3.2: Nomenclature of unsaturated hydrocarbons
- Page ID
- 225772
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Alkenes
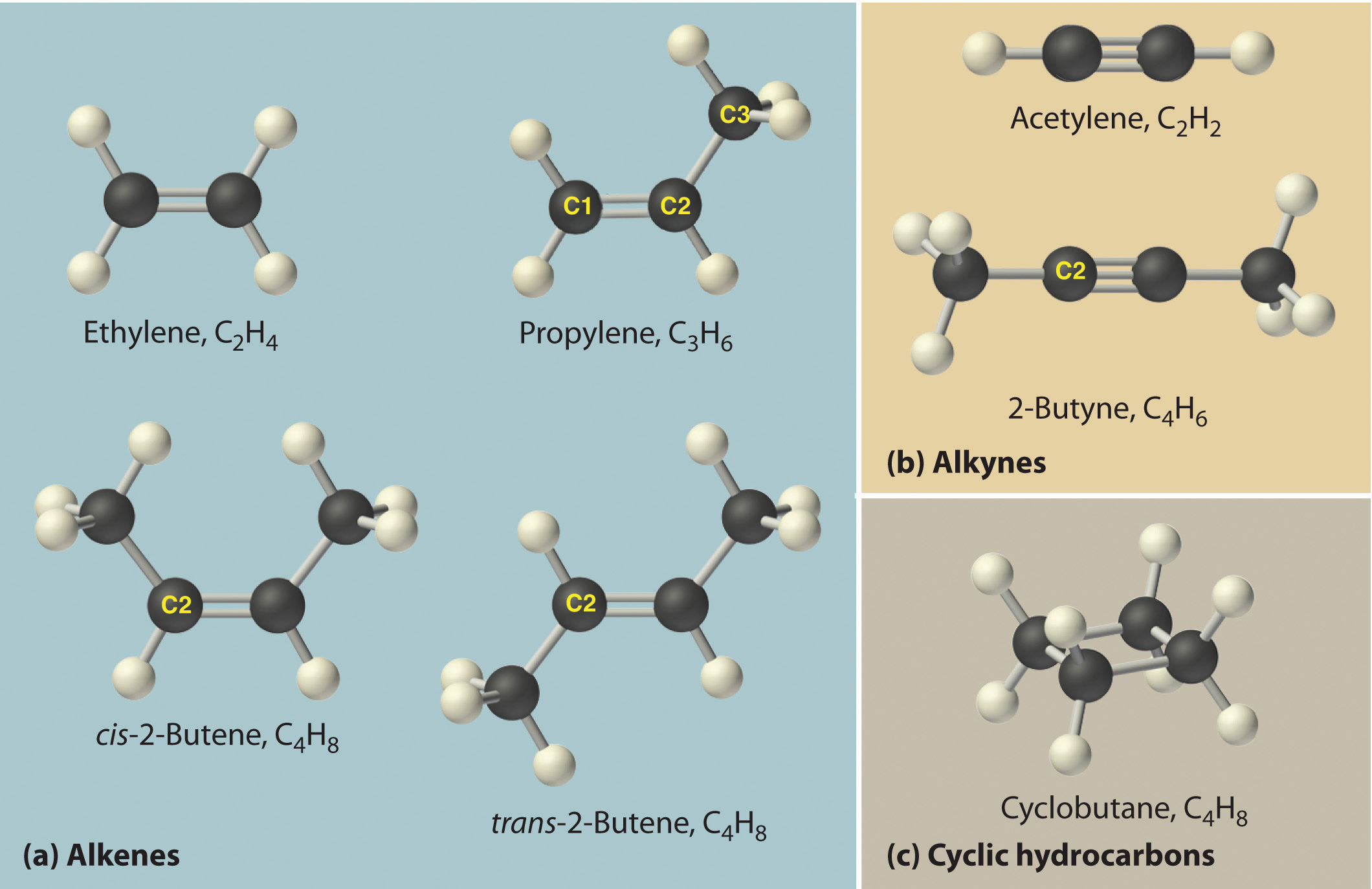
The simplest alkenes are ethene (traditionally called ethylene), C2H4 or CH2=CH2, and propene (or propylene), C3H6 or CH3CH=CH2 (part (a) in the figure below). The names of alkenes that have more than three carbon atoms use the same stems as the names of the alkanes (see table above, “The First 10 Straight-Chain Alkanes”) but end in –ene instead of –ane.
