22.1 Introduction to Nucleophilic Acyl Substitution
- Page ID
- 28411
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Background and Properties
The important classes of organic compounds known as alcohols, phenols, ethers, amines and halides consist of alkyl and/or aryl groups bonded to hydroxyl, alkoxyl, amino and halo substituents respectively. If these same functional groups are attached to an acyl group (RCO–) their properties are substantially changed, and they are designated as carboxylic acid derivatives. Carboxylic acids have a hydroxyl group bonded to an acyl group, and their functional derivatives are prepared by replacement of the hydroxyl group with substituents, such as halo, alkoxyl, amino and acyloxy. Some examples of these functional derivatives were displayed earlier.
The following table lists some representative derivatives and their boiling points. An aldehyde and ketone of equivalent molecular weight are also listed for comparison. Boiling points are given for 760 torr (atmospheric pressure), and those listed as a range are estimated from values obtained at lower pressures. As noted earlier, the relatively high boiling point of carboxylic acids is due to extensive hydrogen bonded dimerization. Similar hydrogen bonding occurs between molecules of 1º and 2º-amides (amides having at least one N–H bond), and the first three compounds in the table serve as hydrogen bonding examples.
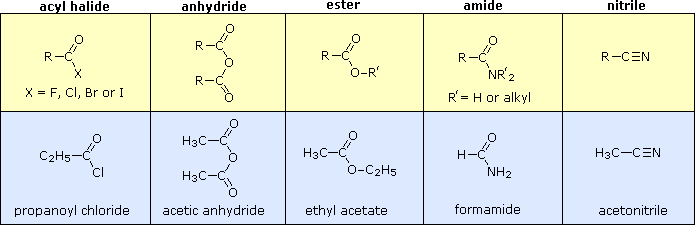
Other functional group combinations with the carbonyl group can be prepared from carboxylic acids, and are usually treated as related derivatives. Five common classes of these carboxylic acid derivatives are listed in the following table. Although nitriles do not have a carbonyl group, they are included here because the functional carbon atoms all have the same oxidation state. The top row (yellow shaded) shows the general formula for each class, and the bottom row (light blue) gives a specific example of each. As in the case of amines, amides are classified as 1º, 2º or 3º, depending on the number of alkyl groups bonded to the nitrogen.
Acyl Group Substitution
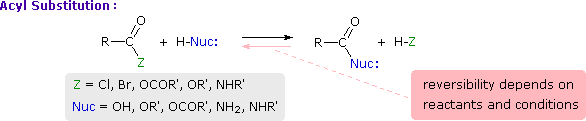
This is probably the single most important reaction of carboxylic acid derivatives. The overall transformation is defined by the following equation, and may be classified either as nucleophilic substitution at an acyl group or as acylation of a nucleophile. For certain nucleophilic reagents the reaction may assume other names as well. If Nuc-H is water the reaction is often called hydrolysis, if Nuc–H is an alcohol the reaction is called alcoholysis, and for ammonia and amines it is called aminolysis.
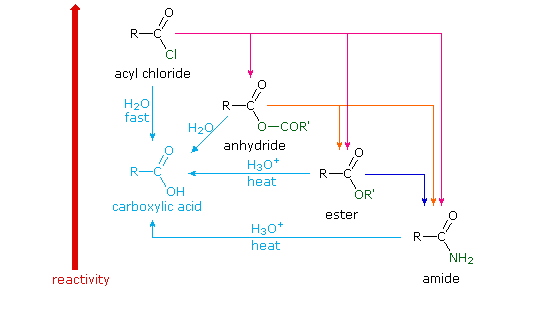
Different carboxylic acid derivatives have very different reactivities, acyl chlorides and bromides being the most reactive and amides the least reactive, as noted in the following qualitatively ordered list. The change in reactivity is dramatic. In homogeneous solvent systems, reaction of acyl chlorides with water occurs rapidly, and does not require heating or catalysts. Amides, on the other hand, react with water only in the presence of strong acid or base catalysts and external heating.
