2.4: Question 2.E.58 PASS - electron configuration, orbital diagrams, Aufbau's Principle Hund's Rule
- Page ID
- 452270
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Consider magnesium (Mg), silicon (Si), and sulfur (S).
Which of the following atoms has two unpaired electrons?
a) Mg
b) Si
c) S
d) Both Mg and S
e) Both Si and S
- Answer
-
e) Both Si and S
- Strategy Map
-
Step Hint 1. Find your element on the Periodic Table and note its atomic number
The atomic number (Z) describes the number of electrons the atom has
Recall (LibreText section 3.1): The Periodic Table
2. Create an orbital box diagram for each elements electrons Orbital diagrams are pictorial representations of the electron configuration, showing the individual orbitals as boxes and the pairing arrangement of electrons as arrows
Begin to fill your box orbital diagram starting with the lowest energy '1s' orbital and stop when you “run out” of electrons
- Follow Hund's Rule and Aufbau Principle
3. Count the number of unpaired electrons Any orbitals containing one single electron are described as unpaired electrons
- Solution
-
Magnesium (Mg): 12 electrons
![Empty box orbital diagram for Mg boxes from left to right increasing energy [1s] [2s] [2p][2p][2p] [3s]](https://chem.libretexts.org/@api/deki/files/457112/correctMg-EmptyBoxOrbitals.png?revision=1&size=bestfit&width=416&height=143)
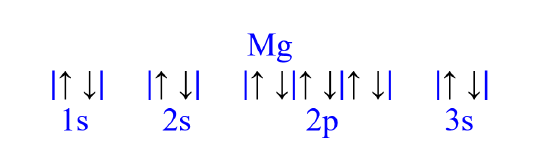
Mg has no unpaired electrons
Silicon (Si): 14 electrons
![Empty box orbital diagram for Si boxes from left to right increasing energy [1s]-[2s]-[2p][2p][2p]-[3s]-[3p][3p][3p]](https://chem.libretexts.org/@api/deki/files/457114/correctSi-EmptyBoxOrbitals.png?revision=1&size=bestfit&width=481&height=130)

Silicon has two unpaired electrons
Sulfur (S): 16 electrons
![Empty box orbital diagram for S boxes from left to right increasing energy [1s]-[2s]-[2p][2p][2p]-[3s]-[3p][3p][3p]](https://chem.libretexts.org/@api/deki/files/457116/correctS-EmptyBoxOrbitals.png?revision=1&size=bestfit&width=469&height=127)

Sulfur has 2 unpaired electrons
Answer
e) Both Si and S
- Guided Solution
-
Guided Solution Hint This problem relies on concepts from Electronic Structure of Atoms (Electron Configurations)
Refer to LibreText 2.6, Electronic Structure of Atoms (Electron Configurations)
What does the question ask you to find?
Question
Consider magnesium (Mg), silicon (Si), and sulfur (S).
Which of the following atoms has two unpaired electrons?
Consider the circumstances in which electrons are paired or unpaired
- Aufbau Principle: Fill electrons into the lowest available energy orbital
- Paired electrons: If the lowest energy orbital available already has one electron, you will add a second electron to it with an opposite spin
- represented by an arrow pointed in the opposite direction
- Paired electrons: If the lowest energy orbital available already has one electron, you will add a second electron to it with an opposite spin
- Hund's Rule: when degenerate (same energy) orbitals are available, maximize the number of unpaired electrons with the same spin
- Unpaired electrons: pay attention when filling the first, second, and third electrons into degenerate p-orbitals
How many electrons does each element have?
Mg: 12 electrons
Si: 14 electrons
S: 16 electrons
Find the atomic number (Z) on the Periodic Table to determine the number of electrons for each element
Complete a Box Orbital diagram to determine the number of unpaired electrons for each element
![Empty box orbital diagram for Mg boxes from left to right increasing energy [1s] [2s] [2p][2p][2p] [3s]](https://chem.libretexts.org/@api/deki/files/457112/correctMg-EmptyBoxOrbitals.png?revision=1&size=bestfit&width=416&height=143)
![Empty box orbital diagram for Si boxes from left to right increasing energy [1s]-[2s]-[2p][2p][2p]-[3s]-[3p][3p][3p]](https://chem.libretexts.org/@api/deki/files/457114/correctSi-EmptyBoxOrbitals.png?revision=1)
![Empty box orbital diagram for S boxes from left to right increasing energy [1s]-[2s]-[2p][2p][2p]-[3s]-[3p][3p][3p]](https://chem.libretexts.org/@api/deki/files/457116/correctS-EmptyBoxOrbitals.png?revision=1&size=bestfit&width=469&height=127)
Common mistake: having an even number of electrons does not always mean that they are all paired!
The orbitals in the box diagram are arranged from left to right in order of increasing energy
Fill the appropriate number of electrons into each box diagram
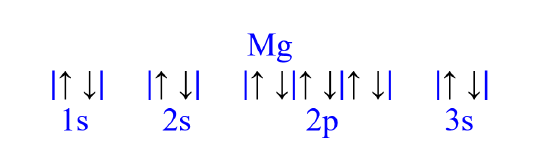


Add electrons from left to right
- maximize the number of electrons in each lowest energy available orbital before moving on
- each orbital can hold a maximum of two electrons
- the two electrons must have opposite spin (pointed up and down)
- if degenerate empty orbitals are available, fill those first with unpaired electrons with the same spin before pairing electrons
See LibreText Example 2.6.1: Quantum Numbers and Electron Configurations
Complete Solution:
Mg
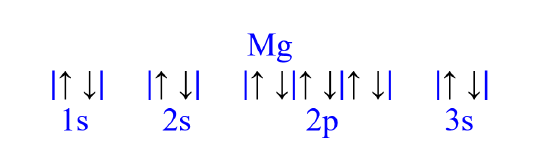
Magnesium, atomic number 12, has a total of twelve electrons. The diagram is filled starting with a pair of electrons in the 1s orbital box, followed by 2s, the 2p then 3s. All electrons are paired in magnesium.
Si

Silicon, atomic number 14, has a total of fourteen electrons. The diagram is filled starting with the 1s orbital box, followed by 2s, 2p, 3s then 3p. The 3p orbital has 2/6 electrons, meaning silicon has two unpaired electrons.
S

Sulfur, atomic number 16, has a total of sixteen electrons. The diagram is filled starting with the 1s orbital box, followed by 2s, 2p, 3s then 3p. The 3p orbital has 4/6 electrons, meaning sulfur has two unpaired electrons.
When filling degenerate (same enegy) orbitals, Hund's Rule must be obeyed to maintain the most stable (lowest energy) electron configuration.
Hund's Rule is shown below:

Check your work!
When completing the box orbital diagram, count the number of arrows; be sure it matches the number of electrons determined from the atomic number!
(question source adapted question 6.4.14 from 6.E: Electronic Structure and Periodic Properties (Exercises): https://chem.libretexts.org/Bookshelves/General_Chemistry/Chemistry_1e_(OpenSTAX)/06%3A_Electronic_Structure_and_Periodic_Properties_of_Elements/6.E%3A_Electronic_Structure_and_Periodic_Properties_(Exercises), shared under a CC BY 4.0 license, authored, remixed, and/or curated by OpenStax, original source question 59 https://openstax.org/books/chemistry-2e/pages/6-exercises, Access for free at https://openstax.org/books/chemistry/pages/1-introduction)
- Aufbau Principle: Fill electrons into the lowest available energy orbital