As with alkanes, more than one structure is possible for alkenes with four or more carbon atoms. For example, an alkene with four carbon atoms has three possible structures. One is CH2=CHCH2CH3 (1-butene), which has the double bond between the first and second carbon atoms in the chain. The other two structures have the double bond between the second and third carbon atoms and are forms of CH3CH=CHCH3 ((but-2-ene, formerly called 2-butene). All four carbon atoms in but-2-ene lie in the same plane, so there are two possible structures (part (a) in Figure 3.7.2). If the two methyl groups are on the same side of the double bond, the compound is cis-but-2-ene or cis-2-butene (from the Latin cis, meaning “on the same side”). If the two methyl groups are on opposite sides of the double bond, the compound is trans-but-2-ene or trans-2-butene (from the Latin trans, meaning “across”). (NB: We will learn more complete naming for such isomers in section 4.3.) These are distinctly different molecules: cis-but-2-ene melts at −138.9°C, whereas trans-but-2-ene melts at −105.5°C.
Just as a number indicates the positions of branches in an alkane, the number in the name of an alkene specifies the position of the first carbon atom of the double bond. The name is based on the lowest possible number starting from either end of the carbon chain, so CH3CH2CH=CH2 is called but-1-ene (formerly 1-butene), not but-3-ene. Note that CH2=CHCH2CH3 and CH3CH2CH=CH2 are different ways of writing the same molecule (but-1-ene) in two different orientations.
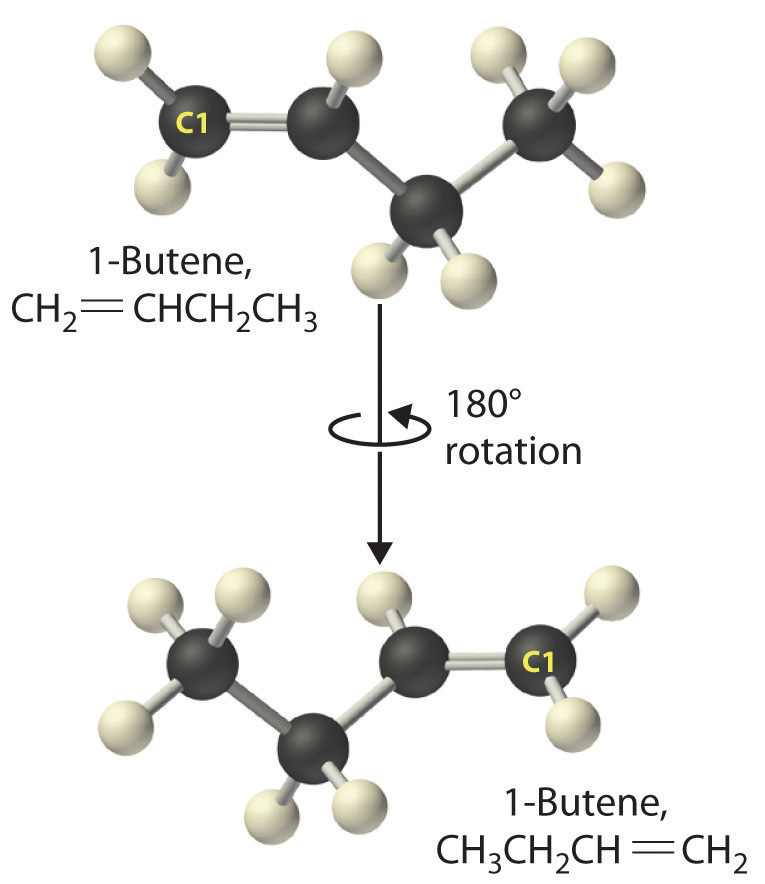
The name of a compound does not depend on its orientation. As illustrated for 1-butene, both condensed structural formulas and molecular models show different orientations of the same molecule. It is important to be able to recognize the same structure no matter what its orientation.
