6.2: The Brønsted-Lowry Scheme
- Last updated
- Save as PDF
- Page ID
- 43431
Learning Objectives
- Explain the characterization of aqueous solutions as acidic, basic, or neutral
- To know the relationship between acid or base strength and the magnitude of \(K_a\), \(K_b\), \(pK_a\), and \(pK_b\).
- Express hydronium and hydroxide ion concentrations on the pH and pOH scales
- Perform calculations relating pH and pOH
We now turn our attention to acid–base reactions to see how the concepts of chemical equilibrium and equilibrium constants can deepen our understanding of this kind of chemical behavior. We begin with a qualitative description of acid–base equilibria in terms of the Brønsted–Lowry model and then proceed to a quantitative description the following sections.
Free Hydrogen Ions do not Exist in Water
Owing to the overwhelming excess of \(H_2O\) molecules in aqueous solutions, a bare hydrogen ion has no chance of surviving in water. The hydrogen ion in aqueous solution is no more than a proton, a bare nucleus. Although it carries only a single unit of positive charge, this charge is concentrated into a volume of space that is only about a hundred-millionth as large as the volume occupied by the smallest atom. (Think of a pebble sitting in the middle of a sports stadium!) The resulting extraordinarily high charge density of the proton strongly attracts it to any part of a nearby atom or molecule in which there is an excess of negative charge. In the case of water, this will be the lone pair (unshared) electrons of the oxygen atom; the tiny proton will be buried within the lone pair and will form a shared-electron (coordinate) bond with it, creating a hydronium ion, \(H_3O^+\). In a sense, \(H_2O\) is acting as a base here, and the product \(H_3O^+\) is the conjugate acid of water:
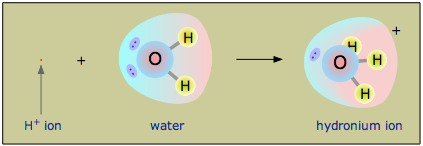
Although other kinds of dissolved ions have water molecules bound to them more or less tightly, the interaction between H+ and \(H_2O\) is so strong that writing “H+(aq)” hardly does it justice, although it is formally correct. The formula \(H_3O^+\) more adequately conveys the sense that it is both a molecule in its own right, and is also the conjugate acid of water. However, the equation
\[\ce{HA → H^{+} + A^{–}} \nonumber\]
is so much easier to write that chemists still use it to represent acid-base reactions in contexts in which the proton donor-acceptor mechanism does not need to be emphasized. Thus it is permissible to talk about “hydrogen ions” and use the formula H+ in writing chemical equations as long as you remember that they are not to be taken literally in the context of aqueous solutions.
Interestingly, experiments indicate that the proton does not stick to a single \(H_2O\) molecule, but changes partners many times per second. This molecular promiscuity, a consequence of the uniquely small size and mass the proton, allows it to move through the solution by rapidly hopping from one \(H_2O\) molecule to the next, creating a new \(H_3O^+\) ion as it goes. The overall effect is the same as if the \(H_3O^+\) ion itself were moving. Similarly, a hydroxide ion, which can be considered to be a “proton hole” in the water, serves as a landing point for a proton from another \(H_2O\) molecule, so that the OH– ion hops about in the same way.
The hydronium ion is an important factor when dealing with chemical reactions that occur in aqueous solutions. Because hydronium and hydroxide ions can “move without actually moving” and thus without having to plow their way through the solution by shoving aside water molecules as do other ions, solutions which are acidic or alkaline have extraordinarily high electrical conductivities. The hydronium ion has a trigonal pyramidal geometry and is composed of three hydrogen atoms and one oxygen atom. There is a lone pair of electrons on the oxygen giving it this shape. The bond angle between the atoms is 113 degrees.
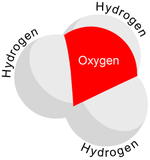
As H+ ions are formed, they bond with \(H_2O\) molecules in the solution to form \(H_3O^+\) (the hydronium ion). This is because hydrogen ions do not exist in aqueous solutions, but take the form the hydronium ion, \(H_3O^+\). A reversible reaction is one in which the reaction goes both ways. In other words, the water molecules dissociate while the OH- ions combine with the H+ ions to form water. Water has the ability to attract H+ ions because it is a polar molecule. This means that it has a partial charge, in this case the charge is negative. The partial charge is caused by the fact that oxygen is more electronegative than hydrogen. This means that in the bond between hydrogen and oxygen, oxygen "pulls" harder on the shared electrons thus causing a partial negative charge on the molecule and causing it to be attracted to the positive charge of H+ to form hydronium. Another way to describe why the water molecule is considered polar is through the concept of dipole moment. The electron geometry of water is tetrahedral and the molecular geometry is bent. This bent geometry is asymmetrical, which causes the molecule to be polar and have a dipole moment, resulting in a partial charge.
