9.4: Physical Properties of Alcohols, Ethers and Epoxides
- Page ID
- 28198
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Physical properties of alcohols
Boiling Points
The chart below shows the boiling points of the following simple primary alcohols with up to 4 carbon atoms:
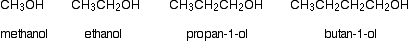
These boiling points are compared with those of the equivalent alkanes (methane to butane) with the same number of carbon atoms.
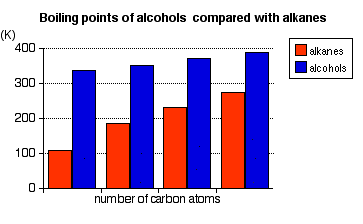
Notice that:
- The boiling point of an alcohol is always significantly higher than that of the analogous alkane.
- The boiling points of the alcohols increase as the number of carbon atoms increases.
The patterns in boiling point reflect the patterns in intermolecular attractions.
Hydrogen bonding
Hydrogen bonding occurs between molecules in which a hydrogen atom is attached to a strongly electronegative element: fluorine, oxygen or nitrogen. In the case of alcohols, hydrogen bonds occur between the partially-positive hydrogen atoms and lone pairs on oxygen atoms of other molecules.
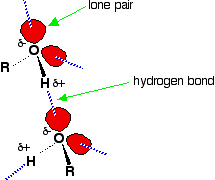
The hydrogen atoms are slightly positive because the bonding electrons are pulled toward the very electronegative oxygen atoms. In alkanes, the only intermolecular forces are van der Waals dispersion forces. Hydrogen bonds are much stronger than these, and therefore it takes more energy to separate alcohol molecules than it does to separate alkane molecules. This the main reason for higher boiling points in alcohols.
The effect of van der Waals forces
- Boiling points of the alcohols: Hydrogen bonding is not the only intermolecular force alcohols experience. There are also van der Waals dispersion forces and dipole-dipole interactions. The hydrogen bonding and dipole-dipole interactions are much the same for all alcohols, but dispersion forces increase as the alcohols get bigger. These attractions get stronger as the molecules get longer and have more electrons. This increases the sizes of the temporary dipoles formed. This is why the boiling points increase as the number of carbon atoms in the chains increases. It takes more energy to overcome the dispersion forces, and thus the boiling points rise.
- Comparison between alkanes and alcohols: Even without any hydrogen bonding or dipole-dipole interactions, the boiling point of the alcohol would be higher than the corresponding alkane with the same number of carbon atoms.
Compare ethane and ethanol:
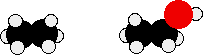
Ethanol is a longer molecule, and the oxygen atom brings with it an extra 8 electrons. Both of these increase the size of the van der Waals dispersion forces, and subsequently the boiling point. A more accurate measurement of the effect of the hydrogen bonding on boiling point would be a comparison of ethanol with propane rather than ethane. The lengths of the two molecules are more similar, and the number of electrons is exactly the same.
Solubility of alcohols in water
Small alcohols are completely soluble in water; mixing the two in any proportion generates a single solution. However, solubility decreases as the length of the hydrocarbon chain in the alcohol increases. At four carbon atoms and beyond, the decrease in solubility is noticeable; a two-layered substance may appear in a test tube when the two are mixed.
Consider ethanol as a typical small alcohol. In both pure water and pure ethanol the main intermolecular attractions are hydrogen bonds.
In order to mix the two, the hydrogen bonds between water molecules and the hydrogen bonds between ethanol molecules must be broken. Energy is required for both of these processes. However, when the molecules are mixed, new hydrogen bonds are formed between water molecules and ethanol molecules.
The energy released when these new hydrogen bonds form approximately compensates for the energy needed to break the original interactions. In addition, there is an increase in the disorder of the system, an increase in entropy. This is another factor in deciding whether chemical processes occur. Consider a hypothetical situation involving 5-carbon alcohol molecules.
The hydrocarbon chains are forced between water molecules, breaking hydrogen bonds between those water molecules. The -OH ends of the alcohol molecules can form new hydrogen bonds with water molecules, but the hydrocarbon "tail" does not form hydrogen bonds. This means that many of the original hydrogen bonds being broken are never replaced by new ones.
In place of those original hydrogen bonds are merely van der Waals dispersion forces between the water and the hydrocarbon "tails." These attractions are much weaker, and unable to furnish enough energy to compensate for the broken hydrogen bonds. Even allowing for the increase in disorder, the process becomes less feasible. As the length of the alcohol increases, this situation becomes more pronounced, and thus the solubility decreases.
Comparisons of Alcohols and Ethers
Ether molecules have no hydrogen atom on the oxygen atom (that is, no OH group). Therefore there is no intermolecular hydrogen bonding between ether molecules, and ethers therefore have quite low boiling points for a given molar mass. Indeed, ethers have boiling points about the same as those of alkanes of comparable molar mass and much lower than those of the corresponding alcohols (Table 14.4 "Comparison of Boiling Points of Alkanes, Alcohols, and Ethers").
Table 14.4 Comparison of Boiling Points of Alkanes, Alcohols, and Ethers
| Condensed Structural Formula | Name | Molar Mass | Boiling Point (°C) | Intermolecular Hydrogen Bonding in Pure Liquid? |
|---|---|---|---|---|
| CH3CH2CH3 | propane | 44 | –42 | no |
| CH3OCH3 | dimethyl ether | 46 | –25 | no |
| CH3CH2OH | ethyl alcohol | 46 | 78 | yes |
| CH3CH2CH2CH2CH3 | pentane | 72 | 36 | no |
| CH3CH2OCH2CH3 | diethyl ether | 74 | 35 | no |
| CH3CH2CH2CH2OH | butyl alcohol | 74 | 117 | yes |
Ether molecules do have an oxygen atom, however, and engage in hydrogen bonding with water molecules. Consequently, an ether has about the same solubility in water as the alcohol that is isomeric with it. For example, dimethyl ether and ethanol (both having the molecular formula C2H6O) are completely soluble in water, whereas diethyl ether and 1-butanol (both C4H10O) are barely soluble in water (8 g/100 mL of water).
Contributors
Jim Clark (Chemguide.co.uk)