Note
The positions of groups or multiple bonds are always indicated by the lowest number possible.
Alkynes
The simplest alkyne is ethyne, traditionally called acetylene, C2H2 or HC≡CH (part (b) in the figure above). Because a mixture of acetylene and oxygen burns with a flame that is hot enough (>3000°C) to cut metals such as hardened steel, acetylene is widely used in cutting and welding torches. The names of other alkynes are similar to those of the corresponding alkanes but end in –yne. For example, HC≡CCH3 is propyne, and CH3C≡CCH3 is but-2-yne because the multiple bond begins on the second carbon atom.
Note
The number of bonds between carbon atoms in a hydrocarbon is indicated in the suffix:
- alkane: only carbon–carbon single bonds
- alkene: at least one carbon–carbon double bond
- alkyne: at least one carbon–carbon triple bond
Aromatic hydrocarbons
Alkanes, alkenes, alkynes, and cyclic hydrocarbons are generally called aliphatic hydrocarbons. The name comes from the Greek aleiphar, meaning “oil,” because the first examples were extracted from animal fats. In contrast, the first examples of aromatic hydrocarbons, also called arenes, were obtained by the distillation and degradation of highly scented (thus aromatic) resins from tropical trees.
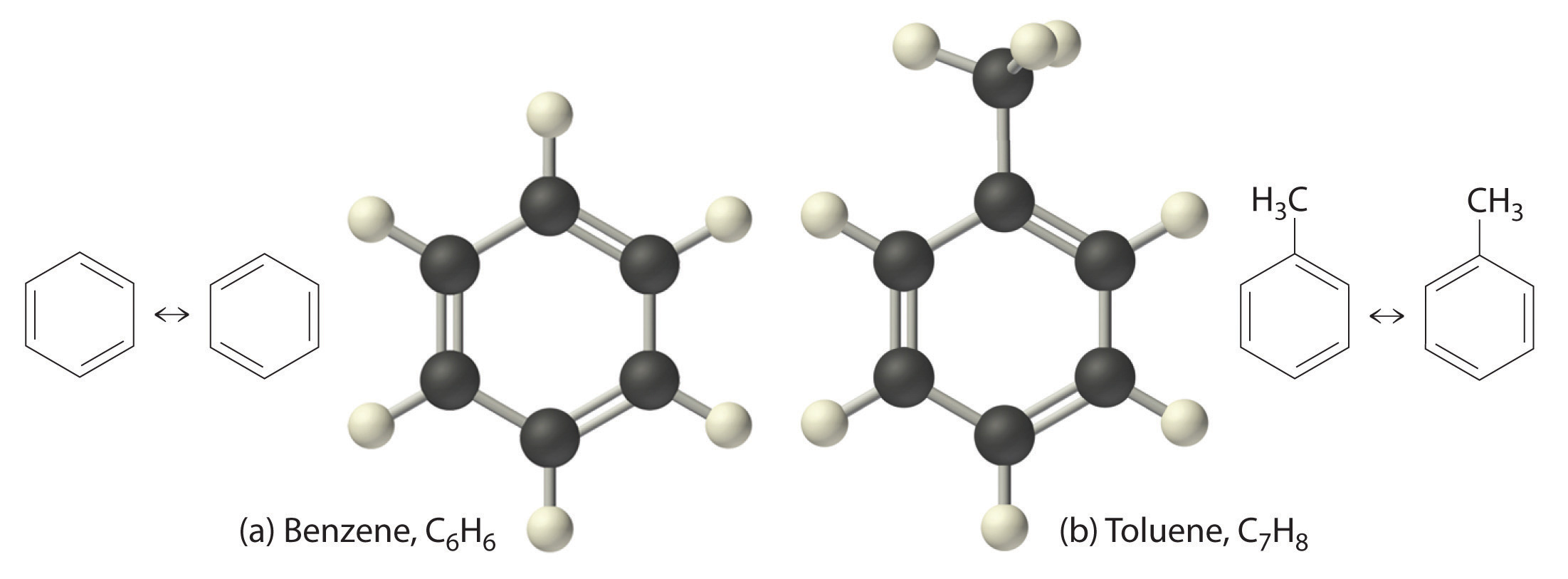
The simplest aromatic hydrocarbon is benzene (C6H6), which was first obtained from a coal distillate. The word aromatic now refers to benzene and structurally similar compounds. As shown in part (a) in Figure 3.7.4, it is possible to draw the structure of benzene in two different but equivalent ways, depending