The Robinson Annulation
Many times the product of a Michael addition produces a dicarbonyl which can then undergo an intramolecular aldol reaction. These two processes together in one reaction creates two new carbon-carbon bonds and also creates a ring. Ring-forming reactions are called annulations after the Latin work for ring annulus. The reaction is named after English chemist Sir Robert Robinson (1886-1975) who developed it. He received the Nobel prize in chemistry in 1947. Remember that during annulations five and six membered rings are preferred.
The nucleophilic enolate donor is typically an enolate ion or enamine of a cyclic ketone, β-keto ester or β-diketone. The electrophilic acceptor is usually an α, β-unsaturated ketone. In the example below, 2-methyl-1,3-cyclohexanedione is deprotonated to form an enolate which affects a Michael reaction addition on 3-buten-2-one forming a C-C bond. The product contains a 1,5-diketone fragment which can undergo an intramolecular aldol condensation. This occurs through creation of a new enolate at the methyl ketone which undergoes an intramolecular aldol reaction. A new C-C bond is formed to one of the ring carbonyls, creating a new six-membered ring. In the last step, the resulting alcohol is eliminated to form a α, β-unsaturated ketone as the final Robinson annulation product. Because the Robinson Annulation involves an aldol reaction, a full equivalent of base is required.
Example

Worked Example
Draw the product of the following Robinson Annulation.

- Answer
-
Analysis: When considering the product of a Robinson annulation it is usually best to consider each reaction individually. Use the steps discussed in Section 23.10 to convert the starting materials into the product of a Michael reaction, then into the product of an intramolecular aldol condensation. If the starting materials are drawn in a skeletal structure it may be helpful to convert to a condensed formula to better keep track of carbons and hydrogens.
Formation of the Michael reaction product

Formation of the intramolecular Aldol condensation product

Planning a Synthesis Using a Robinson Annulation
The presence of a cyclic, six-membered α, β-unsaturated ketone in a target molecule suggest that a Robinson Annulation may be utilized in its synthesis. The key bond cleavages are the C=C bond of the α, β-unsaturated ketone and the C-C bond between carbons in the a and g on the opposite alkyl chain of the ketone.

Worked Example
What would be the starting materials used if the following molecule was made using a Robinson annulation?

- Answer
-
Analysis
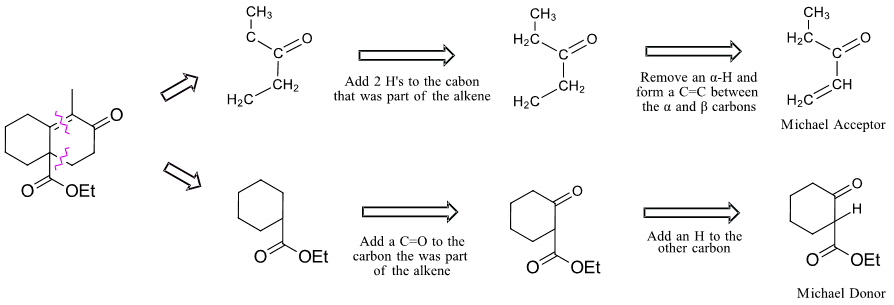
Solution
It is necessary to use sodium ethoxide as the reaction base due to the presence of an ester.

Exercise \(\PageIndex{1}\)
1) Provide products of the following Robinson annulations.
a)

b)

c)

2) What would be the starting materials required to make the following molecule using a Robinson annulation?

- Answers
-
1)
a)

b)

c)

2)



