9.2: Common nucleophilic substitution reactions
- Page ID
- 225809
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Most of this chapter focuses on specific reagents and conditions for performing nucleophilic substitutions in the lab or plant. Remember some of the main lessons from the previous chapter: Higher temperatures favor elimination (where this can happen) and colder conditions favor SN2 (as long as the reactants can still react at that temperature). For SN2, you will normally want to use a polar aprotic solvent (DMSO is my favorite).
All of these reactions fall into a common pattern of electrophile + nucleophile gives substitution product. You may see either reactant – electrophile or nucleophile – given above the arrow as a “reagent.” Consider the following:
The electrophile
Most often this has an alkyl halide or sulfonate serving as “RX”. An alkyl bromide is probably the most useful for you to know; it’s stable, fairly cheap, easy to make using PBr3 (see section 9.3. which shows how to make all different RX). Remember that a primary or secondary alkyl halide will work best with SN2, and for a tertiary alkyl halide you will have to use SN1 methods. Most of the reactions that are effective in synthesis involve SN2, because these are usually the cleanest, especially with primary alkyl halides.
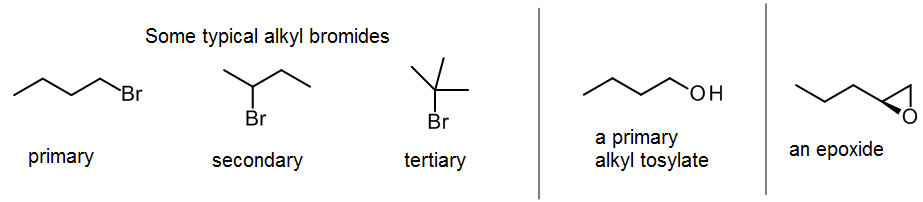
In section 9.6, we’ll learn about an alternative electrophile, the epoxide or oxirane. Epoxides are ethers that contain a strained three-membered ring that makes them quite reactive towards nucleophiles. The incoming group displaces the oxygen from one of the carbons (usually the less substituted one), leaving that oxygen attached to the other (neighboring) carbon.
The nucleophile
A wide variety of nucleophiles is available to react with alkyl halides. Some of the most common are shown below, using bromobutane as a typical primary alkyl halide. The nucleophile is shown on the far left. Note that most nucleophiles (except amines/NH3) are anions, but the actual reagents you use are usually sodium or potassium salts, which are always ionic. For example, when you see –OH in a scheme this will usually mean NaOH or KOH.
Preparation of alcohols
Hydroxide ion is very effective for primary and secondary alcohols. For tertiary alcohols, you have to use water to avoid elimination.

Preparation of ethers
The Williamson ether synthesis (see section 9.5) uses alkoxide ion with primary and secondary alkyl halides. For making tertiary ethers, you have to use ROH to avoid elimination.

Preparation of amines
See section 9.4. Amination of alkyl halides can be done using NH3, RNH2 or R2NH, but a very large excess of the nitrogen compound must be used to avoid mixtures of products. A cleaner reaction can be done using azide ion (N3–) followed by reduction.
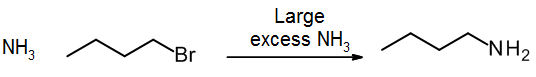
Preparation of alkyl iodides
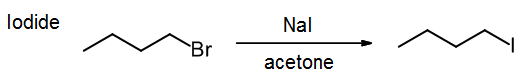
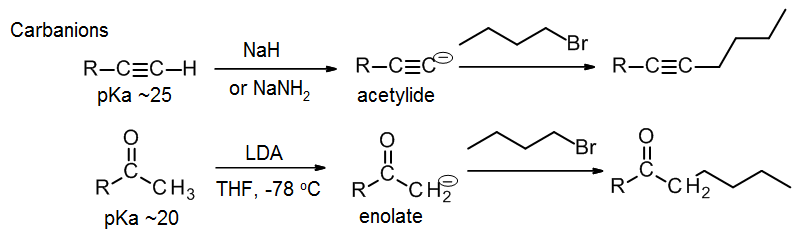
Reactions using carbanions or carbanion equivalents
These are useful for making new C-C connections. See section 9.8. for reactions of acetylides, which in two steps turn a terminal alkyne into a longer chain internal alkyne. A strong base (NaH or NaNH2) is used to make the carbanion nucleophile (the acetylide), which is then reacted with the alkyl halide. Only primary alkyl halides work.
See section 9.7 for reactions of enolates. Here a strong base (LDA) is used to make the carbanion nucleophiile (an enolate) from a ketone or similar compound. This reacts with the alkyl halide to produce a new ketone with a longer carbon chain. Again, only primary alkyl halides work.
We will now examine some of these reactions in more detail.
References
Joel Karty, “Organic Chemistry: Principles and Mechanisms,” First Edition, Norton.
- Authored by: Martin Walker. Provided by: SUNY Potsdam. Located at: http://directory.potsdam.edu/?function=user=walkerma. License: CC BY-SA: Attribution-ShareAlike