on which carbon atoms are connected by double bonds or single bonds. Toluene is similar to benzene, except that one hydrogen atom is replaced by a –CH3 group; it has the formula C7H8 (part (b) in Figure 3.7.4). The chemical behavior of aromatic compounds differs from the behavior of aliphatic compounds. Benzene and toluene are found in gasoline, and benzene is the starting material for preparing substances as diverse as aspirin and nylon.
Figure 3.7.5 illustrates two of the molecular structures possible for hydrocarbons that have six carbon atoms. As shown, compounds with the same molecular formula can have very different structures.

Example 3.7.1
Write the condensed structural formula for each hydrocarbon.
- n-heptane
- 2-pentene
- 2-butyne
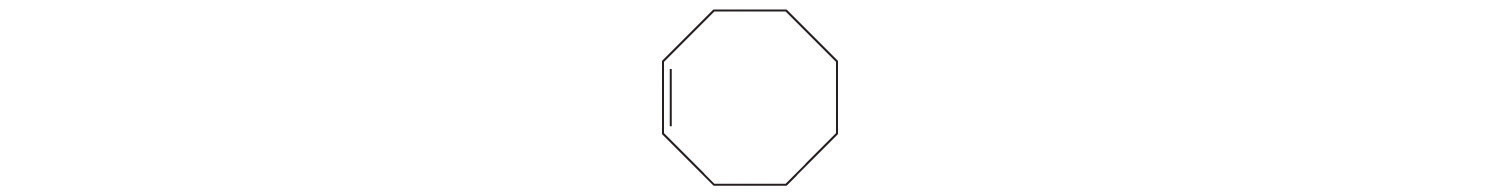
- cyclooctene (give the skeletal formula)
Given: name of hydrocarbon
Asked for: condensed structural formula
Strategy:
- Use the prefix to determine the number of carbon atoms in the molecule and whether it is cyclic. From the suffix, determine whether multiple bonds are present.
- Identify the position of any multiple bonds from the number(s) in the name and then write the condensed structural formula.
[reveal-answer q=”86147″]Show Solution[/reveal-answer]
[hidden-answer a=”86147″]
a. A The prefix hept- tells us that this hydrocarbon has seven carbon atoms, and n- indicates that the carbon atoms form a straight chain. The suffix -ane tells that it is an alkane, with no carbon–carbon double or triple bonds. B The condensed structural formula is CH3CH2CH2CH2CH2CH2CH3, which can also be written as $$CH_3(CH_2)_5CH_3$$.
