26.4 Application: Natural and Synthetic Rubber
- Page ID
- 30978
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Rubber is an example of an elastomer type polymer, where the polymer has the ability to return to its original shape after being stretched or deformed. The rubber polymer is coiled when in the resting state. The elastic properties arise from the its ability to stretch the chains apart, but when the tension is released the chains snap back to the original position. The majority of rubber polymer molecules contain at least some units derived from conjugated diene monomers (see Polymerization of Conjugated Dienes). Such conjugated diene monomers have a constructive backbone of at least four carbon atoms with a double-single-double bond reactive core (C=C-C=C ). Most if not practically all such dienes undergo 1,4-addition to the polymer chain, where 1 and 4 refer to the 1st and 4th carbons of the backbone unit, which become single-bonded to the rest of the polymer chain. The diene's double bonds turn into single bonds, and the single bond between them turns into a Z or E configured double bond, depending on the polymerization conditions. The unit's backbone thus becomes like this (-C-C=C-C-). Rubber gets its elasticity when the formed double bond gets the Z configuration. For 1,3-butadiene, Z is equivalent to a cis and E is equivalent to a trans configuration.
Natural Rubber
Natural rubber is an addition polymer that is obtained as a milky white fluid known as latex from a tropical rubber tree. Natural rubber is from the monomer isoprene (2-methyl-1,3-butadiene), which is a conjugated diene hydrocarbon as mentioned above. In natural rubber, most of the double fonds formed in the polymer chain have the Z configuration, resulting in natural rubber's elastomer qualities.
Charles Goodyear accidentally discovered that by mixing sulfur and rubber, the properties of the rubber improved in being tougher, resistant to heat and cold, and increased in elasticity. This process was later called vulcanization after the Roman god of fire. Vulcanization causes shorter chains to cross link through the sulfur to longer chains. The development of vulcanized rubber for automobile tires greatly aided this industry.
Synthetic Rubber
Important conjugated dienes used in synthetic rubbers include isoprene (2-methyl-1,3-butadiene), 1,3-butadiene, and chloroprene (2-chloro-1,3-butadiene). Polymerized 1,3-butadiene is mostly referred to simply as polybutadiene. Polymerized chloroprene was developed by DuPont and given the trade name Neoprene.
In a number of cases, monomers which are not dienes are also used for certain types of synthetic rubber, often copolymerized with dienes. Some of the most commercially important addition polymers are the copolymers. These are polymers made by polymerizing a mixture of two or more monomers. An example is styrene-butadiene rubber (SBR) - which is a copolymer of 1,3-butadiene and styrene which is mixed in a 3 to 1 ratio, respectively.
SBR rubber was developed during World War II when important supplies of natural rubber were cut off. SBR is more resistant to abrasion and oxidation than natural rubber and can also be vulcanized. More than 40% of the synthetic rubber production is SBR and is used in tire production. A tiny amount is used for bubble-gum in the unvulcanized form.
Nitrile rubber is copolymerized from butadiene and acrylonitrile (H2C=CH-CN). Butyl rubber is copolymerized from isobutylene [which is methylpropene H2C=C(CH3)2 ] and a small percentage of isoprene. Silicone rubber and other compounds, chemically called polysiloxanes, are not from conjugated dienes but have repeating units like -O-SiR2- where R is some organic radical group like methyl. There is a separate page on Silicone Polymers.
Conjugated dienes (alkenes with two double bonds and a single bond in between) can be polymerized to form important compounds like rubber. This takes place, in different forms, both in nature and in the laboratory. Interactions between double bonds on multiple chains leads to cross-linkage which creates elasticity within the compound.
Polymerization of 1,3-Butadiene
For rubber compounds to be synthesized, 1,3-butadiene must be polymerized. Below is a simple illustration of how this compound is formed into a chain. The 1,4 polymerization is much more useful to polymerization reactions.

Above, the green structures represent the base units of the polymers that are synthesized and the red represents the bonds between these units which form these polymers. Whether the 1,3 product or the 1,4 product is formed depends on whether the reaction is thermally or kinetically controlled.
Synthetic Rubber
The most important synthetic rubber is Neoprene which is produced by the polymerization of 2-chloro-1,3-butadiene.
.png?revision=1&size=bestfit&width=350&height=133)
In this illustration, the dashed lines represent repetition of the same base units, so both the products and reactants are polymers. The reaction proceeds with a mechanism similar to the Friedel-Crafts mechanism. Cross-linkage between the chlorine atom of one chain and the double bond of another contributes to the overall elasticity of neoprene. This cross-linkage occurs as the chains lie next to each other at random angles, and the attractions between double bonds prevent them from sliding back and forth.
Natural Rubber
The synthesis of rubber in nature in somewhat similar the artificial synthesis of rubber except that it takes place within a plant. Instead of the 2-chloro-1,3-butadiene used in the synthesis of neoprene, natural rubber is synthesized from 2-methyl-1,3-butadiene. As an electrophile, the plant synthesizes the pyrophosphate 3-methyl-3-butenyl pyrophosphate is from phosphoric acid and 3-methyl-3-buten-1-ol. This pyrophosphate then catalyzes the reaction that leads to natural rubber.
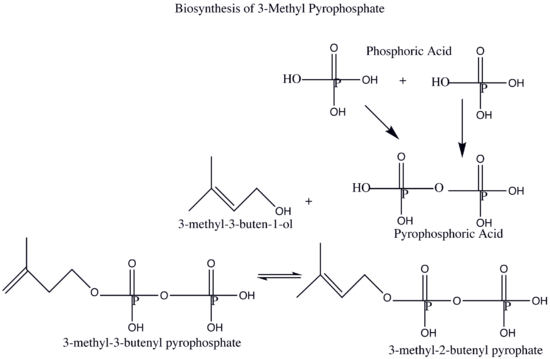
The 3-methyl-3-butenyl pyrophosphate (OPP) is then used in the polymerization of natural rubber as it pulls electrons off 2-methyl-1,3-butadiene (see questions section for this process.)
Outside links
- "Dienes," http://en.wikipedia.org/wiki/Diene
- "Rubber," http://en.wikipedia.org/wiki/Rubber
- "Neoprene," http://en.wikipedia.org/wiki/Neoprene
References
- Vollhardt, Peter, and Neil E. Schore. Organic Chemistry: Structure and Function. New York: W. H. Freeman & Company, 2007.
- Buehr, Walter. Rubber: Natural and Synthetic. Morrow, 1964.
Problem
Draw out the mechanism for the natural synthesis of rubber from 3-methyl-3-butenyl pyrophosphate and 2-methyl-1,3-butadiene. Show the movement of electrons with arrows.
Answer
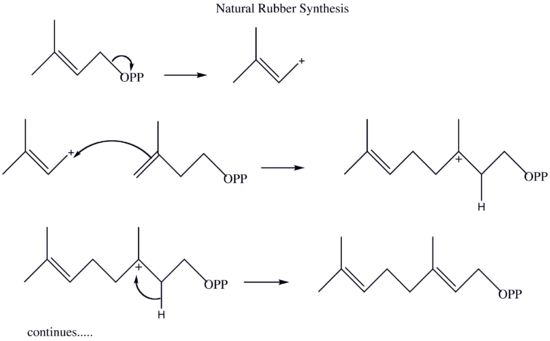
Contributors
- Charles Ophardt, Professor Emeritus, Elmhurst College; Virtual Chembook
- Layne A. Morsch - University of Illinois Springfield
- User:H Padleckas