The pH Scale
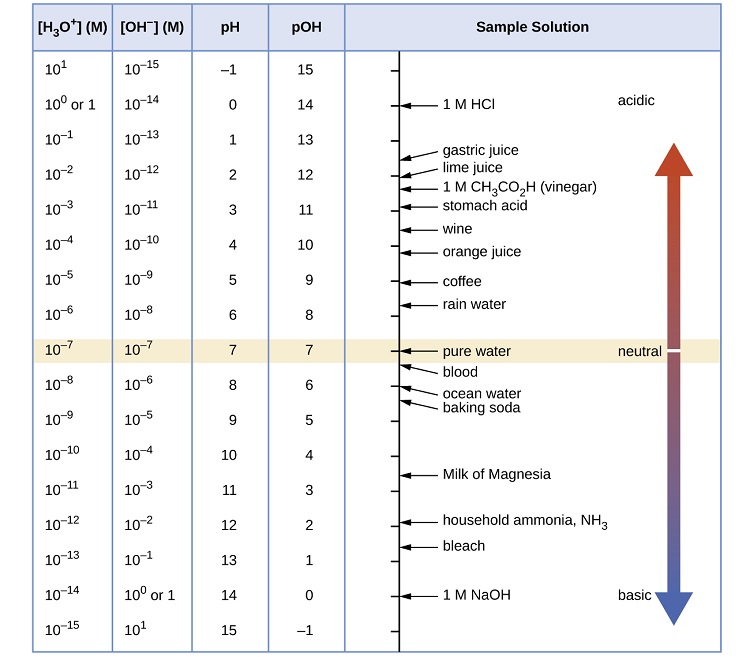
As discussed earlier, hydronium and hydroxide ions are present both in pure water and in all aqueous solutions, and their concentrations are inversely proportional as determined by the ion product of water (Kw). The concentrations of these ions in a solution are often critical determinants of the solution’s properties and the chemical behaviors of its other solutes, and specific vocabulary has been developed to describe these concentrations in relative terms. A solution is neutral if it contains equal concentrations of hydronium and hydroxide ions; acidic if it contains a greater concentration of hydronium ions than hydroxide ions; and basic if it contains a lesser concentration of hydronium ions than hydroxide ions.
A common means of expressing quantities, the values of which may span many orders of magnitude, is to use a logarithmic scale. One such scale that is very popular for chemical concentrations and equilibrium constants is based on the p-function, defined as shown where “X” is the quantity of interest and “log” is the base-10 logarithm:
\[\mathrm{pX=−\log X} \label{\(\PageIndex{1}\)}\]
The pH of a solution is therefore defined as shown here, where [H3O+] is the molar concentration of hydronium ion in the solution:
\[\mathrm{pH=-\log[H_3O^+]}\label{\(\PageIndex{2}\)}\]
Rearranging this equation to isolate the hydronium ion molarity yields the equivalent expression:
\[\mathrm{[H_3O^+]=10^{−pH}}\label{\(\PageIndex{3}\)}\]
Likewise, the hydroxide ion molarity may be expressed as a p-function, or pOH:
\[\mathrm{pOH=-\log [OH^−]}\label{\(\PageIndex{4}\)}\]
or
\[\mathrm{[OH^-]=10^{−pOH}} \label{\(\PageIndex{5}\)}\]
Finally, the relation between these two ion concentration expressed as p-functions is easily derived from the \(K_w\) expression:
\[ \begin{align*} K_\ce{w} &=\ce{[H_3O^+][OH^- ]} \\[4pt] -\log K_\ce{w} &= -\log([H_3O^+][OH^−]) \\[4pt] &=-\log[H_3O^+] + -\log[OH^-] \\[4pt] p \mathit{K}_w &=pH + pOH \end{align*}\]
At 25 °C, the value of \(K_w\) is \(1.0 \times 10^{−14}\) and so:
\[\mathrm{14.00=pH + pOH} \label{\(\PageIndex{9}\)}\]
The hydronium ion molarity in pure water (or any neutral solution) is \( 1.0 \times 10^{-7}\; M\) at 25 °C. The pH and pOH of a neutral solution at this temperature are therefore:
\[\mathrm{pH=-\log[H_3O^+]=-\log(1.0\times 10^{−7}) = 7.00} \label{\(\PageIndex{1}\)0}\]
\[\mathrm{pOH=-\log[OH^−]=-\log(1.0\times 10^{−7}) = 7.00} \label{\(\PageIndex{1}\)1}\]
And so, at this temperature, acidic solutions are those with hydronium ion molarities greater than \( 1.0 \times 10^{-7}\; M\) and hydroxide ion molarities less than \( 1.0 \times 10^{-7}\; M\) (corresponding to pH values less than 7.00 and pOH values greater than 7.00). Basic solutions are those with hydronium ion molarities less than \( 1.0 \times 10^{-7}\; M\) and hydroxide ion molarities greater than \( 1.0 \times 10^{-7}\; M\) (corresponding to pH values greater than 7.00 and pOH values less than 7.00).