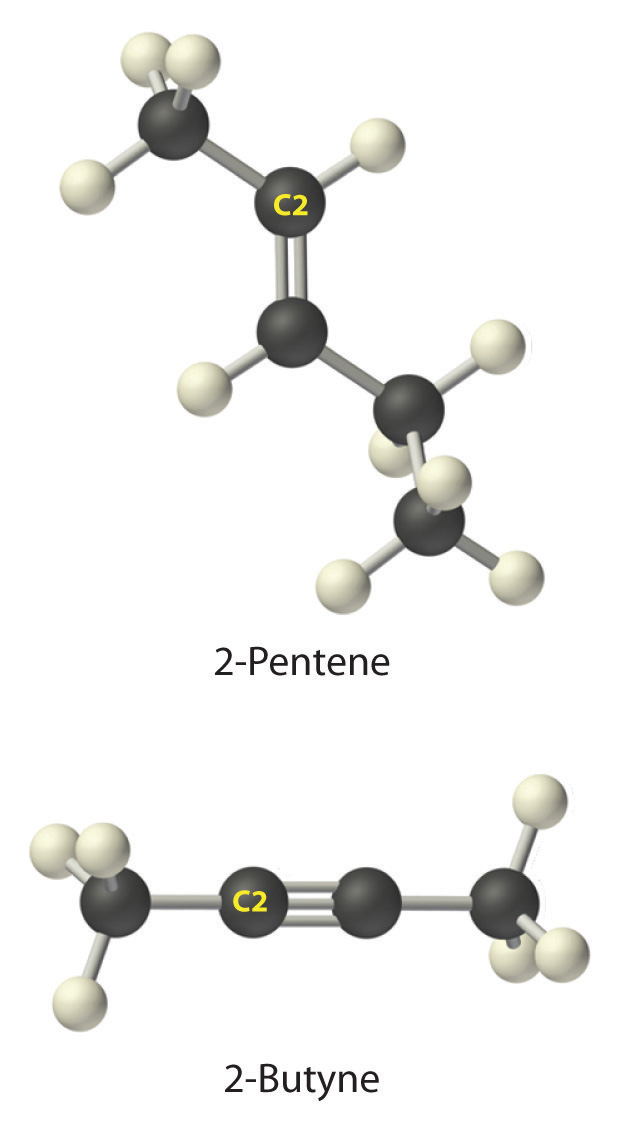
b. A The prefix pent- tells us that this hydrocarbon has five carbon atoms, and the suffix -ene indicates that it is an alkene, with a carbon–carbon double bond. B The 2- tells us that the double bond begins on the second carbon of the five-carbon atom chain. The condensed structural formula of the compound is therefore CH3CH=CHCH2CH3.
c. A The prefix but- tells us that the compound has a chain of four carbon atoms, and the suffix -yne indicates that it has a carbon–carbon triple bond. B The 2- tells us that the triple bond begins on the second carbon of the four-carbon atom chain. So the condensed structural formula for the compound is CH3C≡CCH3.
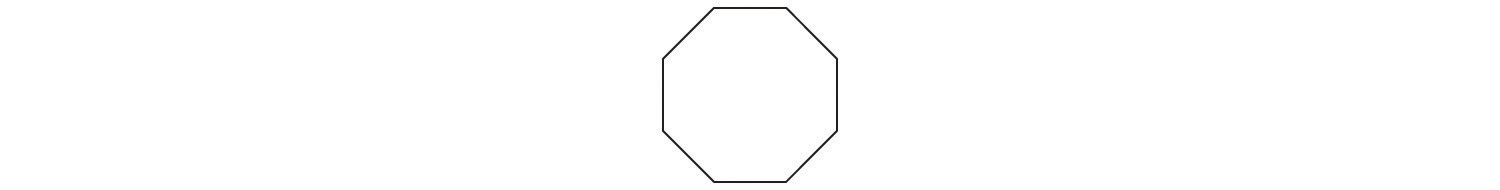
d. A The prefix cyclo- tells us that this hydrocarbon has a ring structure, and oct- indicates that it contains eight carbon atoms, which we can draw as
The suffix -ene tells us that the compound contains a carbon–carbon double bond, but where in the ring do we place the double bond? B Because all eight carbon atoms are identical, it doesn’t matter. We can draw the structure of cyclooctene as
Exercise
Write the condensed structural formula for each hydrocarbon.
