9.7: Other Alternative Renewable Energy Sources
- Page ID
- 189508
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)5.7.1: Hydrogen
Hydrogen gas may be an important clean fuel of the future. Hydrogen is considered an energy carrier, like electricity and batteries, it carries energy that can be converted for use later. Hydrogen gas does not tend to exist freely but rather hydrogen atoms bind to other atoms and molecules becoming incorporated in everything from water to organic compounds. Therefore, to obtain hydrogen gas for fuel, energy is needed to force these substances to release their hydrogen atoms. One such procedure is known as electrolysis in which an electric current is passed through water to decompose the water molecule into oxygen and hydrogen (Figure \(\PageIndex{1}\)). Hydrogen can also be produced from hydrocarbons such as natural gas and coal, fermentation of plant waste material, and using algae.
The nation of Iceland is attempting to become the first hydrogen-based energy economy using its abundant geothermal energy resources. Some energy experts believe that combining hydrogen fuel and electricity could serve as a basis for a clean, safe, and energy efficient energy system. Electricity generated from intermittent renewable sources such as wind and solar can be used to produce hydrogen fuel for fuel cells that would then generate electricity to power vehicles, computers, heat homes and many other uses. An energy system based on hydrogen would alleviate dependence on foreign fuels and help fight climate change. Hydrogen is the most abundant element in the universe and we will never run out of it.
.png?revision=1&size=bestfit&width=685&height=238) Figure \(\PageIndex{1}\): a) An electric current passed through water resulting in the separation of hydrogen atoms from oxygen to produce the hydrogen fuel. b) An electrolyser used in the production of hydrogen by CambridgeBayWeather. After production the hydrogen is used to inflate weather balloons to which a radiosonde is attached. The oxygen produced is vented to the outside.
Figure \(\PageIndex{1}\): a) An electric current passed through water resulting in the separation of hydrogen atoms from oxygen to produce the hydrogen fuel. b) An electrolyser used in the production of hydrogen by CambridgeBayWeather. After production the hydrogen is used to inflate weather balloons to which a radiosonde is attached. The oxygen produced is vented to the outside.
Fuel Cells
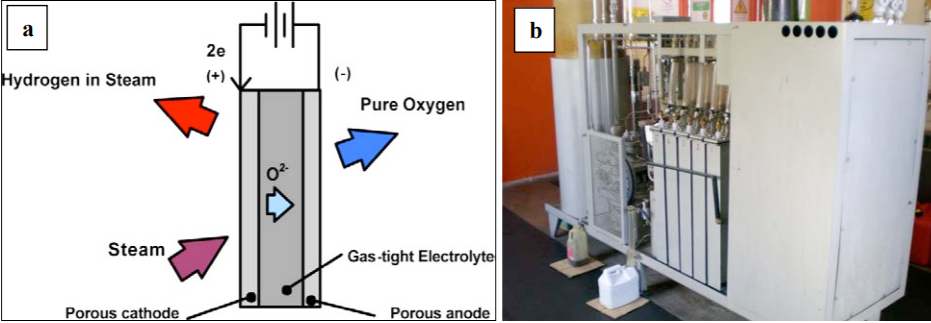
Fuel cells are highly efficient power plants that produce electricity using hydrogen fuel in a chemical reaction that is a reverse of the electrolysis process that produced the hydrogen fuel (Figure \(\PageIndex{2}\)). Energy is released by an exothermic electrochemical reaction that combines hydrogen and oxygen ions through an electrolyte material to generate electricity and heat. Experimental fuel cells that can power automobiles have been developed (Figure \(\PageIndex{2}\)).
.png?revision=1&size=bestfit&width=716&height=292) Figure \(\PageIndex{2}\): The electrochemical processes showing how hydrogen fuel is combined with oxygen generating heat energy along with water as a waste product. Some vehicles that use the fuel cell include buses (photo is a hydrogen fuel bus on Tower Bridge London, photo by Sludge G.), cars (The Toyota Fine N car based on fuel cell technology, photo by Chris 73, CC BY-SA 3.0) and motorcycles (Suzuki Burgman Fuel Cell cutaway model shown here, Photo by Mario CC BY-SA 3.0).
Figure \(\PageIndex{2}\): The electrochemical processes showing how hydrogen fuel is combined with oxygen generating heat energy along with water as a waste product. Some vehicles that use the fuel cell include buses (photo is a hydrogen fuel bus on Tower Bridge London, photo by Sludge G.), cars (The Toyota Fine N car based on fuel cell technology, photo by Chris 73, CC BY-SA 3.0) and motorcycles (Suzuki Burgman Fuel Cell cutaway model shown here, Photo by Mario CC BY-SA 3.0).
Challenges of Hydrogen
Currently, the infrastructure for using hydrogen fuel is lacking and converting a nation such as the United States to hydrogen would require massive and costly development of facilities to produce, store, transport, and provide the fuel. The environmental impact of hydrogen production itself depends on the source of material used to supply the hydrogen. For example, biomass and fossil fuel sources result in carbon-based emissions. Some research suggests that leakage of hydrogen from its production, transport, and use could potentially deplete stratospheric ozone. Research into this is still ongoing.
5.7.2: Electric cars
Electric cars are vehicles that solely depend on electricity to work. They are charged using fossil fuel energy sources (Figure \(\PageIndex{3}\) a), but they are very efficient. Although it might seem like science fiction the electric car is already here, and has been here for the last hundred years. The main problem with the electric car is that it cannot go very far before it needs to be recharged, something that takes between three to six hours. Different models have been explored (Figure \(\PageIndex{3}\)b), but for now most cars are equipped as a hybrid with a back up reservoir using fossil fuel.
.png?revision=1) Figure 5.7.3: a) This REVAi, an electric car, gets recharged on a street in London. (credit: Frank Hebbert). b) 1985 Mercedes-Benz/Alpha Real "Tour de Sol" Solarmobile; built for the 1985 Tour de Sol Centennial Rally, solar car race from Lake Constance to Lake Geneva.
Figure 5.7.3: a) This REVAi, an electric car, gets recharged on a street in London. (credit: Frank Hebbert). b) 1985 Mercedes-Benz/Alpha Real "Tour de Sol" Solarmobile; built for the 1985 Tour de Sol Centennial Rally, solar car race from Lake Constance to Lake Geneva.