Since the autoionization constant \(K_w\) is temperature dependent, these correlations between pH values and the acidic/neutral/basic adjectives will be different at temperatures other than 25 °C. For example, the hydronium molarity of pure water at 80 °C is 4.9 × 10−7 M, which corresponds to pH and pOH values of:
\[ \begin{align*} \mathrm{pH} &= -\log[H_3O^+] \\[4pt] &= -\log(4.9\times 10^{−7}) \\[4pt] &=6.31 \label{\(\PageIndex{1}\)2} \end{align*}\]
\[ \begin{align*} \mathrm{pOH} &= -\log[OH^-] \\[4pt] &= -\log(4.9\times 10^{−7}) \\[4pt] &=6.31 \label{\(\PageIndex{1}\)3} \end{align*}\]
At this temperature, then, neutral solutions exhibit pH = pOH = 6.31, acidic solutions exhibit pH less than 6.31 and pOH greater than 6.31, whereas basic solutions exhibit pH greater than 6.31 and pOH less than 6.31. This distinction can be important when studying certain processes that occur at nonstandard temperatures, such as enzyme reactions in warm-blooded organisms. Unless otherwise noted, references to pH values are presumed to be those at standard temperature (25 °C) (Table \(\PageIndex{1}\)).
| Classification | Relative Ion Concentrations | pH at 25 °C | pH at 80 °C |
|---|---|---|---|
| acidic | [H3O+] > [OH−] | pH < 7 | pH < 6.31 |
| neutral | [H3O+] = [OH−] | pH = 7 | pH = 6.31 |
| basic | [H3O+] < [OH−] | pH > 7 | pH > 6.31 |
Figure \(\PageIndex{2}\) shows the relationships between [H3O+], [OH−], pH, and pOH, and gives values for these properties at standard temperatures for some common substances.
Example \(\PageIndex{1}\): Calculation of pH from \(\ce{[H_3O^+]}\)
What is the pH of stomach acid, a solution of HCl with a hydronium ion concentration of \(1.2 \times 10^{−3}\; M\)?
Solution
\[ \begin{align*} \mathrm{pH} &= -\log [H_3O^+] \\[4pt] &= -\log(1.2 \times 10^{−3}) \\[4pt] &=−(−2.92) =2.92 \end{align*} \]
Exercise \(\PageIndex{1}\)
Water exposed to air contains carbonic acid, H2CO3, due to the reaction between carbon dioxide and water:
\[\ce{CO}_{2(aq)}+\ce{H_2O}_{(l)} \rightleftharpoons \ce{H_2CO}_{3(aq)}\nonumber\]
Air-saturated water has a hydronium ion concentration caused by the dissolved \(\ce{CO_2}\) of \(2.0 \times 10^{−6}\; M\), about 20-times larger than that of pure water. Calculate the pH of the solution at 25 °C.
- Answer
-
5.70
Example \(\PageIndex{2}\): Calculation of Hydronium Ion Concentration from pH
Calculate the hydronium ion concentration of blood, the pH of which is 7.3.
Solution
\[ \begin{align*} \mathrm{pH} =-\log [\ce{H_3O^+}] &=7.3 \\[4pt] [\ce{H_3O^+}] &=10^{−7.3} \\[4pt] [\ce{H_3O^+}] &=5\times 10^{−8}\;M \end{align*}\]
(On a calculator take the antilog, or the “inverse” log, of −7.3, or calculate 10−7.3.)