- n-octane
- 2-hexene
- 1-heptyne
- cyclopentane (give the skeletal formula)
[reveal-answer q=”895946″]Show Answer[/reveal-answer]
[hidden-answer a=”895946″]
- CH3(CH2)6CH3
- CH3CH=CHCH2CH2CH3
- HC≡C(CH2)4CH3
-

[/hidden-answer]
The general name for a group of atoms derived from an alkane is an alkyl group. The name of an alkyl group is derived from the name of the alkane by adding the suffix –yl. Thus the –CH3 fragment is a methyl group, the –CH2CH3 fragment is an ethyl group, and so forth, where the dash represents a single bond to some other atom or group. Similarly, groups of atoms derived from aromatic hydrocarbons are aryl groups, which sometimes have unexpected names. For example, the –C6H5 fragment is derived from benzene, but it is called a phenyl group. In general formulas and structures, alkyl and aryl groups are often abbreviated as R.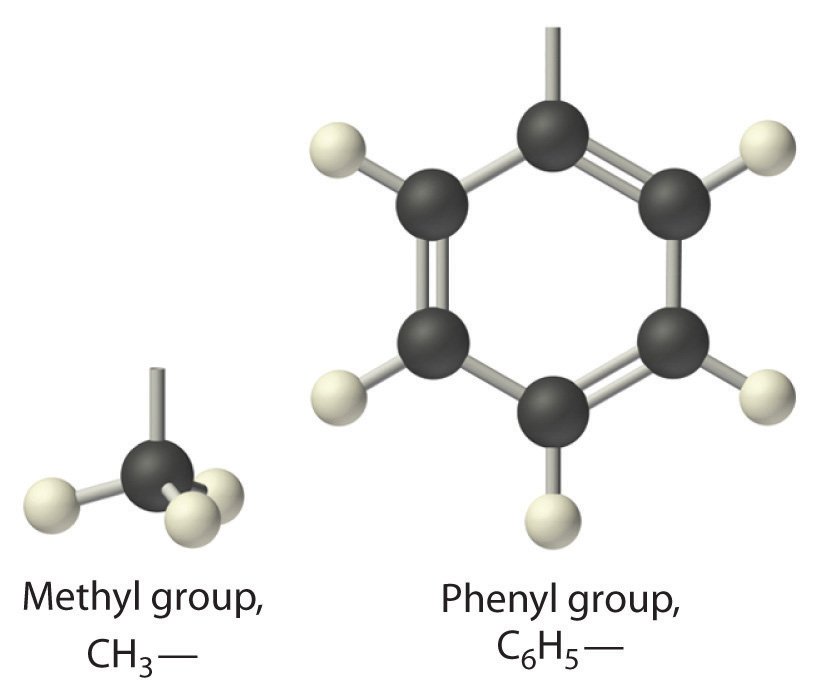
Structures of alkyl and aryl groups. The methyl group is an example of an alkyl group, and the phenyl group is an example of an aryl group.
Summary
The simplest organic compounds are the hydrocarbons, which contain only carbon and hydrogen. Alkanes contain only carbon–hydrogen and carbon–carbon single bonds, alkenes contain at least one carbon–carbon double bond, and alkynes contain one or more carbon–carbon triple bonds. Hydrocarbons can also be cyclic, with the ends of the chain connected to form a ring. Collectively, alkanes, alkenes, and alkynes are called aliphatic hydrocarbons. Aromatic hydrocarbons, or arenes, are another important class of hydrocarbons that contain rings of carbon atoms related to the structure of benzene (C6H6). A derivative of an alkane or an arene from which one hydrogen atom has been removed is called an alkyl group or an aryl group, respectively.
- 3.7: Names of Formulas of Organic Compounds. Located at: https://chem.libretexts.org/Textbook_Maps/General_Chemistry/Map%3A_General_Chemistry_(Petrucci_et_al.)/03%3A_Chemical_Compounds/3.7%3A__Names_of_Formulas_of_Organic_Compounds. Project: Chemistry LibreTexts. License: CC BY-NC-SA: Attribution-NonCommercial-ShareAlike