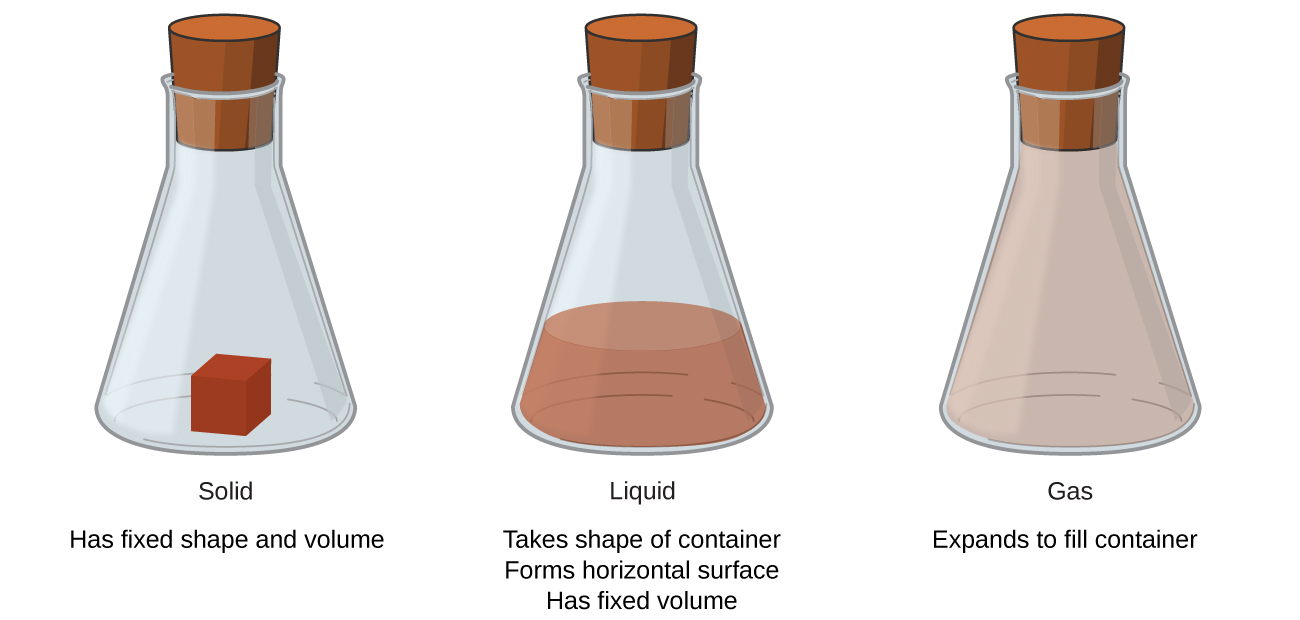
Phases or Physical Properties of Matter
Concept Review Exercises
-
Explain the differences between the physical properties of matter and the chemical properties of matter.
-
What is the difference between a heterogeneous mixture and a homogeneous mixture? Give an example of each.
-
Give at least two examples of a phase change and state the phases involved in each.
Answers
-
Physical properties describe the existence of matter, and chemical properties describe how substances change into other substances.
-
A heterogeneous mixture is obviously a mixture, such as dirt; a homogeneous mixture behaves like a single substance, such as saltwater.
-
solid to liquid (melting) and liquid to gas (boiling) (answers will vary)
Key Takeaways
- Matter can be described with both physical properties and chemical properties.
- Matter can be identified as an element, a compound, or a mixture.
Exercises
-
Does each statement refer to a chemical property or a physical property?
- Balsa is a very light wood.
- If held in a flame, magnesium metal burns in air.
- Mercury has a density of 13.6 g/mL.
- Human blood is red.
-
Does each statement refer to a chemical property or a physical property?
- The elements sodium and chlorine can combine to make table salt.
- The metal tungsten does not melt until its temperature exceeds 3,000°C.
- The ingestion of ethyl alcohol can lead to disorientation and confusion.
- The boiling point of isopropyl alcohol, which is used to sterilize cuts and scrapes, is lower than the boiling point of water.
-
Define element. How does it differ from a compound?
-
Define compound. How does it differ from an element?
-
Give two examples of a heterogeneous mixture.
-
Give two examples of a homogeneous mixture.
-
Identify each substance as an element, a compound, a heterogeneous mixture, or a solution.
- xenon, a substance that cannot be broken down into chemically simpler components
- blood, a substance composed of several types of cells suspended in a salty solution called plasma
- water, a substance composed of hydrogen and oxygen
-
Identify each substance as an element, a compound, a heterogeneous mixture, or a solution.
- sugar, a substance composed of carbon, hydrogen, and oxygen
- hydrogen, the simplest chemical substance
- dirt, a combination of rocks and decaying plant matter
-
Identify each substance as an element, a compound, a heterogeneous mixture, or a solution.
- air, primarily a mixture of nitrogen and oxygen
- ringer’s lactate, a standard fluid used in medicine that contains salt, potassium, and lactate compounds all dissolved in sterile water
- tartaric acid, a substance composed of carbon, hydrogen, and oxygen
-
Identify each material as an element, a compound, a heterogeneous mixture, or a solution.
- equal portions of salt and sand placed in a beaker and shaken up
- a combination of beeswax dissolved in liquid hexane
- hydrogen peroxide, a substance composed of hydrogen and oxygen
-
What word describes each phase change?
- solid to liquid
- liquid to gas
- solid to gas
-
What word describes each phase change?
- liquid to solid
- gas to liquid
- gas to solid
Answers
-
- physical property
- chemical property
- physical property
- physical property
2.
- chemical property
- physical property
- chemical property
- physical property
-
An element is a substance that cannot be broken down into chemically simpler components. Compounds can be broken down into simpler substances.
4. A compound is composed of two or more elements combined in a fixed ratio. An element is the simplest chemical substance.
-
a salt and pepper mix and a bowl of cereal (answers will vary)
6. vinegar and rubbing alcohol (answers will vary)
-
- element
- heterogeneous mixture
- compound
8.
- compound
- element
- heterogeneous mixture
-
- solution
- solution
- compound
10.
- heterogeneous mixture
- solution
- compound
11.
- melting or fusion
- boiling or evaporation
- sublimation
12.
- freezing
- condensation
- deposition