Exercise \(\PageIndex{2A}\)
Calculate the hydronium ion concentration of a solution with a pH of −1.07.
- Answer
-
12 M
This uses the definition of pH that we commonly use:
\[pH=-\log[H_3O^+] \nonumber\]
So for this solution:
\[ \begin{align*} \mathrm{pH} =-\log [\ce{H_3O^+}] &= -1.07 \\[4pt] [\ce{H_3O^+}] & = 10^{+1.07} \\[4pt] &= 12\, (\text{with significant figure}) \end{align*}\]
However, at this high concentration, the solution will be non-ideal and we have to use the proper definition in terms of hydronium activities
\[pH=-\log a\{H_3O^+\} \nonumber \]
See this module for more details.
Exercise \(\PageIndex{2B}\)
The ionization constant of water \(\ce{K_w}\) at 37 °C is \(2.42 \times 10^{-14}\). What is the pH for a neutral solution at this human physiological temperature? Is the water acidic, basic or neutral?
- Answer
-
\[ \ce{K_w} = \ce{[H3O^{+}] [OH^{-}]} = 2.42 \times 10^{-14}\nonumber\]
and
\[ \begin{align*} \ce{[H3O^{+}]} = \ce{[OH^{-}]} &= \sqrt{2.42 \times 10^{-14} } \\[4pt] &= 1.55 \times 10^{-7} \end{align*} \]
\[pH=-\log[H_3O^+] = -\log 1.55 \times 10^{-7} = 6.81 \]
If we use the definition of acidic systems like in Figure \(\PageIndex{2}\), then we would (incorrectly) argue the solution is acidic. However, since \(\ce{[OH^{-}]} = \ce{[H3O^{+}]}\), the solution is still neutral. This is only a strange idea, if one ignores the temperature dependence of \(\ce{K_w}\).
Environmental Science
Normal rainwater has a pH between 5 and 6 due to the presence of dissolved CO2 which forms carbonic acid:
\[\ce{H_2O}_{(l)}+\ce{CO}_{2(g)}⟶\ce{H_2CO}_{3(aq)} \label{\(\PageIndex{1}\)4}\]
\[\ce{H_2CO}_{3(aq)} \rightleftharpoons \ce{H^+}_{(aq)} + \ce{HCO^-}_{3(aq)} \label{\(\PageIndex{1}\)5}\]
Acid rain is rainwater that has a pH of less than 5, due to a variety of nonmetal oxides, including CO2, SO2, SO3, NO, and NO2 being dissolved in the water and reacting with it to form not only carbonic acid, but sulfuric acid and nitric acid. The formation and subsequent ionization of sulfuric acid are shown here:
\[\ce{H_2O}_{(l)} + \ce{SO}_{3(g)} ⟶\ce{H_2SO}_{4(aq)} \label{\(\PageIndex{1}\)6}\]
\[\ce{H_2SO}_{4(aq)} ⟶ \ce{H^+}_{(aq)} + \ce{HSO}_{4(aq)}^- \label{\(\PageIndex{1}\)7}\]
Carbon dioxide is naturally present in the atmosphere because we and most other organisms produce it as a waste product of metabolism. Carbon dioxide is also formed when fires release carbon stored in vegetation or when we burn wood or fossil fuels. Sulfur trioxide in the atmosphere is naturally produced by volcanic activity, but it also stems from burning fossil fuels, which have traces of sulfur, and from the process of “roasting” ores of metal sulfides in metal-refining processes. Oxides of nitrogen are formed in internal combustion engines where the high temperatures make it possible for the nitrogen and oxygen in air to chemically combine.

Figure \(\PageIndex{3}\): (a) Acid rain makes trees more susceptible to drought and insect infestation, and depletes nutrients in the soil. (b) It also is corrodes statues that are carved from marble or limestone. (credit a: modification of work by Chris M Morris; credit b: modification of work by “Eden, Janine and Jim”/Flickr)
Acid rain is a particular problem in industrial areas where the products of combustion and smelting are released into the air without being stripped of sulfur and nitrogen oxides. In North America and Europe until the 1980s, it was responsible for the destruction of forests and freshwater lakes, when the acidity of the rain actually killed trees, damaged soil, and made lakes uninhabitable for all but the most acid-tolerant species. Acid rain also corrodes statuary and building facades that are made of marble and limestone (Figure \(\PageIndex{3}\)). Regulations limiting the amount of sulfur and nitrogen oxides that can be released into the atmosphere by industry and automobiles have reduced the severity of acid damage to both natural and manmade environments in North America and Europe. It is now a growing problem in industrial areas of China and India.