Reactivity: acyl halides > anhydrides >> esters ≈ acids >> amides
Because of these differences, the conversion of one type of acid derivative into another is generally restricted to those outlined in the following diagram. Methods for converting carboxylic acids into these derivatives were shown in a previous section, but the amide and anhydride preparations were not general and required strong heating. A better and more general anhydride synthesis can be achieved from acyl chlorides, and amides are easily made from any of the more reactive derivatives. Specific examples of these conversions will be displayed by clicking on the product formula. The carboxylic acids themselves are not an essential part of this diagram, although all the derivatives shown can be hydrolyzed to the carboxylic acid state (light blue formulas and reaction arrows). Base catalyzed hydrolysis produces carboxylate salts.
Before proceeding further, it is important to review the general mechanism by means of which all these acyl transfer or acylation reactions take place. Indeed, an alert reader may well be puzzled by the facility of these nucleophilic substitution reactions. After all, it was previously noted that halogens bonded to sp2 or sp hybridized carbon atoms do not usually undergo substitution reactions with nucleophilic reagents. Furthermore, such substitution reactions of alcohols and ethers are rare, except in the presence of strong mineral acids. Clearly, the mechanism by which acylation reactions occur must be different from the SN1 and SN2 procedures described earlier.
In any substitution reaction two things must happen. The bond from the substrate to the leaving group must be broken, and a bond to the replacement group must be formed. The timing of these events may vary with the reacting system. In nucleophilic substitution reactions of alkyl compounds examples of bond-breaking preceding bond-making (the SN1 mechanism), and of bond-breaking and bond-making occurring simultaneously (the SN2 mechanism) were observed. On the other hand, for most cases of electrophilic aromatic substitution bond-making preceded bond-breaking.
As illustrated in the following diagram, acylation reactions generally take place by an addition-elimination process in which a nucleophilic reactant bonds to the electrophilic carbonyl carbon atom to create a tetrahedral intermediate. This tetrahedral intermediate then undergoes an elimination to yield the products. In this two-stage mechanism bond formation occurs before bond cleavage, and the carbonyl carbon atom undergoes a hybridization change from sp2 to sp3 and back again. The facility with which nucleophilic reagents add to a carbonyl group was noted earlier for aldehydes and ketones.
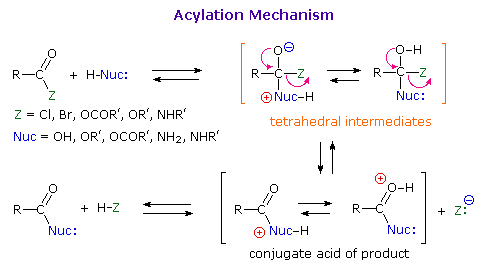
Acid and base-catalyzed variations of this mechanism will be displayed in turn as the "Mechanism Toggle" button is clicked. Also, a specific example of acyl chloride formation from the reaction of a carboxylic acid with thionyl chloride will be shown. The number of individual steps in these mechanisms vary, but the essential characteristic of the overall transformation is that of addition followed by elimination. Acid catalysts act to increase the electrophilicity of the acyl reactant; whereas, base catalysts act on the nucleophilic reactant to increase its reactivity. In principle all steps are reversible, but in practice many reactions of this kind are irreversible unless changes in the reactants and conditions are made. The acid-catalyzed formation of esters from carboxylic acids and alcohols, described earlier, is a good example of a reversible acylation reaction, the products being determined by the addition or removal of water from the system. The reaction of an acyl chloride with an alcohol also gives an ester, but this conversion cannot be reversed by adding HCl to the reaction mixture.
Contributors
William Reusch, Professor Emeritus (Michigan State U.), Virtual Textbook of Organic Chemistry
- Layne A. Morsch - University of Illinois Springfield