The acidity of a solution is typically assessed experimentally by measurement of its pH. The pOH of a solution is not usually measured, as it is easily calculated from an experimentally determined pH value. The pH of a solution can be directly measured using a pH meter (Figure \(\PageIndex{4}\)).

Figure \(\PageIndex{4}\): (a) A research-grade pH meter used in a laboratory can have a resolution of 0.001 pH units, an accuracy of ± 0.002 pH units, and may cost in excess of $1000. (b) A portable pH meter has lower resolution (0.01 pH units), lower accuracy (± 0.2 pH units), and a far lower price tag. (credit b: modification of work by Jacopo Werther).
Example \(\PageIndex{3}\): Calculation of pOH
What are the pOH and the pH of a 0.0125-M solution of potassium hydroxide, KOH at 25 °C?
Solution
Potassium hydroxide is a highly soluble ionic compound and completely dissociates when dissolved in dilute solution, yielding [OH−] = 0.0125 M:
\[\mathrm{pOH=-\log[OH^− ]=-\log 0.0125}\]
\[=−(−1.903)=1.903\]
The pH can be found from the \(\ce{pOH}\):
\[\mathrm{pH+pOH=14.00}\]
\[\mathrm{pH=14.00−pOH=14.00−1.903=12.10}\]
Exercise \(\PageIndex{3}\)
The hydronium ion concentration of vinegar is approximately \(4 \times 10^{−3}\; M\) at 25 °C. What are the corresponding values of pOH and pH?
- Answer
-
pOH = 11.6, pH = 2.4
The pH of a solution may also be visually estimated using colored indicators (Figure \(\PageIndex{5}\)).

Figure \(\PageIndex{5}\): (a) A universal indicator assumes a different color in solutions of different pH values. Thus, it can be added to a solution to determine the pH of the solution. The eight vials each contain a universal indicator and 0.1-M solutions of progressively weaker acids: HCl (pH = l), CH3CO2H (pH = 3), and NH4Cl (pH = 5), deionized water, a neutral substance (pH = 7); and 0.1-M solutions of the progressively stronger bases: KCl (pH = 7), aniline, C6H5NH2 (pH = 9), NH3 (pH = 11), and NaOH (pH = 13). (b) pH paper contains a mixture of indicators that give different colors in solutions of differing pH values. (credit: modification of work by Sahar Atwa).
Summary
The concentration of hydronium ion in a solution of an acid in water is greater than \( 1.0 \times 10^{-7}\; M\) at 25 °C. The concentration of hydroxide ion in a solution of a base in water is greater than \( 1.0 \times 10^{-7}\; M\) at 25 °C. The concentration of H3O+ in a solution can be expressed as the pH of the solution; \(\ce{pH} = -\log \ce{H3O+}\). The concentration of OH− can be expressed as the pOH of the solution: \(\ce{pOH} = -\log[\ce{OH-}]\). In pure water, pH = 7.00 and pOH = 7.00
Key Equations
- \(\ce{pH}=-\log[\ce{H3O+}]\)
- \(\ce{pOH} = -\log[\ce{OH-}]\)
- [H3O+] = 10−pH
- [OH−] = 10−pOH
- pH + pOH = pKw = 14.00 at 25 °C
Glossary
- acidic
- describes a solution in which [H3O+] > [OH−]
- basic
- describes a solution in which [H3O+] < [OH−]
- neutral
- describes a solution in which [H3O+] = [OH−]
- pH
- logarithmic measure of the concentration of hydronium ions in a solution
- pOH
- logarithmic measure of the concentration of hydroxide ions in a solution
Contributors and Attributions
Paul Flowers (University of North Carolina - Pembroke), Klaus Theopold (University of Delaware) and Richard Langley (Stephen F. Austin State University) with contributing authors. Textbook content produced by OpenStax College is licensed under a Creative Commons Attribution License 4.0 license. Download for free at http://cnx.org/contents/85abf193-2bd...a7ac8df6@9.110).

